Water Pollution
Water Pollution
**We use about 6400 L of water/day**
What pollutes water?
- Oil spills
- Microplastic
- Sewage
- Solid waste
Point source
- Pollution discharged from a single source
- Ex. sewage, industrial, oil, mining
Non-Point source
- Pollution discharged from many sources
- Ex. agriculture runoff, road salt, animal waste, precipitation
- Harder to control
The water cycle
- Pollution in one lake doesn't just stay there, but transmits to other bodies of water due to the connection of water around the world
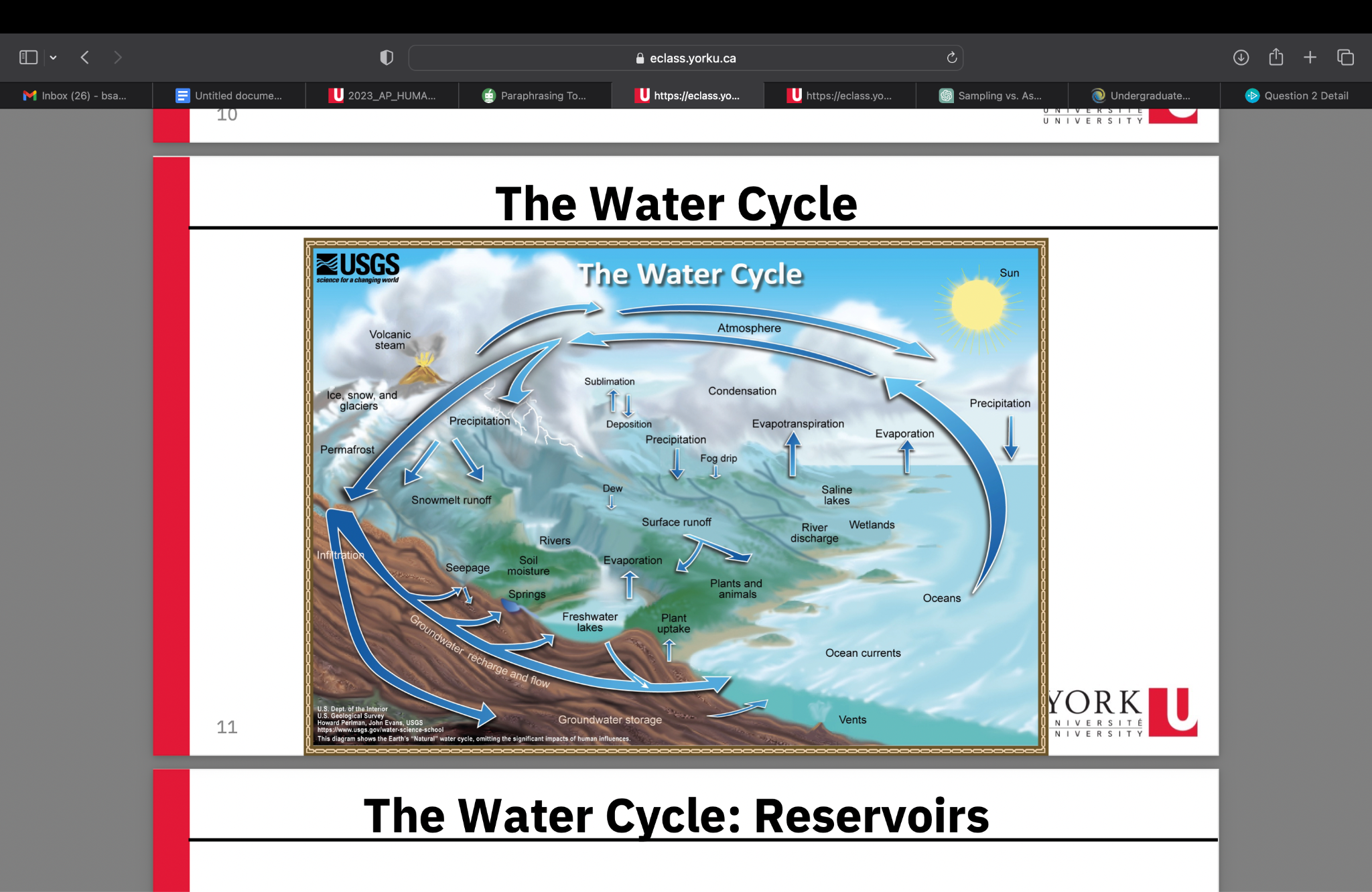
Reservoir: Location where water is stored
Major water Reservoirs:
- Oceans
- Freshwater
- Groundwater
- Glaciers
- Surface waters (lakes, rivers)
- Atmosphere
Transport process of the water cycle (TO ATMOSPHERE)
Evaporation: as the sun heats up water (ocean) it changes from a liquid to a gas (vapour), transporting water into the atmosphere
Sublimation: ice and snow can sublimate directly into water vapor
