Cancer Biology & Cancer Epidemiology
Cancer Scenario
Patient: Jimmy, 56 years old male.
History and Symptoms:
3 packs per day smoker for 20 years.
Presents with cough, shortness of breath (SOB), blood-tinged sputum, and lethargy.
Vital signs stable (VSS).
Examination indicates wheezing in the right middle and lower lobes, cervical and axial lymphadenopathy.
Diagnostic Considerations:
Diagnosis: Should consider the type of cancer (lung likely due to smoking history).
Cause: Likely due to smoking-related damage leading to malignancy.
Treatment: Standard protocols would include chemotherapy, radiation, and possibly surgery depending on staging.
Side Effects: Common side effects of cancer treatment include nausea, vomiting, hair loss, and immune suppression.
Overview of Cancer
Definition:
Cancer (NOT A TUMOR) is characterized as an abnormal growth caused by uncontrolled cell proliferation. Also referred to as a neoplasm, meaning 'new growth'.
Key Characteristics of Tumors:
Benign Tumors:
Slow-growing, well-defined capsule, not invasive, well-differentiated, does not metastasize, Low mitotic index.
Malignant Tumors:
Rapid growth, not encapsulated, invasive, poorly differentiated (anaplasia), variable size/shape (pleomorphic), high mitotic index, can spread distantly (metastasis).
Classification and Nomenclature of Cancer
Benign Tumors:
Named according to the tissues from which they arise with suffix '-oma'.
Example:
Lipoma: Fat
Leiomyoma: Smooth muscle
May have potential to progress to cancer.
Malignant Tumors: Named by cell type:
Carcinoma: Epithelium
Adenocarcinoma: Ductal or glandular tissues
Sarcoma: Mesenchymal tissues
Lymphoma: Lymphatic tissues
Leukemia: Blood-forming cells
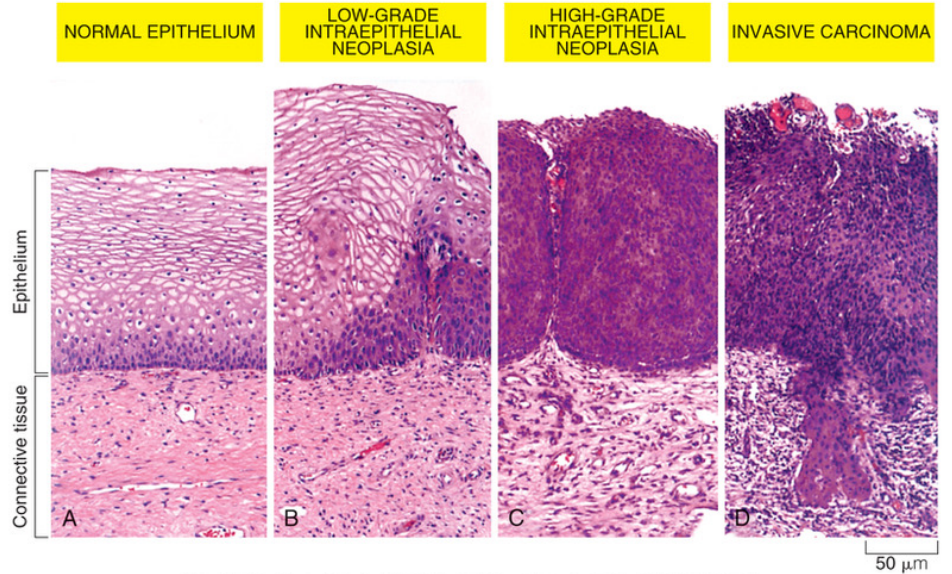
Carcinoma in Situ (CIS):
Early-stage cancer, preinvasive epithelial tumors of glandular or squamous cell origin.
Characteristics:
Have not broken through the basement membrane.
Outcomes may include remaining stable, progressing to invasive cancer, or regressing and disappearing.
Cancer Development Stages
Tumor Initiation:
Normal cells become cancer cells, influenced by epigenetics.
Tumor Promotion:
Cancer cells become more complex, influenced by genetic changes and the tumor environment.
Tumor Progression:
Leads to the spread of the tumor (metastasis), associated with genetic changes and tumor environment.
Human Carcinogenesis
Three key Genetic Mechanisms (there are more):
Activation of proto-oncogenes leads to hyperactivity of oncogenes (mutated growth-related gene products).
