Chapter_18_Apoptosis
Chapter 18: Cell Death
1. Overview of Cell Death
Cell death is an essential biological process that serves to remove unnecessary, damaged, or infected cells and tissues, playing a critical role in maintaining homeostasis. This process is not only pivotal during development, where it shapes organ structures and removes transient structures, but also during tissue remodeling and in response to cellular stress or injury.
2. Types of Cell Death
Apoptosis: Also known as programmed cell death, apoptosis is a highly regulated and controlled process that involves a cascade of molecular events leading to cellular dismantling without triggering an inflammatory response. This is a vital process in development and in cellular turnover in tissues.
Necrosis: In contrast, necrosis refers to uncontrolled cell death that occurs as a result of severe pathological conditions such as injury, lack of blood supply, or infection. Necrosis often results in inflammation and damage to surrounding tissues.
3. Significance of Regulated Apoptosis
The regulation of apoptosis is crucial; disruptions can lead to significant health issues. For instance, mutations in the gene encoding Caspase 9 inhibit apoptosis, resulting in:
Over-proliferation of neurons, which can contribute to neurodevelopmental disorders and cancers.
Abnormal brain structures, including structural anomalies such as brain protrusions, which can impact cognitive function.
Persistence of webbing in limbs, indicative of a failure to appropriately undergo apoptosis during embryonic development, known as syndactyly.
4. Morphological and Biochemical Features of Apoptosis
Key features of apoptosis include:
Blebbing of Plasma Membrane: The cell membrane exhibits outward bulging, or blebbing, as the cell shrinks.
Pyknosis: During apoptosis, the nucleus undergoes condensation, resulting in dense, dark-staining nuclei.
Golgi Fragmentation: The Golgi apparatus disassembles, disrupting normal cellular processes.
DNA Fragmentation: Cells undergoing apoptosis show characteristic fragmentation of nuclear DNA into oligonucleosomal bands.
5. Changes in Plasma Membrane During Apoptosis
During apoptosis, phosphatidylserine, a phospholipid normally found on the inner leaflet of the plasma membrane, translocates to the outer leaflet. This change serves as a signal for phagocytic cells, such as macrophages, to recognize and engulf the dying cell, thereby preventing inflammation.
6. Mediators of Apoptosis
Caspases: These are a family of cysteine aspartic acid proteases that play essential roles in the apoptosis pathway. Initiator caspases activate other caspases known as effector caspases through a process called proteolytic cleavage.
Types of Caspases:
Initiator Caspases: These include Caspase 8 and Caspase 9, which are crucial for activating the apoptotic cascade.
Executioner Caspases: These caspases, such as Caspase 3, are responsible for cleaving various cellular substrates leading to the morphological changes associated with apoptosis. Activation occurs from inactive precursors, referred to as procaspases.
7. Initiator Caspase Activation
Caspases have specific protein interaction domains that allow them to participate in apoptosis signaling:
Death Domain (DD): Mediates interaction with receptor signaling pathways.
Caspase Recruitment Domain (CARD): Facilitates the assembly of caspases into active complexes.
Death Effector Domain (DED): Helps bind to adaptor proteins that facilitate signaling.
8. Consequences of Caspase Activation
The activation of caspases leads to cellular destruction through several mechanisms:
Cleavage of ICAD (Inhibitor of Caspase Activated DNase) releases CAD, which then cleaves nuclear DNA.
Cleavage of nuclear lamins disrupts the nuclear architecture, contributing to cell disassembly and further progression of apoptosis.
9. Pathways of Apoptosis
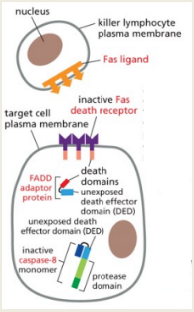
Extrinsic Pathway: This pathway is initiated by the binding of extrinsic ligands to death receptors belonging to the tumor necrosis factor (TNF) receptor family. This leads to the recruitment of adaptor proteins like FADD, which combine to form the Death-Inducing Signaling Complex (DISC), activating Caspase 8 and subsequently triggering downstream caspases.
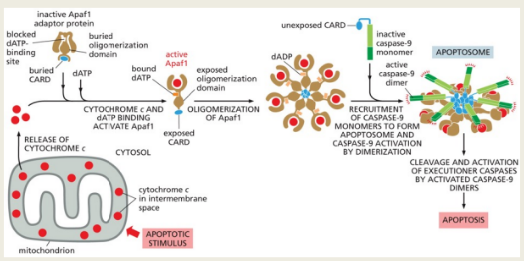
Intrinsic Pathway: This pathway is activated in response to internal stress signals leading to mitochondrial outer membrane permeabilization (MOMP). This results in the release of cytochrome c from mitochondria into the cytosol, where it forms a complex with Apaf-1, leading to the activation of Caspase 9 and subsequent effector caspases.
10. Regulation of Apoptosis
Bcl Proteins: This family of proteins plays a pivotal role in regulating the intrinsic apoptotic pathway through their pro- and anti-apoptotic actions. Pro-apoptotic Bcl proteins cause MOMP when activated, while anti-apoptotic Bcl members inhibit the activity of these pro-apoptotic factors, maintaining cell survival.
11. Interaction of Pathways
One important interaction is mediated by the Bid protein, which, when cleaved and activated by Caspase 8 in the extrinsic pathway, promotes MOMP by activating pro-apoptotic members like Bax and Bak.
12. Inhibitors of Apoptosis (IAPs)
IAPs are important for regulating apoptosis as they inhibit active caspases and prevent premature cell death. They have BIR domains that facilitate binding to caspases and RING domains involved in the polyubiquitination and degradation of target proteins. Apoptotic stimuli can overwhelm IAP function:
In Drosophila: Proteins like Grim, Reaper, and HID are known to induce apoptosis by antagonizing IAPs.
In Mammals: Proteins Smac and Omi, released from mitochondria during apoptosis, counteract the effects of IAPs.
13. Survival Pathways
The PI3K/AKT signaling pathway promotes cell survival by activating various downstream targets. Through the phosphorylation of PIP2 into PIP3, AKT is activated, phosphorylating and thereby inactivating pro-apoptotic proteins like Bad, leading to increased survival.
14. Inhibition of Apoptosis by Survival Factors
The increased production and activation of survival factors, including growth factors and cytokines, enhances pathways that inhibit apoptosis, allowing cells to resist programmed death even under conditions that typically induce apoptosis.