18/3 Gluconeogenesis, Glycogen & Glucagon Signalling
LI
gluconeogenesis - glucose production from non-carbs
glycogen stores glucose
gluconeogenesis controlled by glucagon
key steps of gluconeogensis is controlled by phosphorylation
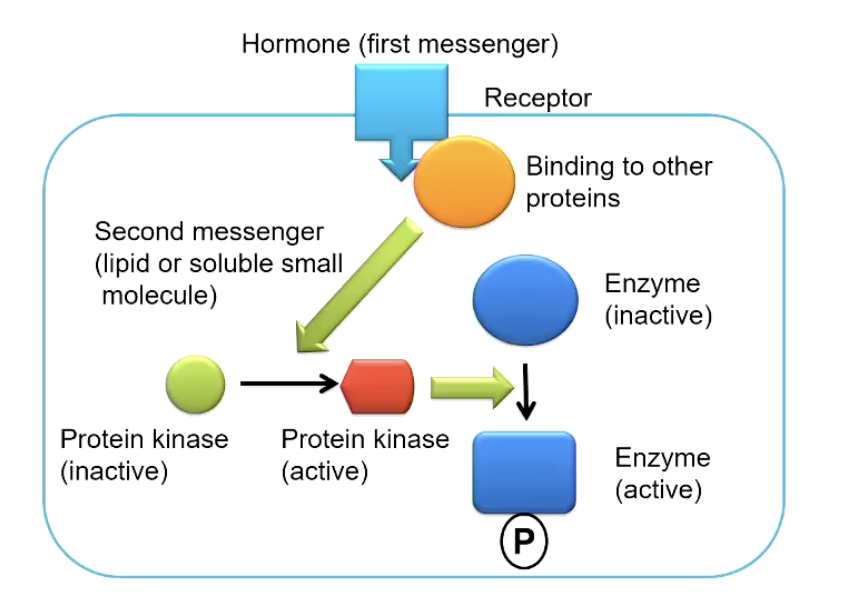
signal transduction
Physiological Context & Importance of Glucose
Humans can survive ~1 month without ingesting calories provided water is available.
Maintaining blood glucose at ≈4–5 mM is critical.
Mild hypoglycaemia (≈3–4 mM): lethargy, hunger.
Severe hypoglycaemia (<3 mM): poor judgement ➝ disorientation ➝ coma ➝ death.
Two complementary strategies keep plasma glucose constant when dietary carbohydrate is absent:
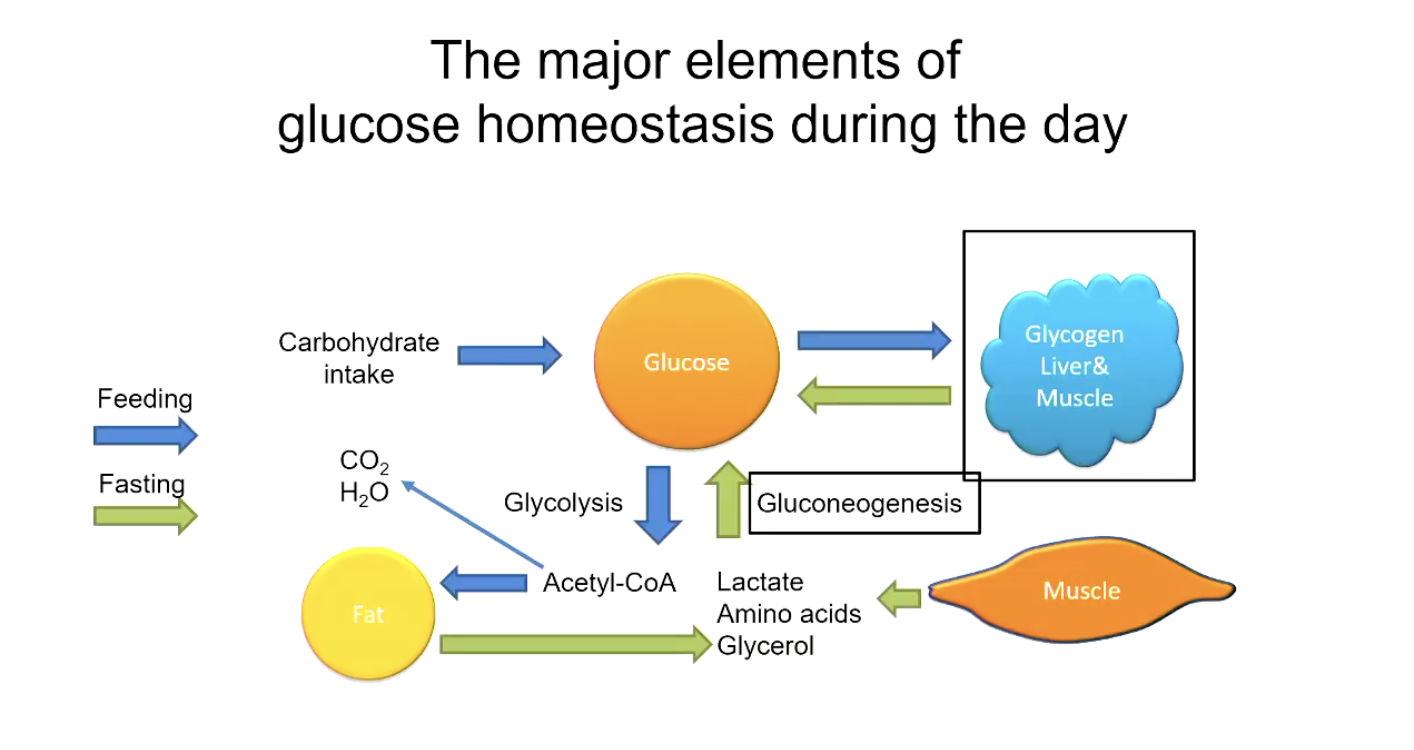
Glycogenolysis – mobilises stored glycogen (rapid, short-term).