Mutation of tumor-suppressor genes results in loss of function, inhibiting growth regulation.
Mutation leading to overexpression of apoptosis inhibitors allows for continued tumor growth.
Cancer-Causing Mutations
Genetic events are the primary basis of carcinogenesis.
Types of mutations accumulate with age, increasing cancer risk with advancing age.
Multiple mutations (estimated 4 to 7 'hits') are typically required before cancer develops, indicating a step-wise accumulation of alterations.
Driver Mutations:
Approximately 140 different types, associated directly with cancer development.
Passenger Mutations:
Random mutation events that cause cancer.
Types of Mutated Genes
Point Mutations:
Changes in one or a few nucleotide base pairs.
Example: Ras gene mutation converts it from a regulated to an unregulated oncogene, accelerating growth.
Chromosome Translocation:
A piece from one chromosome is transferred to another.
Gene Amplification:
A small chromosome segment is duplicated multiple times, increasing oncogene expression.
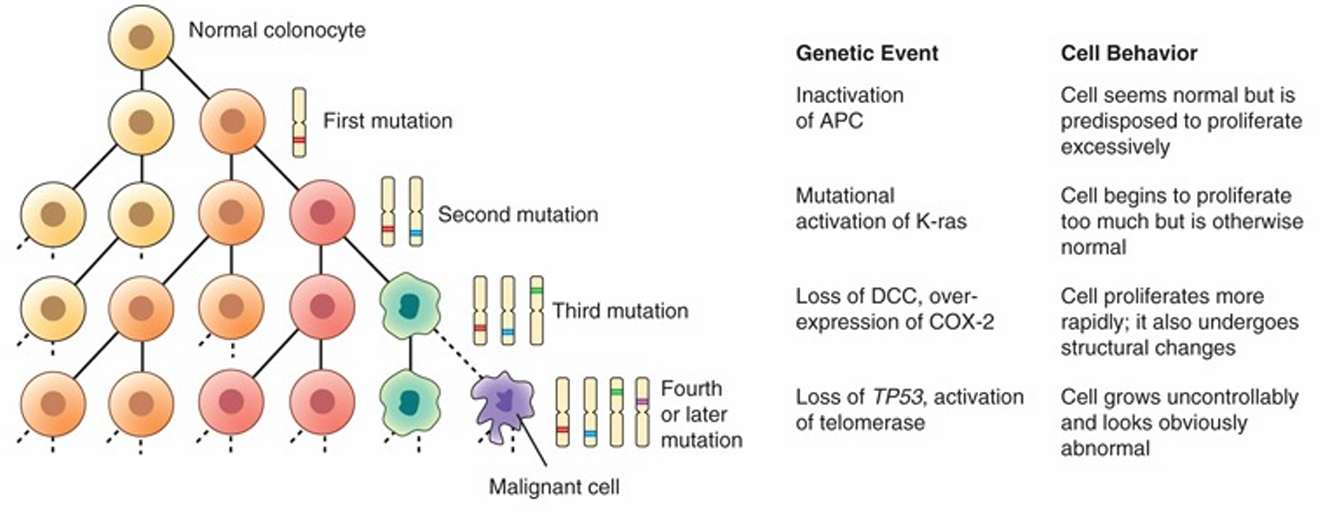
Cancer-Causing Mutations (colon cancer example)
Genetic events and cell behavior
Inactivation of APC (adenomatous polyposis coli)
Cell seems normal but is predisposed to proliferate excessively.
Mutational activation of K-ras (a member of the RAS oncogene family)
Cell begins to proliferate too much but is otherwise normal.
Loss of DCC (Tumor suppressor gene), over expression of COX-2
Cell proliferates more rapidly; it also undergoes structural changes.
Loss of TP53 (tumor suppressor gene), activation of telomerase
Cell grows uncontrollably and looks obviously abnormal.
Tumor Environment
Development of cancer is analogous to wound healing, initiating inflammatory responses and recruitment of immune cells (primarily T lymphocytes and macrophages).
These cells cause formation of a stroma or tumor microenvironment occurs.
Over time cancer heterogeneity develops through ongoing proliferation and mutations, resulting in altered cellular signaling and phenotype.
Tumor responds differently than normal cells.
May have a different genetic expression.
May respond differently to cellular signaling.
May take on a new phenotype and have genetic diversity.
Cancer Stem Cells (CSC): May arise, contributing to tumor variability.