(solid water -> gaseous water)
Evapotranspiration: evaporated water specifically from plants and soils
Transport process of the water cycle (FROM ATMOSPHERE)
Condensation: water vapour in the atmosphere cools and condenses into clouds
Precipitation: if a cloud grows large enough it is able to fall out of the sky as precipitation (snow, rain, hail)
Deposition: transfer of water vapour directly to a solid water (ice/snow)
Transport process of the water cycle (ALONG earth's surface)
Surface runoff: because of gravity, precipitation flows over the ground until it ends up in a river/stream (will flow into a lake/ocean)
Infiltration: because of gravity, precipitation can leak into groundwater and recharge into the lake/ocean
Conventional Pollutants
- Biochemical oxygen demand (BOD)
- Nutrients
- Suspended soils
- pH
- Oil and grease
- Pathogenic microorganisms
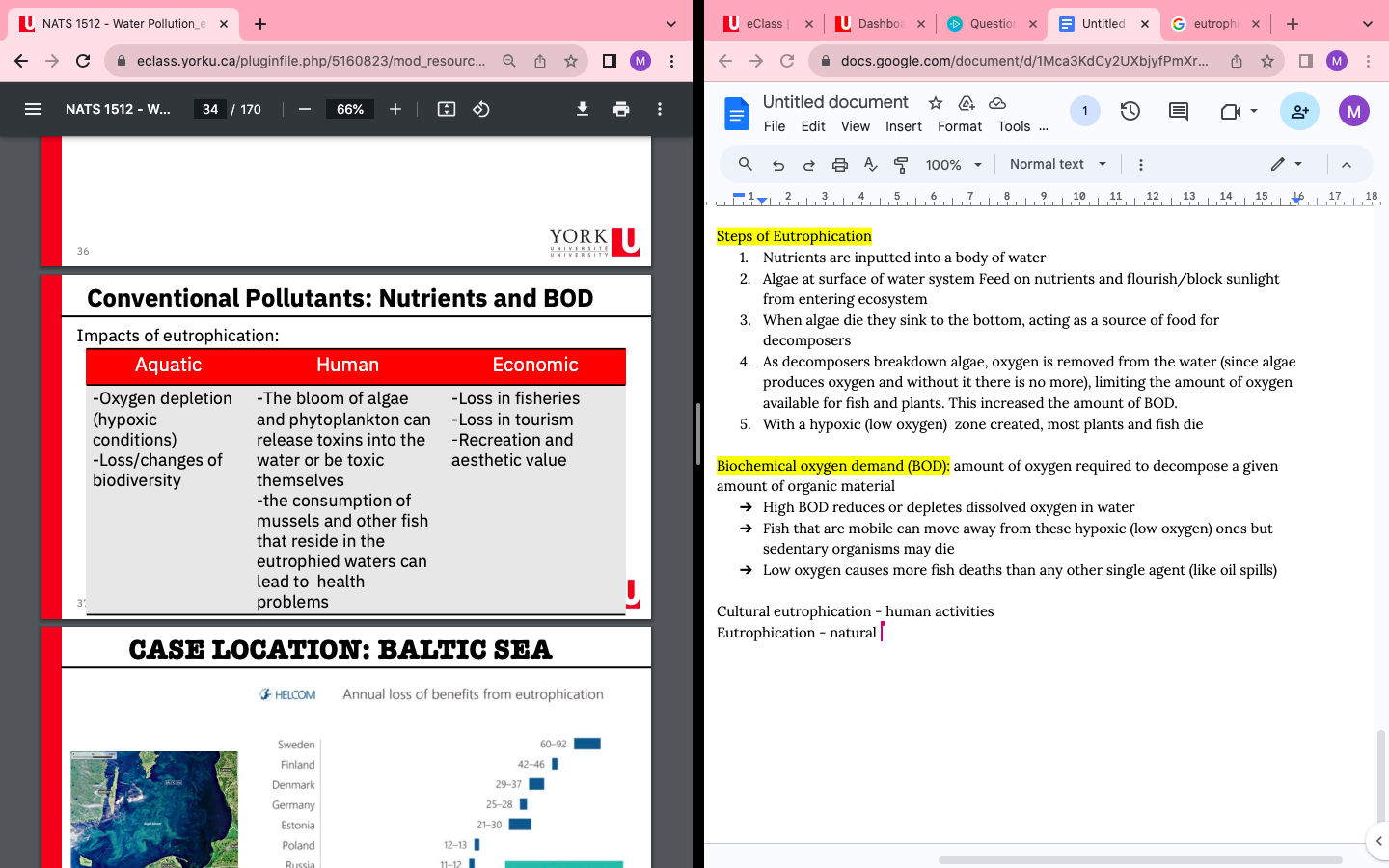
Conventional Pollutants: Nutrients and BOD
- Nutrients such as Nitrogen and phosphorus are naturally occurring in our environment BUT excessive inputs of N and P into water systems is a direct result of human activities
Primary sources of nutrient pollution
- Runoff of fertilizers from agricultural fields
- Animal manure
- Sewage and wastewater
- Laundry detergents and soap
- Excess nitrogen in a water systems can lead to cultural eutrophication:
- Eutrophication: Process of nutrient enrichment and subsequent ecosystem degradation (natural process, can occur without human activities)
Steps of Eutrophication
- Nutrients are inputted into a body of water
- Algae at surface of water system Feed on nutrients and flourish/block sunlight from entering ecosystem
- When algae die they sink to the bottom, acting as a source of food for decomposers
- As decomposers breakdown algae, oxygen is removed from the water (since algae produces oxygen and without it there is no more), limiting the amount of oxygen available for fish and plants. This increased the amount of BOD.
