M6L2 ECM targeting therapies
Fibroblasts in the ECM:
Large secretome - cytokines, growth factors, ECM remodelling enzymes etc to maintain homeostasis in ECM
ECM production
Collagen - most abundant ECM protein
28 types - collagen I (fibrillar, for support), collagen IV (basal lamina) most well known
Homo/heterotrimers
Fibrils and network forming collagens
Integrins can bind to collagen for structural support and signalling (proliferation, adhesion/migration, survival, differentiation pathways etc)
Fibronectin
ECM glycoprotein
Binds to other ECM proteins, eg fibrin
Contains RGD motifs which binds cell surface receptors
Important in TBFb deposition
Glycoproteins, proteoglycans, glycosaminoglycans (GAGs)
Proteoglycans - protein core with GAGs (negatively charged sugars) attached to it
5 main GAGs - hyaluronic acid most important
Important role in lubrication, local sequestration of GFs and cytokines
Binds CD44
ECM modifying enzymes
MMPs - LOX, ADAMs
Degradation, modification, enzyme activation, stiffness/crosslinking, GF regulation
Can be secreted or be on cell surface
ECM in wound healing
Coagulation/inflammation - transient fibrin matrix, recruitment of neutrophils and later macrophages
Tissue formation - keratinocytes (re-epithelialisation), MMP production, myofiobroplasts, angiogenesis
Most relevant for cancer - cancer as the ‘never healing wound’, perpetual inflammation and extensive remodelling by MMPs
Remodeling - collagen formation, wound contraction, scar maturation
ECM in cancer
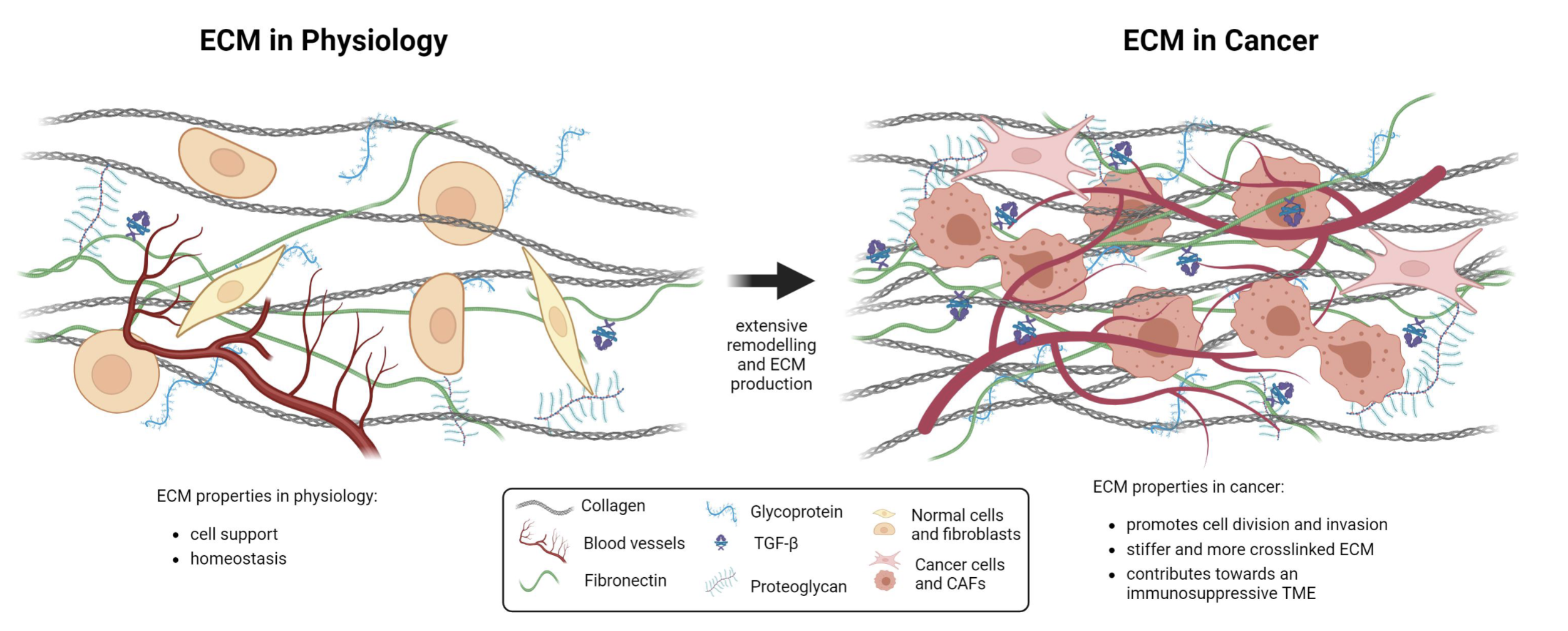
ECM becomes an active player in cancer, containing the niche and protecting the tumour from immune infiltration and drug delivery
Upon tissue invasion, a layer of ECM helps cancer to behave the way that it does
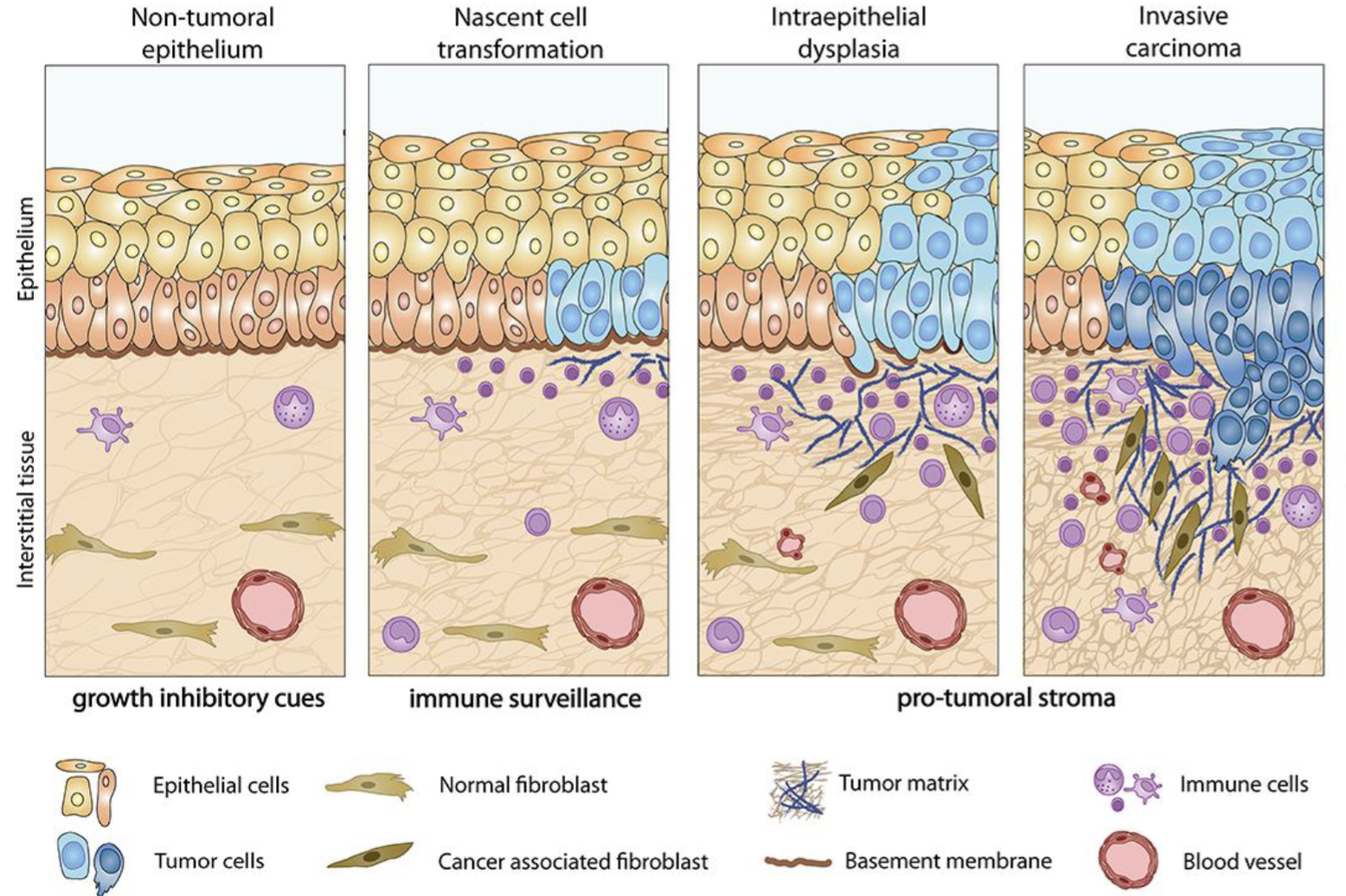
CAFs
Produce tumour ECM together with cancer cells