Hallmarks of Cancer
Emerging Hallmarks & enabling characteristics:
Phenotypic Plasticity: Adaptation of phenotypes in response to environmental factors.
Disrupted Differentiation: Interference with normal maturation processes due to epigenetic factors.
Nonmutational Epigenetic Reprogramming: Changes in gene expression not due to mutations.
Senescent Cells: Inflammatory mediators released by cells that stop multiplying but fail to die.
Polymorphic Microbiomes: Diversity of microbial systems associated with the host.
Established Hallmarks:
Sustaining proliferative signaling
Evading growth suppressors
Avoiding immune destruction
Enabling replicative immortality
Tumor-promoting inflammation
Activating invasion & metastasis
Inducing or accessing vasculature
Genome instability & mutation
Resisting cell death
Deregulating cellular metabolism
Hallmark 1: Sustained Proliferative Signaling
Key characteristic of cancer is uncontrolled cellular proliferation.
In cancer, mutated proto-oncogenes lead to oncogenes contributing to sustained growth.
Example: RAS oncogene stimulates continual growth, mutations prevent normal regulatory shut-off.
Autocrine Stimulation: Cancer cells may secrete growth factors that stimulate their own proliferation (e.g., HER2).
Oncogenes and tumor-suppressor genes
Proto-oncogene
A normal, nonmutant gene that codes for cellular growth.
Ex. Epithelial growth factor.
Oncogenes (GO!!)
MUTANT proto-oncogenes that direct protein synthesis and cellular growth.
Tumor-suppressor genes (STOP!!)
Encode proteins that negatively regulate proliferation.
Also referred to as anti-oncogenes.
Hallmark 2: Evading Growth Suppressors
Inactivation of tumor suppressor genes
BRCA1 and BRCA 2
Inactivation of Rb tumor suppressor (Retinoblastoma)
Anti-proliferative activity of Rb depends on the degree of protein phosphorylation.
When hyperphosphorylated, Rb loses its ability to bind to E2F transcription factors, leading to uncontrolled progression through the cell cycle, which can contribute to tumorigenesis.
When hypo phosphorylated, Rb retains its functionality and effectively sequesters E2F transcription factors, thereby inhibiting the transcription of genes required for cell cycle progression and promoting cell cycle exit, which helps prevent tumor formation.
Mutation in the p53 gene
Caretaker gene, maintains cell.
In cancer, inactivation of p53, leads to increased mutation rates and cancer.
Hallmark 3: Genomic Instability
Cancer cells demonstrate increased tendency of mutations and alterations in the genome.
Caretaker genes decrease
Encode for proteins that are involved in repairing damaged DNA.
Chromosome instability
Increased in malignant cells.
Results in chromosome loss, loss of heterozygosity, and chromosome amplification.
Hallmark 4: Enabling Replicative Immortality
Body cells are not immortal and can only divide a limited number of times Exception: germ cells (testes, etc...).
Telomeres are protective caps on each chromosome, held in place by telomerase, and are important for cellular replication signaling.
Telomeres become smaller and smaller with each cell division and eventually become so small they signal the cell to stop replication.
Cancer cells ACTIVATE and SUSTAIN telomerase which helps preserve the telomere on the cancer cell chromosome. This allows for continued replication.
Telomerase activity restored in 90% of cancer
Hallmark 5: Cellular Adaptations
Angiogenesis: Formation of new blood vessels, allowing tumor growth. “Neovascularization”.
Advanced cancers can secrete angiogenic factors to facilitate feeding of the tumor (similar to wounds).
Vascular endothelial growth factor (VEGF)
Platelet-derived growth factor (PDGF)
Basic fibroblast growth factor (bFGF)
Vessels are more porous and fragile
Prone to hemorrhage.
Allow for passage of cancer cells into vessels and tissues.
Hallmark 6: Reprogramming Energy Metabolism
Cancer cells alter their metabolism to support uncontrolled proliferation, helping them withstand nutrient deprivation and hypoxia.
Oncogenes can drive the process by enabling cells to:
Maintaining deregulated proliferation and growth.
Withstand oxygen and nutrient limitations.
Maintain abnormal gene expression.
Corrupt the tumor environment to assist with tumor growth and dissemination.
Hallmark 7: Resistance to Destruction
Resist Death: mutation in the p53 gene.
Tumor promotion of inflammation:
Tumor associated macrophage
Higher amount, the worse the prognosis.