- With a hypoxic (low oxygen) zone created, most plants and fish die
Biochemical oxygen demand (BOD): amount of oxygen required to decompose a given amount of organic material
- High BOD reduces or depletes dissolved oxygen in water
- Fish that are mobile can move away from these hypoxic (low oxygen) ones but sedentary organisms may die
- Low oxygen causes more fish deaths than any other single agent (like oil spills)
Cultural eutrophication - human activities
Eutrophication - natural
Conventional Pollutants: Suspended soils
- Vegetation and root structure protects soil from erosion and transport
- Mining, construction, land clearing, etc removes vegetation and the protection becomes exposed to wat4r
- Low SS (no sand or soil that blocks the sunlight, this way photosynthesis still continues)
- High SS (high sand or soil which buries aquatic organisms that are attached to rocks removed by sediments washing along the bottom)
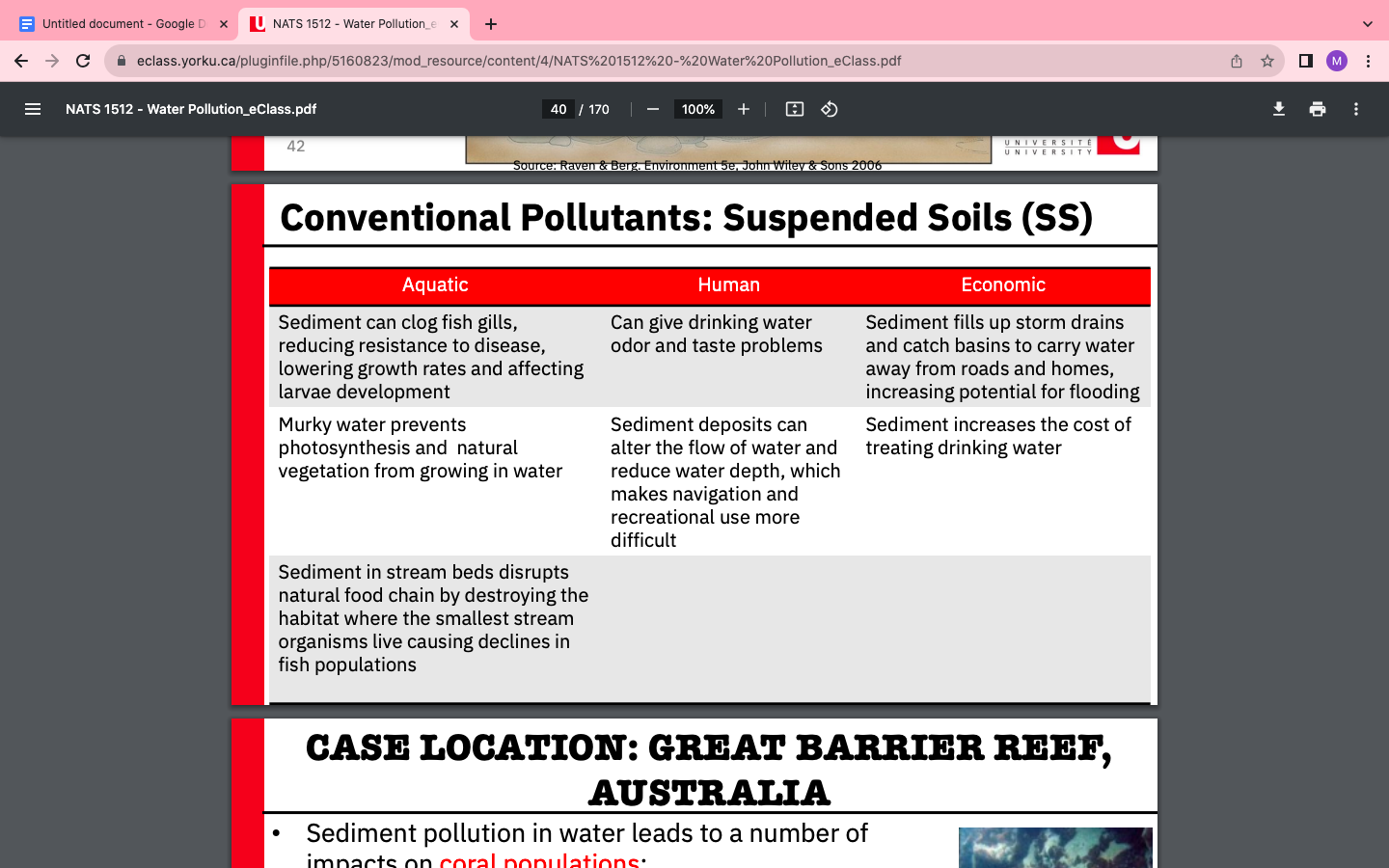
Sediment pollution in water leads to a number of impacts on coral populations:
- • Reduced photosynthetic efficiency (from shading caused by sediment)
- • Change in coral colour arising from changes in the density of photosynthetic pigments (coral bleaching!)