Very heterogenous (many progenitors - adipocytes, endothelial cells, epithelial cells, mesenchymal cells etc)
Produce cytokines and ECM remodelling enzymes
Collagen in cancer ECM
Cancer —> more collagen deposition by CAFs or cancer cells
Collagen fibres are crosslinked to a higher extent (stiffer)
Alignment of collegen fibres change
Tumour associated collagen signatures (TACS)
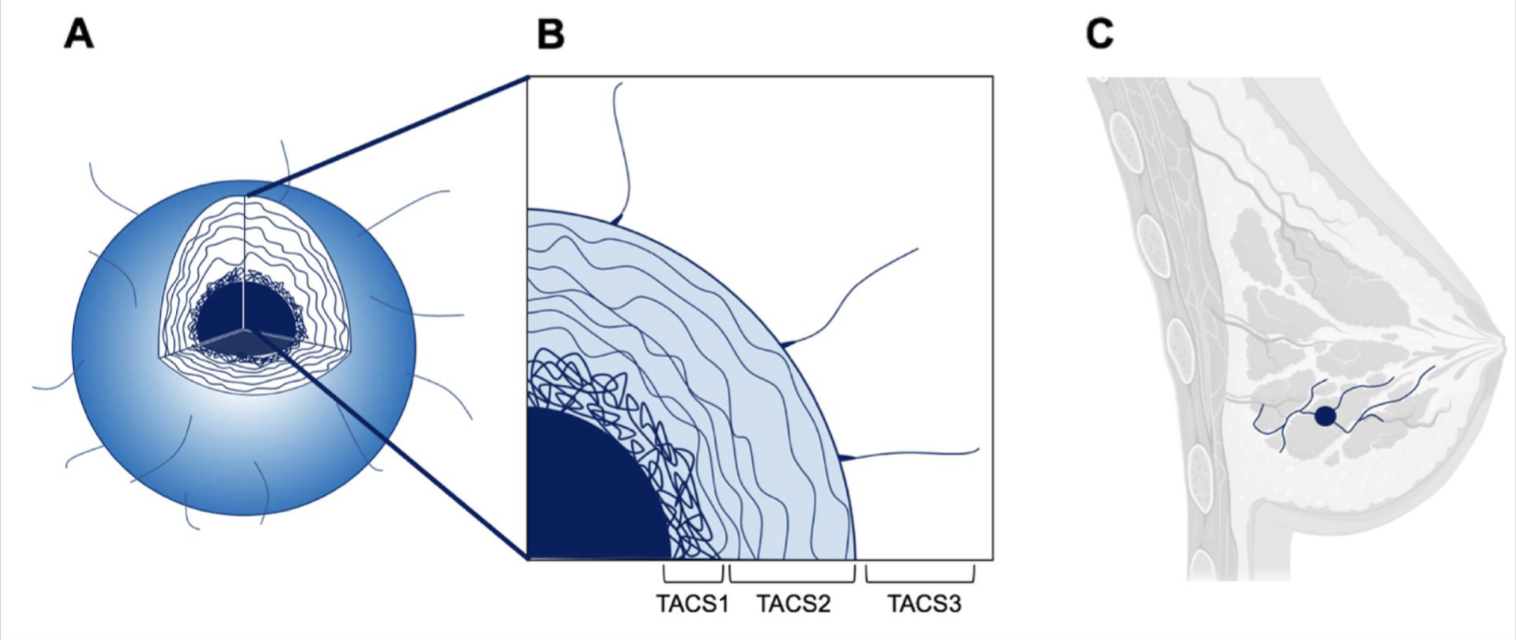
TACS1 - random alignment of collagen directly adjacent to tumour core
TACS2 - slightly more aligned
TACS3 - aligned collagen tracts which give cancer cells a path for invasion
Fibronectin in cancer
Higher expression of extra domain B fibronectin (EDB-FN)
Expressed in development (oncofoetal)
Vascularisation, angioghenesis
Can be used as predictive marker of cancer progression (more expression = more invasion, resistance…)
Not in all cancers, but about ~50%
Glycoproteins, proteoglycans, GAGs in cancer
Upregulated
Often coupled with upregulated proteoglycans