Mimics M2 phenotype.
Diminish cytotoxic response.
Block T-cytotoxic cells and natural killer cell function.
Produce cytokines that are advantageous for cellular growth and spread.
Secrete growth factors.
Secrete angiogenic factors.
Avoid immune destruction.
Hallmark 8: Activating Invasion and Metastasis
Spread of cancer from a primary site of origin to a distant site
Steps:
Direct or continuous extension.
Penetration into lymphatics, blood vessels, or body cavities.
Transport into lymph or blood –”seeding”.
Transport to secondary sites.
Entry and growth in secondary sites.
Metastasis often occurs in the first capillary bed encountered by circulating cells.
Organ tropism
Preferential growth of cancerous cells in certain organs.
Growth factors, chemokines, hormones, tissue selective homing receptors, and chemotactic factors.
Metastasis Manifestations and Organ Tropism
Metastatic patterns include specific organ preferences:
Lung to Brain
Colorectal to Liver to Lungs
Prostate to Bone to Liver
Breast to Bone
Head/Neck to lymph
Ovarian to peritoneal cavity and omentum
Inflammation and Cancer
Chronic inflammation is an important factor in the development of cancer.
Increased Reactive Oxygen Species (ROS).
Increased free radicals.
Promotion of oxidative stress.
Alteration of cellular proteins.
Promotion of DNA damage.
Viruses and Cancer
HIV-1: Non-Hodgkin lymphoma, squamous cell carcinomas, Kaposi sarcoma.
Hepatitis B and hepatitis C: Hepatocellular carcinoma.
Epstein-Barr virus: B-cell non-Hodgkin lymphoma, Burkitt lymphoma, nasopharyngeal carcinoma.
KSHV/HHV8 and immunodeficiency: Kaposi sarcoma.
HPV-16,-18,-31,others: Cervical, anogenital.
Causes virtually all cervical cancer.
HTLV-1: Adult T-cell leukemia/lymphoma.
Bacterial cause of cancer
Helicobacter pylori
Chronic infections are associated with:
Peptic ulcer disease
Stomach carcinoma
Mucosa-associated lymphoid tissue lymphomas
Cancer Epigenetics
The study of gene expression changes that occur due to mechanisms other than alterations in DNA sequence.
Aberrant Methylation: Silences tumor suppressor genes.
Use of anti-methylation drugs as a reversal agent.
Histone Modifications: Can affect gene activity.
MicroRNAs: Target gene expression.
Influenced by nutrition, environment, and lifestyle factors.
1 mutated cell does not equal cancer.
Many years of promoting events cause cancer to develop.
Early life events influence later life susceptibility.
Promotors: Endogenous hormones (ex. estrogen), food additive (ex. nitrates), cigarrettes, and alcohol.
Promoting agents accelerate proliferation of mutated cell.
Some promoting agents may deactivate tumor suppressor gene.
In Utero and Early Life conditions
Conditions that increase susceptibility to cancer include:
Prenatal exposure
Parental exposures before conception
In utero exposure: Diet, drugs, toxins
Possible toxins in breast milk after birth
Gene and environment interactions
Developmental plasticity: Degree to which development is contingent on its environment.
Reducing cancer risk must start early in life: Avoid sun exposure during peak hours, cover the skin, increase physical exercise, and avoid high risk sexual practices.
Environmental Factors and Their Impact
Ionizing Radiation: Causes gene mutations and chromosomal aberrations.
Emission from x-rays, radioisotopes, and other radioactive sources.
Exposure causes cell death, gene mutations, and chromosome aberrations.
Ultraviolet Radiation: Major cause of skin cancers through inflammation and free radical production.
Causes basal cell carcinoma, squamous cell carcinoma, and melanoma.
principal source is sunlight.
Ultraviolet A (UVA) and ultraviolet B (UVB).
Promotes skin inflammation and release of free radicals.
Alcohol Consumption: Increases risks for various cancers, particularly related to oral and liver cancers.
Lifestyle and Environmental Risk Factors
Alcohol consumption:
Risk factor for oral cavity, pharynx, hypopharynx, larynx, esophagus, and liver cancers.
Cigarette/alcohol combination increases a person’s risk.
Alcohol Dehydrogenase (ADH): When alcohol (ethanol) is consumed, it is primarily metabolized in the liver by the enzyme alcohol dehydrogenase (ADH). This enzyme converts ethanol into acetaldehyde, a toxic and carcinogenic compound.