- • Increased ciliary or polyp activity to remove sediment
- • Increased mucus production to remove sediment
- • Injury to coal tissue
- • Mortality of small sized colonies, decreased density of coral cover
Conventional Pollutants: pH - A scale of acidity from 0 to 14
- Acidic solutions have a pH less than 7.00
- Basic/Alkaline solutions have a pH greater than 7.00
- Neutral solutions have a pH = 7.00 50 Conventional Pollution
- PH 4 is 1000 times more acidic than pH 7
- pH 2 is 100,000 times more acidic than pH 7
- Count by 0 (if there's 8 in between each number, than there should be 8 zeros)

NOx → nitric acid
SO2 → sulphuric acid
- Typically, a lake unaffected by human activities has a pH of 6-7
- If H2SO4 or HNO3 is delivered into an aquatic ecosystem (in significant quantities) it can lower the natural pH of the ecosystem (making the water more acidic)
- The problem: certain fish and microorganisms can only survive in particular pH ranges!
- Acidic waters have also been associated with reproductive failures as well as mutations in fish and amphibians
- Reproductive failures and mutations in fish and amphibians are associated with acidic water
Indirect Impact of pH: Aluminium stress
- If acid interacts with soil near an aquatic ecosystem, it can dissolve aluminum (Al) present in the soil, freeing it to runoff into nearby water bodies
- High concentrations of Al in water bodies causes stress on fish species, causing higher rates of mortality Indirect Impact of pH: Aluminum stress
- Figure shows the impact of pH on fish samples in each of the 1460 Adirondack lakes
Case study: Walkerton Ontario
- Contamination of drinking water has an effect on individuals health
- Caused 2300 people to be sick, flu like symptoms, irregular bowel movements
- These residents can no longer open their tap and drink from it
Conventional Pollutant: Pathogen
- A bacterium, virus, fungus, protozoan, toxic algal species
- Can be found in water
- ex , E-coli (lettuce, undercooked red meat )
- Ex. clostridium perfringens (when food is poorly handled and left to sit in
unsafe temp)
Nonpoint sources of Pathogens
- Pathogenic microbes in a water body often result from human activities
- Storm water runoff