Correlates with poorer therapeutic penetration, eg Abs
Effects on therapies
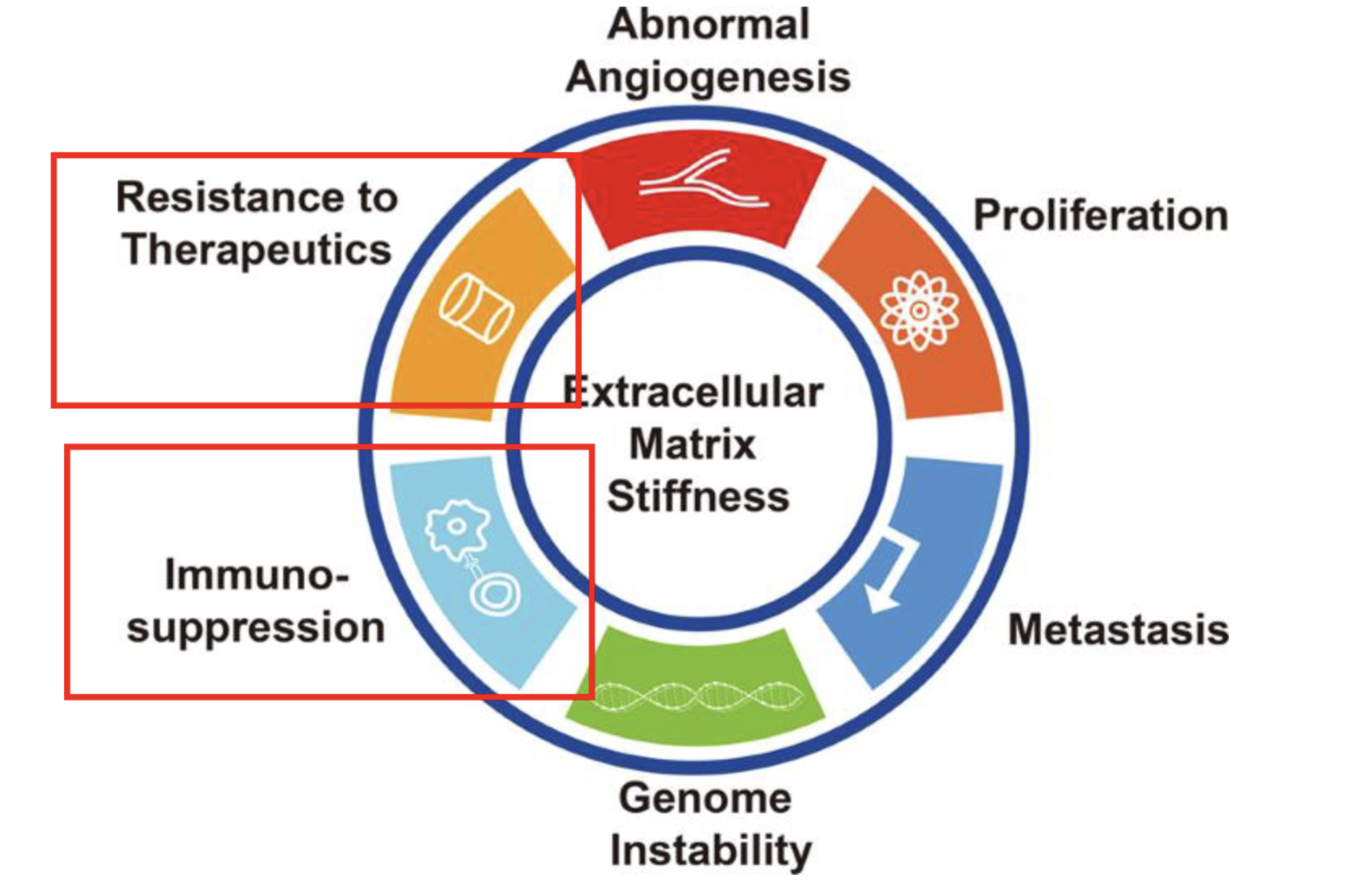
Chemo/radioresistance
LOXL2 (collagen crosslinking enzyme) expression in spheroids significantly reduces doxorubicin penetration vs control
Increasing Gy has a greater killing effect in 2D cultures vs 3D environment
Immunosuppression
Covalent linkage from immunosuppressants to ECM
TGFb - master cytokine
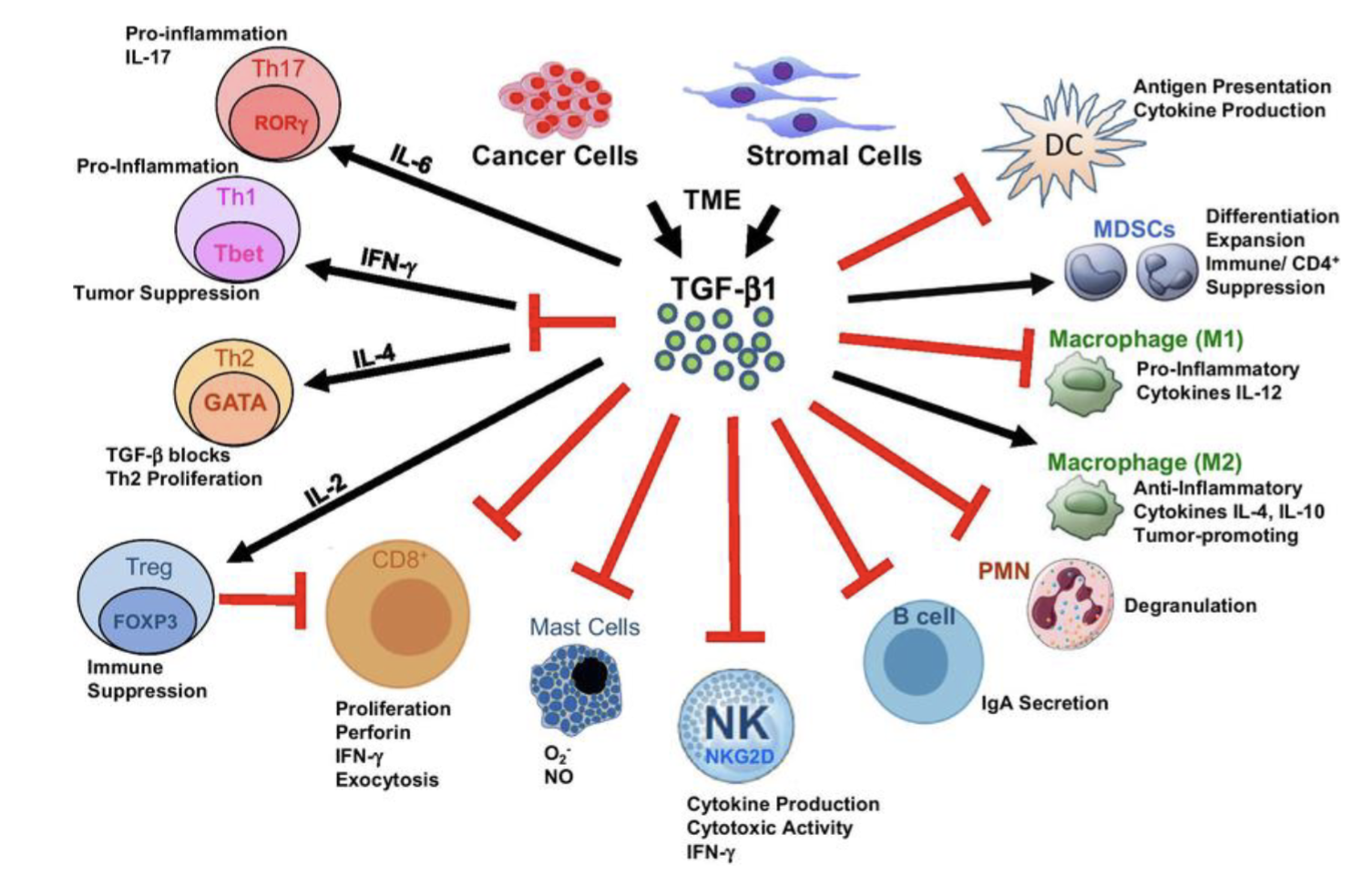
Anti-tumour in early stages, pro-tumour as cancer evolves
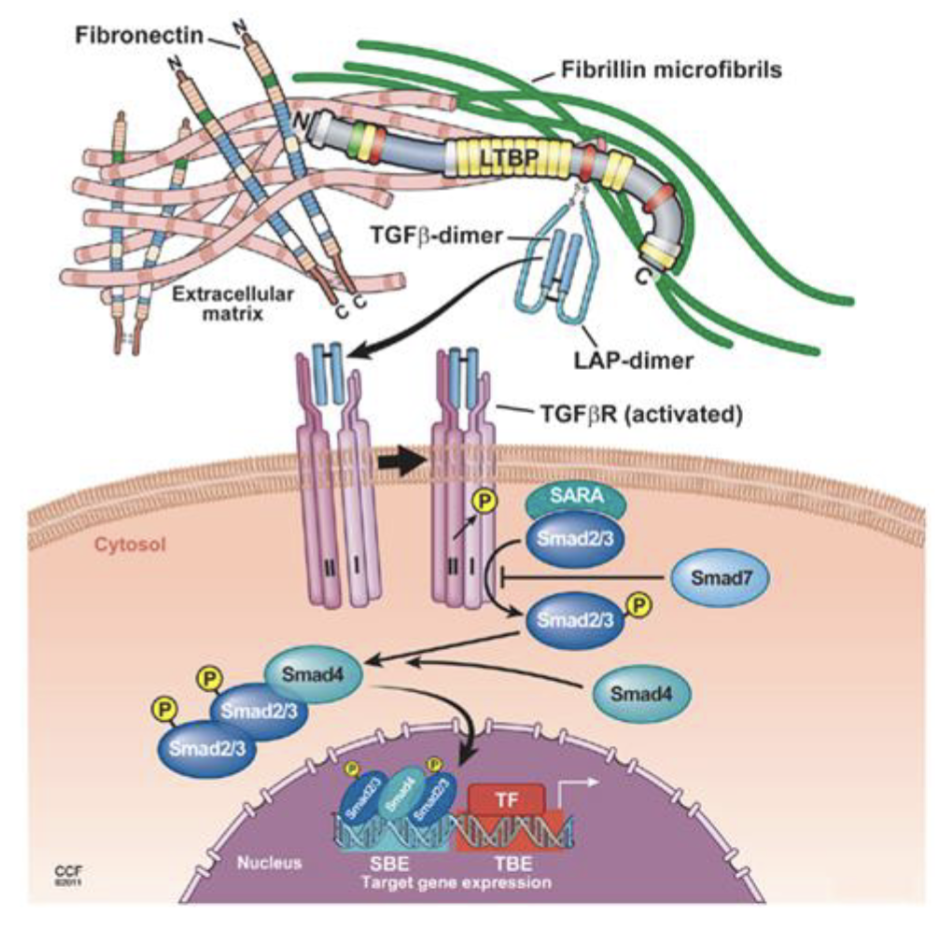
Covalently linked into LTBP - LAP-dimer hides TBFb
Once it is released signalling is rapid due to proximity to cells
Hard to target TGFb as it is hidden
Association of GFs, GAGs, cytokines…
Binds to VEGF, PDGF, FGF, TGF, BMP, NGF, IL-10, IL-6 etc…
Hyaluronan (HA) in naked mole rat is x5 larger vs human or mouse
Introducing cancer cells into skin fibroblasts from naked mole rat does not develop into tumours