Acetaldehyde: Acetaldehyde is highly reactive and can interact with DNA, leading to the formation of DNA adducts. These adducts can cause mutations in the DNA sequence, disrupting normal cell functioning and potentially leading to cancer.
Acetaldehyde Dehydrogenase (ALDH): In normal metabolism, acetaldehyde is further metabolized by another enzyme known as acetaldehyde dehydrogenase (ALDH), which converts it into a non-toxic substance called acetic acid that can be easily eliminated from the body. However, genetic variations in ALDH can lead to decreased enzyme activity, resulting in higher acetaldehyde levels and increased cancer risk.
Additionally, chronic alcohol consumption can lead to oxidative stress, inflammation, and disruption of nutrient absorption, which also contribute to cancer development.
Tobacco Use:
Multipotent carcinogenic mixture
Linked to cancers of the lung, lower urinary tract, aerodigestive tract, liver, kidney, pancreas, cervix uteri, and myeloid leukemia.
Includes E-cigarettes/ vaping.
Sexual reproductive behavior
Carcinogenic types of human papillomavirus (HPV).
High-risk HPV strains such as -16 and -18.
Physical activity
Reduces cancer risk
Decreases insulin and insulin-like growth factors.
Decreases obesity.
Decreases inflammatory mediators and free radicals.
Increased gut motility.
Occupational hazards
Substantial number of occupational carcinogenic agents
Asbestos-mesothelioma
Dyes, rubber, paint, explosives, rubber cement, heavy metals, air pollution, etc.
Radon
Air pollution
Air pollution is linked to lung cancer.
Outdoor pollution: Ozone and particle pollution
Ozone: Is the principle component of smog.
Particle pollution: Causes pulmonary inflammation, oxidative stress and oxidation of DNA, proliferative response, tissue remodeling with fibrosis, and tumor development.
Indoor pollution
Is considered worse than outdoor pollution.
Cigarette smoke, radon: Lung cancer.
Inorganic arsenic: Bladder, skin, and lung cancers.
Diet
Xenobiotics
Toxic, mutagenic, and carcinogenic chemicals in food.
Activated by Phase I activation enzymes.
Defense mechanisms
Phase II detoxification enzymes (decrease carcinogens).
Examples
Compounds produced in the cooking of fat, meat, or proteins.
Alkaloids or mold by-products.
Obesity:
Associated with endometrial, colorectal, kidney, esophageal, breast (postmenopausal), and pancreatic cancers.
Causes a poorer outcome for some cancers.
Energy balance may affect: (1) genomic instability, (2) dysregulated growth signaling and cellular energetics, (3) inhibition of apoptosis and immune surveillance, and (4) angiogenesis.
Increases insulin resistance–producing hyperinsulinemia.
Increases the risk of cancers of the colon, endometrium, and possibly the kidney and pancreas.
Insulin promotes insulin-like growth factor 1.
Increases the risk for prostate cancer.
Adipose tissue secretes adipokines.
Increases inflammation.
Clinical Manifestations of Cancer
Paraneoplastic Syndromes: Symptom complexes due to biologic substances released from tumors, can be life-threatening.
Pain: Often minimal in early malignancy.
Influenced by fear, anxiety, sleep loss, fatigue, and overall physical deterioration.
Mechanisms
Pressure, obstruction, invasion of sensitive structures, stretching of visceral surfaces, tissue destruction, and inflammation.
Fatigue: Common clinical manifestation characterized by tiredness and lethargy. Subjective.
Cachexia Syndrome: Severe malnutrition characterized by weight loss.
Present in 80% of cancer patients at death.
includes
Anorexia, early satiety, weight loss, anemia, asthenia, taste alterations, and altered protein, lipid, and carbohydrate metabolism.
Anemia: Decrease in hemoglobin due to various mechanisms.
Mechanisms
Chronic bleeding resulting in iron deficiency, severe malnutrition, medical therapies, or malignancy in blood-forming organs.
Role of chemotherapy.
Leukopenia and Thrombocytopenia:
Direct tumor invasion to the bone marrow causes both leukopenia and thrombocytopenia.
Chemotherapy drugs are also toxic to the bone marrow.
Infection:
Risk increases when the absolute neutrophil and lymphocyte counts fall.