- Cities with high degree of imperious surfaces are especially susceptible to storm water runoff sources of pathogens
Point Source of Pathogens
- Poorly functioning sewage treatment plants
- During heavy rainfall water treatments plants can be bypassed to avoid flooding
- Deliver untreated water including sewage into body of water
Pathogens can be harmful to other forms of life if:
- • Ingestion (the fecal-oral route)
- • Contact with the water (swimming, bathing, etc.)
- • Inhalation and aspiration
- Presence of these pathogens in the human body can lead to the development of a variety of diseases
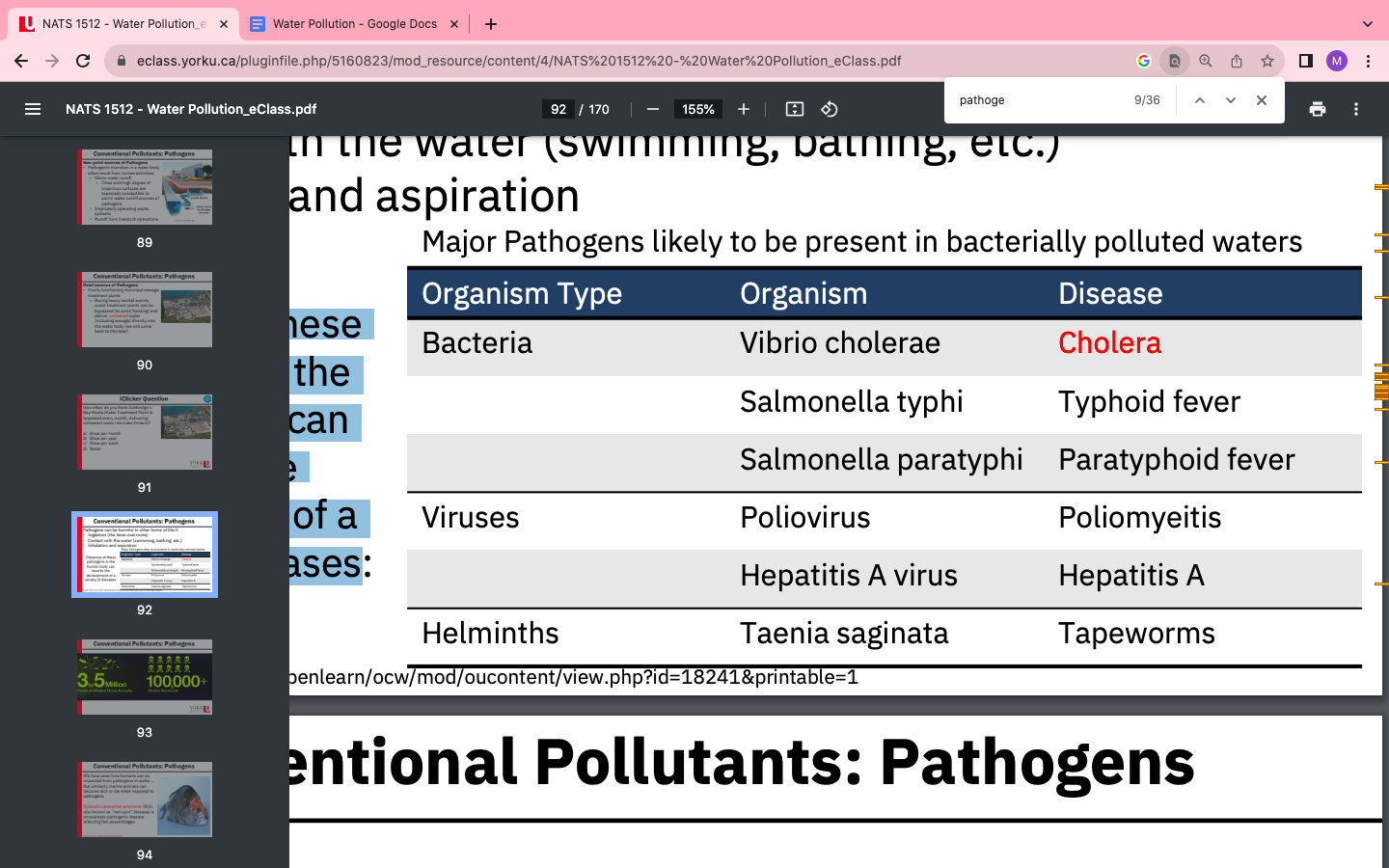
- 3-5 mill cases of Cholera occur annually
Pathogens affect on marine animals
- Epizootic ulcerative syndrome
- (EUS, also known as ”red-spot” disease) is an example pathogenic disease affecting fish assemblages
- Coral Diseases
- Occur in response to biological stresses such as bacteria, fungi and viruses
- Frequency of coral diseases has increased significantly over the last 10 years due to deteriorating water quality associated with human made pollutants
- E.g. Yellow–band disease can rapidly spread over a coral, destroying the tissue and exposing the underlying white coral skeleton (coral bleaching)
- Persistence of pathogens
- Depends on the particular pathogen
- Naturally occurring, free-living pathogens can survive indefinitely
- Other pathogens (e.g. from faecal contamination sources) have limited survival periods outside the host animals/humans
- Sediment acts as a reservoir for viruses
- Factors that influence persistence of pathogens include: water temperature, UV/sunlight, dispersion and dilution and salinity
What happened in Walkerton: E Coli
- E Coli comes from cattle manure
- A farm near the one of the wells where water was supplied to the town was spread on farmland early in May 2000
- After a heavy rainfall the manure made its way into the water supply system
Priority toxic pollutants
- Priority (toxic) pollutants includes 126 pollutants, including the metals: arsenic, cadmium, lead, nickel, copper, chromium, mercury, and zinc
- Also includes other toxic substances such as pesticides (e.g. heptachlor and dieldrin) and polychlorinated biphenyls (PCBs) used for electrical insulation
