Osmosis
Semipermeable membrane
There is a wall containing little balls in the middle of the U shaped tube, this wall allows the smaller substance to pass through but blocks the bigger one.
Phenomena of Osmosis
There is a U shaped tube is separated by the semipermeable membrane and filled with solvent(A) in one half and solute(B) in the other half.The membrane allows the solvent to pass through to both sides but not the solute but due to different concentrations the solvent moves from lower concentration to higher concentration.
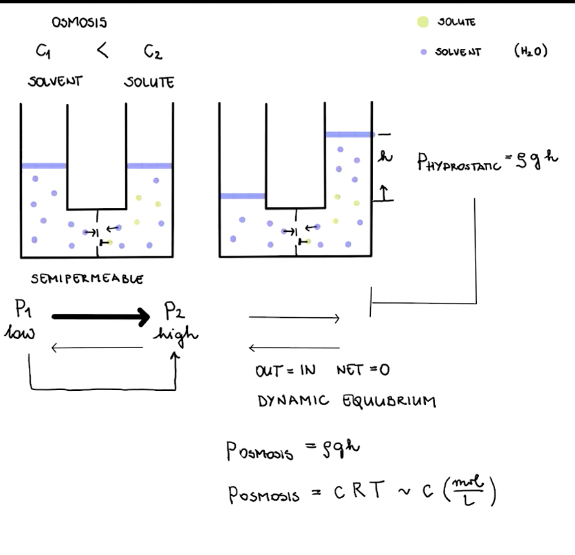
Causes
Due to the concentration difference between the two columns but the solvent is still able to pass through the membrane.
Consequences
Height of liquid decreases in A but increases in B and heat of the column increases in B but decreases in A. Hydrostatic pressure increases as the heat increases and stops net flow of solvent leading to dynamic equilibrium.
Hydrostatic pressure
Pressure due to density of the liquid.
𝑊 = 𝑚𝑔 = 𝜌𝑉𝑔 = 𝜌𝐴h𝑔
𝑝 = 𝜌𝐴h𝑔/𝐴 = 𝜌h𝑔
Osmotic pressure
Pressure is exerted on solution connected to pure solvent by the semipermeable membrane to counteract osmosis.
𝑝𝑜𝑠𝑚𝑜𝑠𝑖𝑠 = 𝜌𝑔h
𝑝𝑜𝑠𝑚𝑜𝑠𝑖𝑠 = 𝑐𝑅𝑇
Van’t Hoffs law
𝑝𝑜𝑠𝑚𝑜𝑠𝑖𝑠𝑉 = 𝑛𝑅𝑇 → 𝑝𝑜𝑠𝑚𝑜𝑠𝑖𝑠 = 𝑛𝑅𝑇/𝑉 = 𝑛/𝑉 x 𝑅𝑇 = 𝑐𝑅𝑇
Increase in Osmotic pressure is proportional to increase in concentration.
Classification of solutions based on osmotic pressure
A) Hypertonic: water is moving out of the cell into the environment ==so it shrinks== and osmotic pressure decreases in the cell.
B) Isotonic: the concentration of the cell and environment are equal so the osmotic pressure is also equal.
C) Hypotonic: water is moving from the environment into the cell so it expands and osmotic pressure increases in the cell.
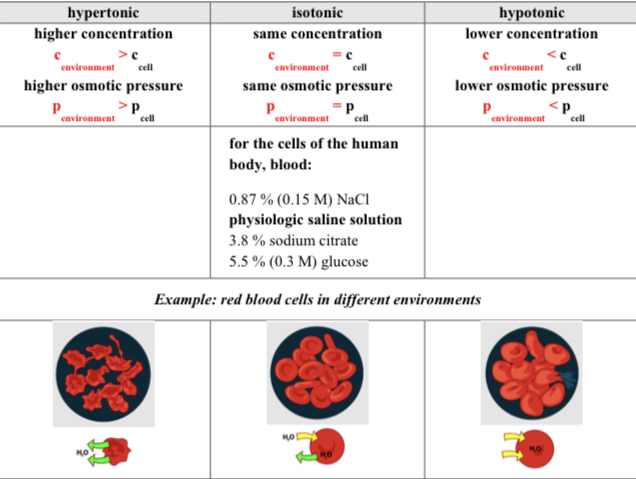
Biological relevance of osmosis
In administration of drugs(isotonic environment), treatment of oedema and constipation(hypertonic environment), hemodialysis.