APES Chp 15 Air Pollution and Stratospheric Ozone Depletion Notes
Air Pollution
Definition: Introduction of chemicals, particulate matter, or microorganisms into the atmosphere at concentrations harmful to plants, animals, humans, and the environment.
Sources:
Natural: Volcanoes, vegetation (e.g., tree leaves), forest fires.
Anthropogenic (Human-made): Automobiles, airplanes, factories.
Global System: Air pollution results from various sources and impacts the atmosphere, biosphere, vegetation, and soil.
Natural Sources of Air Pollution
Types:
Volcanoes
Lightning
Forest Fires
Vegetation
Example: The Great Smoky Mountains, named for visibility issues due to natural pollutants.
Anthropogenic Sources of Air Pollution
Types:
On-road vehicles (including off-road vehicles, trains, planes, boats)
Power plants
Industrial processes
Waste disposal
Major Air Pollutants
Sulfur Dioxide (SO₂)
Source: Combustion of fuels (coal, oil).
Nitrogen Oxides (NOx)
Source: Vehicles, fossil fuel combustion, lightning, forest fires.
Carbon Oxides (CO, CO₂)
Source: Vehicle exhaust and respiration.
Particulate Matter (PM)
Source: Combustion of wood, biofuels, etc.
PM10 and PM 2.5
Ozone (O₃)
Source: Reaction of sunlight with VOCs and NOx.
Volatile Organic Compounds (VOCs)
Source: Gasoline, paints, solvents.
stay in atm for a long time
Lead (Pb)
Source: Previously found in paint and gasoline.
Mercury (Hg)
Source: Coal and oil combustion.
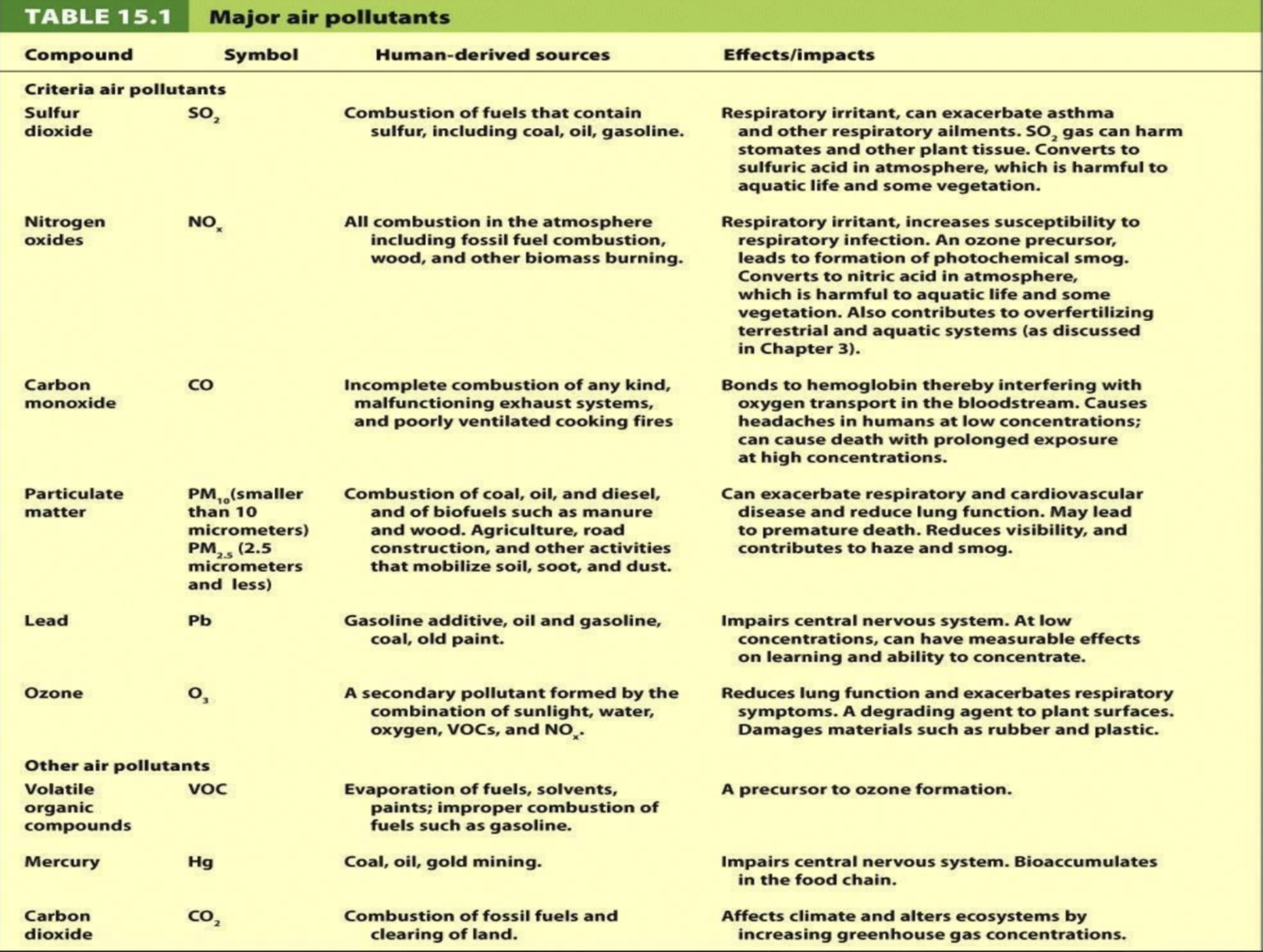
Primary vs. Secondary Pollutants
Primary Pollutants: Directly emitted (e.g., SO₂, CO, NOx, PM).
Secondary Pollutants: Formed from chemical reactions (e.g., ozone).
Formation Factors: Sunlight, water, and temperatures enhance secondary pollutant formation.
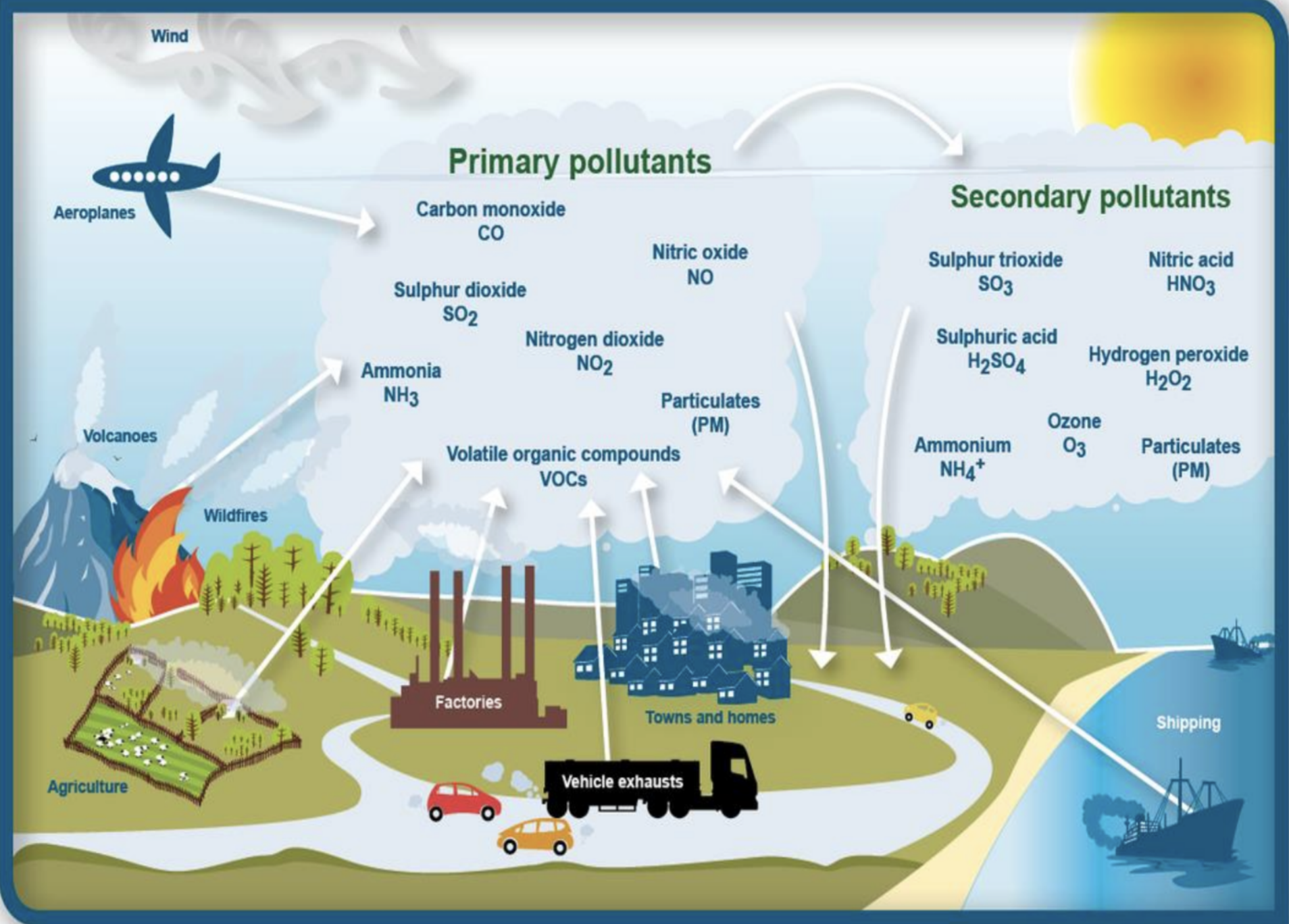
Smog
Definition: A mix of smoke, fog, and sulfur dioxide.