- Regulated by EPA (in USA) or Canadian Environmental Protection Act (CEPA) (in Canada)
A Case Study: DDT
- DDT (dichloro-diphenyltrichloroethane) was developed as the first modern synthetic insecticide in the 1940s
- Initially used to combat malaria, typhus and other insect-borne human diseases among both military and civilian populations
- It was promoted as a “wonder-chemical”
- DDT acts to disturb the function of nerves in insects and humans creating the syndrome: “DDT Jitters”
- The function of the nerve becomes uncontrolled
- largest problem associated with DDT is that is has a long lifetime in the environment (it is persistent)
- may take 5-25 years for soil to lose 95 % of the DDT
Ecological effects of: DDT
- May be slightly toxic to birds
- Very highly toxic to many aquatic invertebrate species and fish species
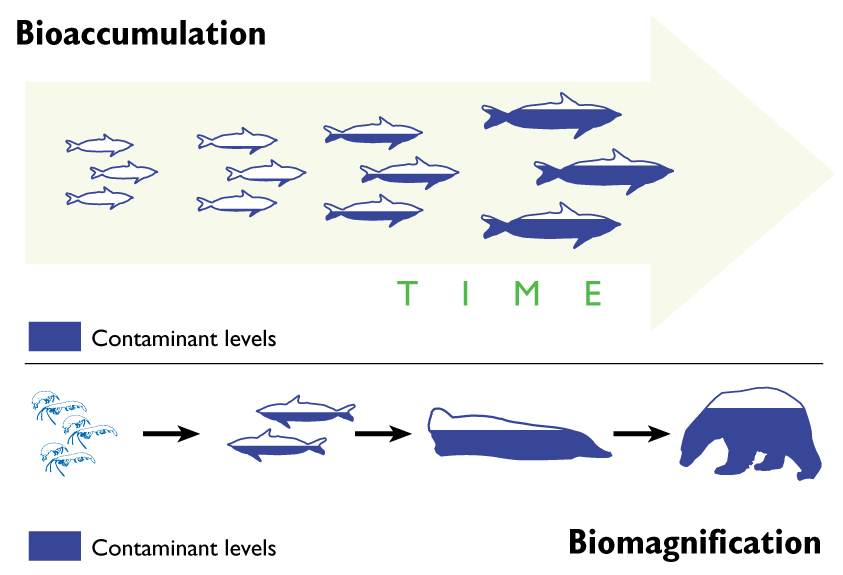
- DDT may bioaccumulate and biomagnify
Bioaccumulation:
- increase in concentration of a pollutant in a single organism over its lifetime
Biomagnification:
- increase in concentration of a pollutant up the food chain
Non-conventional & non toxic pollutants
This third category of pollutants include:
- Road salt
- Iron
- Aluminium
- Colour (dyes)
- Heat
- Etc…
Impacts of road salt on aquatic ecosystem
- Road salt (like our table salt) is made up of sodium (Na) and chlorine (Cl) (i.e. road salt = NaCl)
- After application of road salt on our highways and streets, eventually it will runoff into nearby soil or an aquatic ecosystem
- Chloride is toxic to aquatic life: inhibits aquatic species growth and reproduction, impact food sources and disrupt osmoregulation in amphibians
- It can lead to a hypoxic zone in freshwater systems by blocking the gas exchange within water
- Salt is also corrosive (we this when salt accumulates on our cars in wintertime)
- When water is flowing through pipes that have elevated salt concentrations, it is possible for the pipes to erode, releasing heavy metals into the water (a possible suspect for the Flint, Michigan water crisis)
3 major sources of water pollution
- Conventional water pollutants (nutrients, BOD, suspended soils, pH, oil and grease, pathogens)
- Priority (toxic) pollutants
- Non Conventional and nontoxic pollutants
What is done with this plastic?