GI manifestations:
Oral ulcers caused by decreased cell turnover from chemotherapy and radiation.
Malabsorption.
Diarrhea.
Therapy-induced nausea.
Hair and skin manifestations:
Alopecia from chemotherapy.
Usually temporary.
Skin breakdown and dryness.
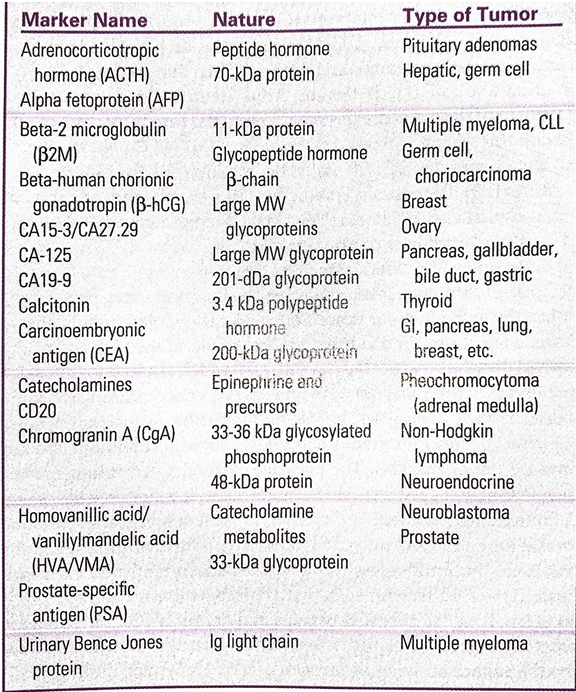
Staging: Cancer/Tumor markers
Are substances produced by benign or malignant cells.
Are found on or in a tumor cell, in the blood, in the spinal fluid, or in urine.
Hormones
Enzymes
Genes
Antigens
Antibodies.
Liver and germ cell tumors
Secrete a protein known as alpha fetoprotein (AFP) into the blood.
Prostate tumors
Secrete prostate-specific antigen (PSA) into the blood.
If a tumor marker, itself, has biologic activity:
Symptoms are expressed.
A phenomenon known as a paraneoplastic syndrome occurs.
Tumor markers are used to:
Screen and identify individuals at high risk for cancer.
Diagnose specific types of tumors.
Observe the clinical course of cancer.
Involves the size of the tumor, degree to which it has invaded, and the extent to which it has spread.
Stage 1
Cancer is confined to its organ of origin.
Stage 2
Locally invasive
Stage 3
Regional structures
Stage 4
Distant sites
Describes the size, invasiveness, and spread of cancer using the TNM system:
T: Tumor size (TX, T0 to T4).
TX: primary tumor cannot be measured.
T0: Primary tumor cannot be found.
T1, T2, T3, or T4: primary tumor has been measured. Higher numbers indicate the tumor is larger or has expanded further into nearby tissue.
N: Regional lymph nodes involvement (NX, N0 to N3).
NX: there is no information about lymph nodes.
N0: Nearby lymph nodes do not contain cancer.
N1, N2 or N3: Cancer is present in the lymph nodes. A higher number indicates the caner has been found in more lymph nodes.
M: Presence of metastasis (MX, M0, M1).
MX: Spread cannot be measured.
M0: Cancer has not spread to other parts of the body.
M1: Cancer has spread to other parts of the body.
Cancer Treatment Modalities
Chemotherapy
Use of nonselective cytotoxic drugs that target vital cellular machinery or metabolic pathways critical to both malignant and normal cell growth and replication.
Induction: Shrink/eradicate tumor.
Adjuvant: eliminate micrometastasis after surgery.
Neoadjuvant: shrink tumor before surgery or radiation to spare more normal tissue.
Salvage: keep metastasis down or prevent rapid growth.
Goal:
Eliminate enough tumor cells so the body’s defense can eradicate any remaining cells.
RAPIDLY REPRODUCING CELLS!!!
Radiation Therapy
Used to kill cancer cells while minimizing the damage to normal structures.
Ionizing radiation
Damages cells by imparting enough ionizing radiation to cause molecular damage to the DNA.
Causes irreversible damage to normal cells.
Lifetime radiation dose
Brachytherapy
Seeds are implanted.
Can cause fertility problems since radiation affects gametes.
Surgery
Is a definitive treatment of cancers that do not spread beyond the limits of surgical excision.