Types: Photochemical smog is formed by sunlight-based reactions, notably in urban areas.
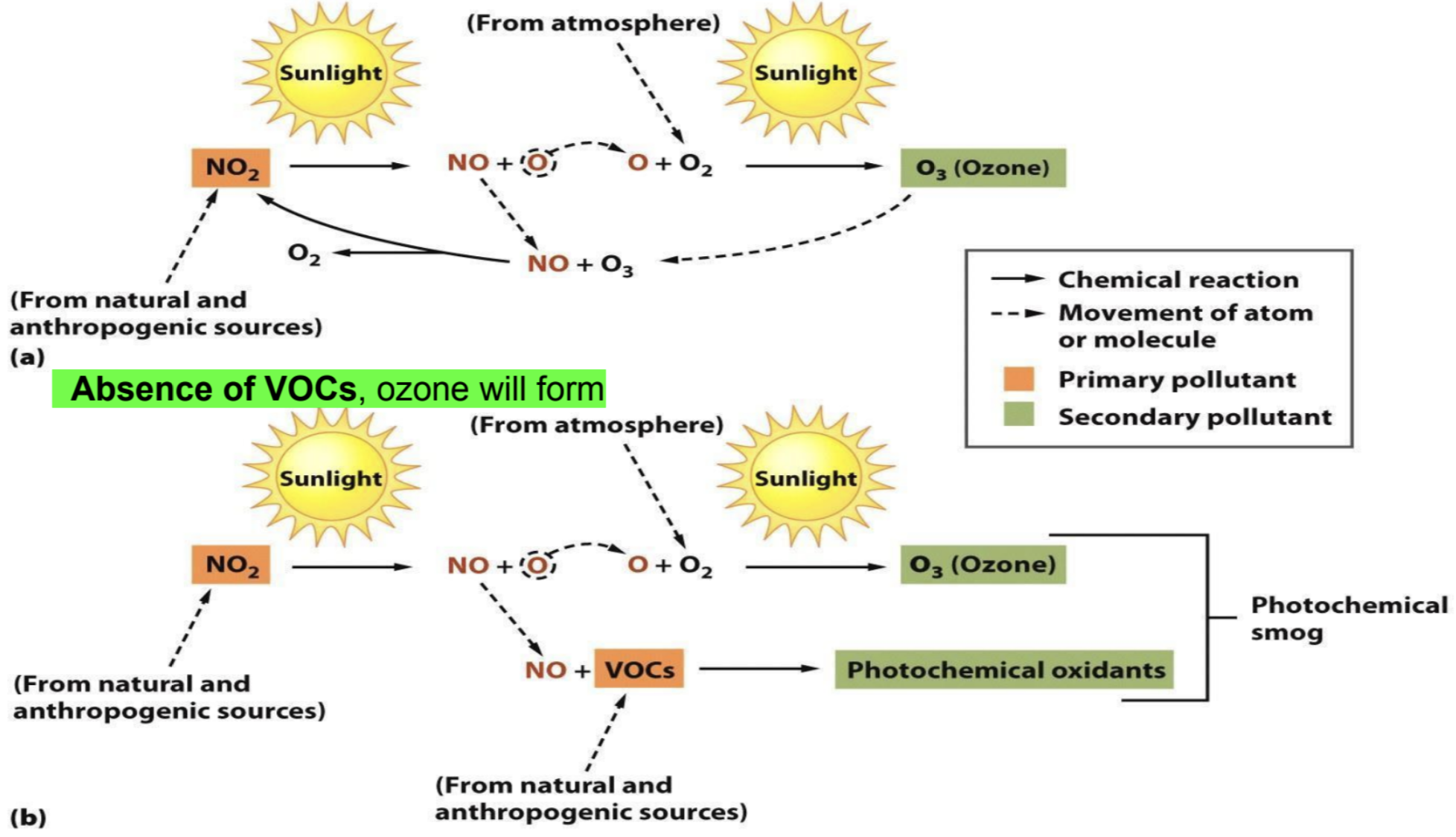
Thermal Inversions
Definition: Occurs when warm air traps colder air below it, leading to confined pollution.
Example: 1998 Northern China incident with coal stoves leading to fatalities from CO poisoning.
Effects: Can exacerbate respiratory issues, increase ground-level ozone concentrations, and contribute to environmental degradation.
Acid Deposition
Definition: Results from NOx and SOx combining with atmospheric components to form acids (e.g., nitric and sulfuric acid).
Effects: Lowers pH of water bodies, reduces species diversity, releases metals from soils, and damages structures.
Stratospheric Ozone
Location: 45-60 km above Earth.
Importance: in stratosphere- Absorbs 99% of UV-B and C radiation, preventing harm to life.
Formation: UV-C radiation breaks oxygen bonds, leading to ozone production.
Ozone Layer Depletion
Causes: CFCs from refrigeration, aerosols breaking down ozone.
Impact: Increase in UV radiation leading to health issues like skin cancer and immune system suppression.
Prevention of Air Pollution
Montreal protocol - agreement that allowed for a reduction and elimination of CFC production and use
Reduce fossil fuel use.
Use fluidized bed combustion to reduce SO₂ emissions.
Install catalytic converters to reduce NOx and CO emissions.
Implement baghouse filters for particulate removal.
Use electrostatic precipitators for particle collection from emissions.
Utilize scrubbers on smokestacks for particle filtration.
Innovative approaches: Montreal Protocol for CFC management.
Indoor Air Pollutants
Sources: Wood, coal stoves, asbestos, carbon monoxide from faulty exhausts, radon from soil and groundwater.
Health Risks: Respiratory problems, cancer from VOCs, nerve damage from lead, and asthma from mold.