CD44 interaction with normal HA vs preventing CD44 interaction with high MW HA could be the reason
HA may be an immunosuppressant
ECM components
Most research is done in 2D - not representative of 3D architecture in vivo (though cheaper, less time consuming, less variable)
No ECM to mature ECM: 2D tissue culture —> spheroids —> organoids —> in vivo (subcutaneous) —> in vivo (orthotopic) —> patient derived tissue slices
ECM targeting
Targeting is difficult - expressed everywhere in the body
Conventional targeting/depletion of ECM proteins - transcriptional depletion, ECM component mimetics, destructiion of ECM producing cells, degradation of ECM
Collagen targeting with halofuginone - decreases alpha smooth muscle actin (marker for CAFs), but drug toxicity
GAG targeting with PEGPH20 - small TI, not much change in survival
Proteoglycan/GAG approach - PG545 (mimetic), activates TLR9 (immune activation), can combine with anti-PD1
Generally conventional approaches have not met goals or terminated due to severe AEs
ECM degradation as supportive strategy - combination with CART cells, OVs etc
OVs with transgene hyaluronidase - increaed spread and efficacy of OV in vivo
CAR-T cell + ECM degradation enzymes are more potent at infiltrating/clearing solid tumours, eg. CAR-T cell against HER2 expressing MMP9/12 and HPSE entering TME through synNotch system
Specific ECM targeting - specific ECM antigens/components
Targeting EDB-FN: EDB-FN x IL-2 could help clear renal cell carcinoma (some patients, not others)
Fibronum by Philogen 0 targeting antibody (L19) + TNF (immunostimulatory and cytotoxic payload)
Targeting tenascin C - anti-FBG + anti-PDL1
Targeting tenascin C with CAR-T cells - in GBM the ECM is very rich in tenascin C (increases with the grade)
PIGF-1 and PIGF-2: 21 AA motif which binds to ECM fused with anti-CTLA4/PDL1 rediuces tumour growth in mice