Biopsy and lymph node sampling
Debulking surgery
Palliative surgery
In selected high-risk diseases, surgery plays a role in the prevention of cancer.
Mutations of the APC gene have close to a 100% lifetime risk of colon cancer: Colectomy.
Women with BRCA1/2 mutations have a significantly increased risk of breast and ovarian cancer: Prophylactic mastectomy or bilateral salpingo-oophorectomy or both.
Surgery must achieve adequate surgical margins; that is, surgery must get it all.
Must carefully place needle tracks and biopsy incision scars (that may be contaminated with cancer cells) to enable their removal in subsequent incisions.
Must avoid the spread of cancer cells during surgical procedures.
Must obtain adequate tissue specimens to confirm diagnosis.
Immunotherapy
Theoretically, antitumor responses can selectively eliminate cancer cells while sparing normal cells.
Immune memory is long lived.
Numerous immunologic mechanisms are capable of rejecting different types of cancer.
Biologic response modifiers (BRMs)
Immune Checkpoint Inhibitors.
Hormone therapy
Receptor activation or blockage.
Interferes with cellular growth and signaling.
Other forms of immunotherapy
Interferon administration
Antigens
Effector cell lymphokines
Monoclonal antibodies
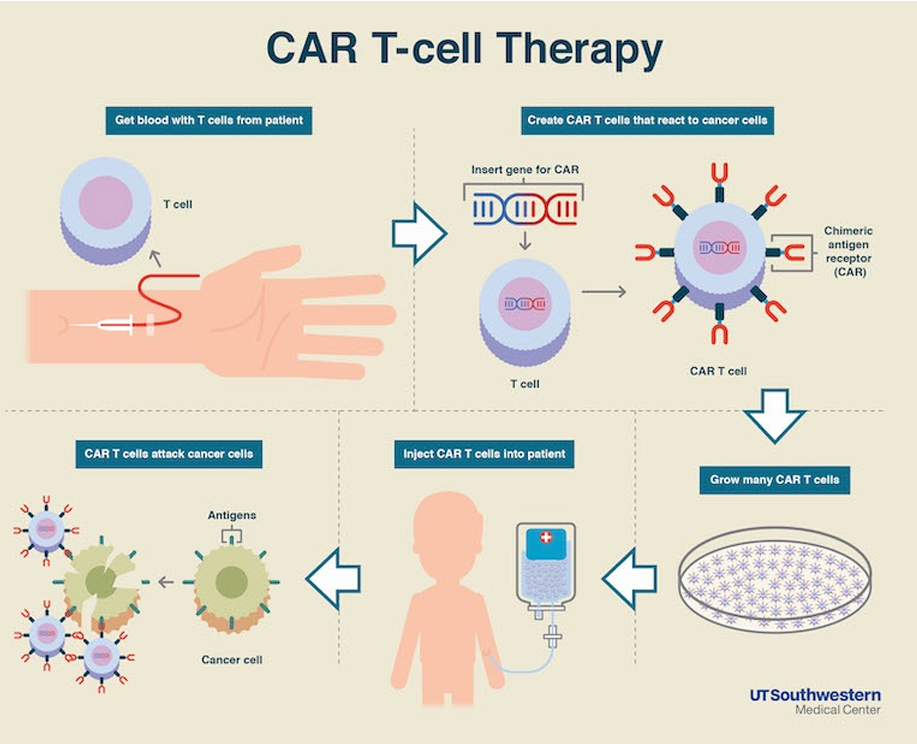
CAR T-cell therapy: a revolutionary treatment that involves genetically modifying a patient's own T-cells to target and destroy cancer cells more effectively.
Conclusion
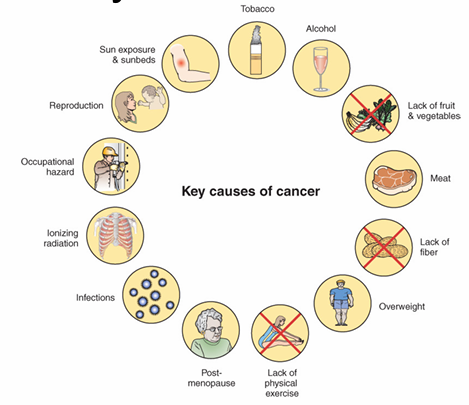
Summary of key causes of cancer includes:
Smoking, alcohol, occupational hazards, diet, lack of physical activity, and sun exposure.