- 1. Recycled(8-9%)
- 2. Incinerated(Waste-to-Energy,12%)
- Atmospheric pollution
- 3. Put in Landfill(79%)
- Aquatic pollution
- 4. Mismanaged(litter)
- Terrestrial and aquatic pollution
Plastic in landfills
- Majority of plastic waste (80-90%) is deposited into a landfill
- Land fill inputs
- Waste
- Water (rainfall)
- Landfill outputs
- Leachate
- As rainwater flows through landfills, it dissolves all the water-soluble compounds that the waste contains
- Leachate from plastic in landfills has been found to create (BPA, phthalates, microplastics, brominated flame retardants
- Does not remain in landfill although it does enter the groundwater being transported to aquatic ecosystems
- Biogas (CH4)
- Leachate
Plastic in oceans
- 14 million tonnes of plastic is floating around in our oceans
- Expose a physical and chemical threat to ecosystems
- Physical threat
- Ingestion of plastic
- Can lead to malnutrition and starvation
- Suffocation
- Drowing
- This is most common in sea turtles
- Entanglement of plastic
- Caught in nets
- Known as ghost fishing (when fish are stuck in the nets)
- Ingestion of plastic
- Chemical threat
- Bioaccumulation
- Increase in pollutant in a single organism over its lifetime
- Biomagnification
- Magnification of pollutant up the food chain
- Microplastic
- Degraded plastic that are broken into smaller pieces
- A major concern associated with ingestion of marine microplastics is that they absorb chemicals from the water, making them vectors of these toxic substances if they are ingested
- Bioaccumulation
Water treatment
- 9 steps of treatment and testing that must be performed from when water in collected from Lake Ontario before it is delivered to homes
- 1. Collection from Lake Ontario
- Water intake pipes extend into Lake Ontario and collect raw water. In some areas of the city, large intake pipes extend as far as 5KM offshore
- 2. Screening
- At the entrance of the plant large travelling screens remove large objects and debris
- 3. Chlorine
- Added to lake water to kill microorganisms (bacteria, algae, fungi)
- Pre-chlorination step
- Alum is added to allow microorganisms to clump together for removal of chemicals (known as floc)
- 4. Settling Basin
- Heavy flocs drop out of water in a settling tank (collect along bottom)
- Floc is removed from water
- Cleaner water is left at surface to be drawn off through the spillways, leading to filtering of basins
- 5. Filtration
- Made up of layers of graded gravel, find sand and carbon
- Layer of sand removes find bits of floc, alega or slit
- Chemicals, physical impurities, or biological impurities are mostly removed
- Layer of activated carbon removes taste and odour-producing chemicals from the water.
- 6. Storage
- Purified water goes into holding basins prior to distribution
- Safe levels of (extra) chlorine are added to kill any harmful microorganisms
- Excess chlorine is removed with sulphur dioxide
- In toronto water is fluoridated to allow for fewer cavities
- 7. Ammonia
- Last step = add ammonia to water
- Ammonia combines with remaining chlorine to stabilize it and keep water safe
- During trips from the plant, through distribution pipes ot peoples homes and buildings
- 8. Quality Tests
- Treated water is tested regularly to ensure quality
- 9. Distribution
- With the help of high lift pumps, reservoirs and pumping stations, water is distributed to homes, apartments, schools and industries.
Guidelines for drinking water (MACs)
- MACs= maximum acceptable concentrations
- Macs must be completed before sending water to residents
- Tests are complete for
- Microbiological parameters
- Chemical and physical parameters
- Radiological parameters
Microbiological Parameters
- These are the highest priority guidelines, ensuring low levels of bacteria, protozoa and viruses
- Ex. e coli, total coliforms
Chemical and Physical Parameters
- Attempts to reduce chemical contaminant should not compromise the effectiveness of disinfection
- Ex. arsenic, atrazine, lead, mercury