Gluconeogenesis (GNG) – de-novo synthesis of glucose from non-carbohydrate precursors (slower, long-term).
Major Pathways – Direction & Location
Glycolysis (red in slides): Glucose ⟶ Pyruvate (energy-yielding, generating ATP). Predominantly cytosolic, universal.
Gluconeogenesis (blue/green arrows): Pyruvate/PEP ⟶ Glucose (energy-consuming). Mainly in liver; minor contribution from kidney cortex (especially from glutamine).
Key organs
Liver: central to both glycogen turnover & gluconeogenesis; exports glucose to blood.
Kidney: backup GNG (≈10 % during prolonged fast).
Skeletal muscle: large glycogen store for local use; lacks glucagon receptors, therefore not a source of blood glucose.

Non-Carbohydrate Precursors Feeding GNG
Lactate – generated during anaerobic glycolysis in muscle/RBCs; enters liver via Cori cycle.
Amino acids (esp. alanine & glutamine) – released from muscle proteolysis.
Glycerol – liberated from adipose triglyceride breakdown; enters pathway at dihydroxy-acetone-phosphate (DHAP).
Fatty-acid carbon cannot yield net glucose, but glycerol backbone can.
Irreversible Steps & Bypass Reactions
Certain glycolytic steps have large negative free energies ( \Delta G^{\circ '} \ll 0 ) and must be circumvented in GNG:
Pyruvate kinase step
Glycolysis: PEP→pyruvate
GNG bypass: Pyruvate →oxaloacetate→PEP (pyruvate carboxylase)
PFK-1 step
Glycolysis: Fructose-6-P ⟶ Fructose-1,6-bis-P.
GNG bypass: Fructose-1,6-bis-P ⟶ Fructose-6-P (Fructose-1,6-bisphosphatase-1).
Hexokinase/Glucokinase step
Glycolysis: Glucose ⟶ Glucose-6-P.
GNG bypass (liver, kidney): Glucose-6-P ⟶ Glucose + P_i (Glucose-6-phosphatase, ER-membrane).
Hormonal Control – Insulin vs Glucagon
insulin - stops sugar production
glucagon - encourages sugar production
Fed state (high glucose):
↑ Insulin; ↓ Glucagon.
Promotes glycogen synthesis & glycolysis; suppresses GNG.
Fasting state (low glucose):
↓ Insulin; ↑ Glucagon.
Activates glycogenolysis, GNG, lipolysis.
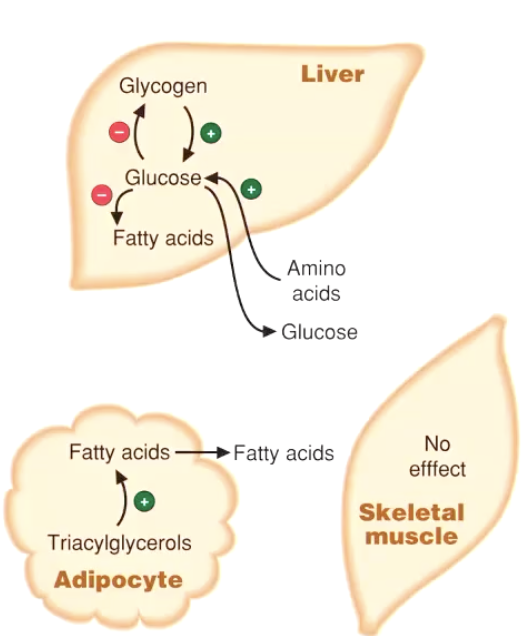
Glucagon target tissues
Liver & adipose tissue: YES.
Skeletal muscle: NO (critical exam fact).
Adrenaline (epinephrine) fulfils glucagon-like role in muscle during exercise.
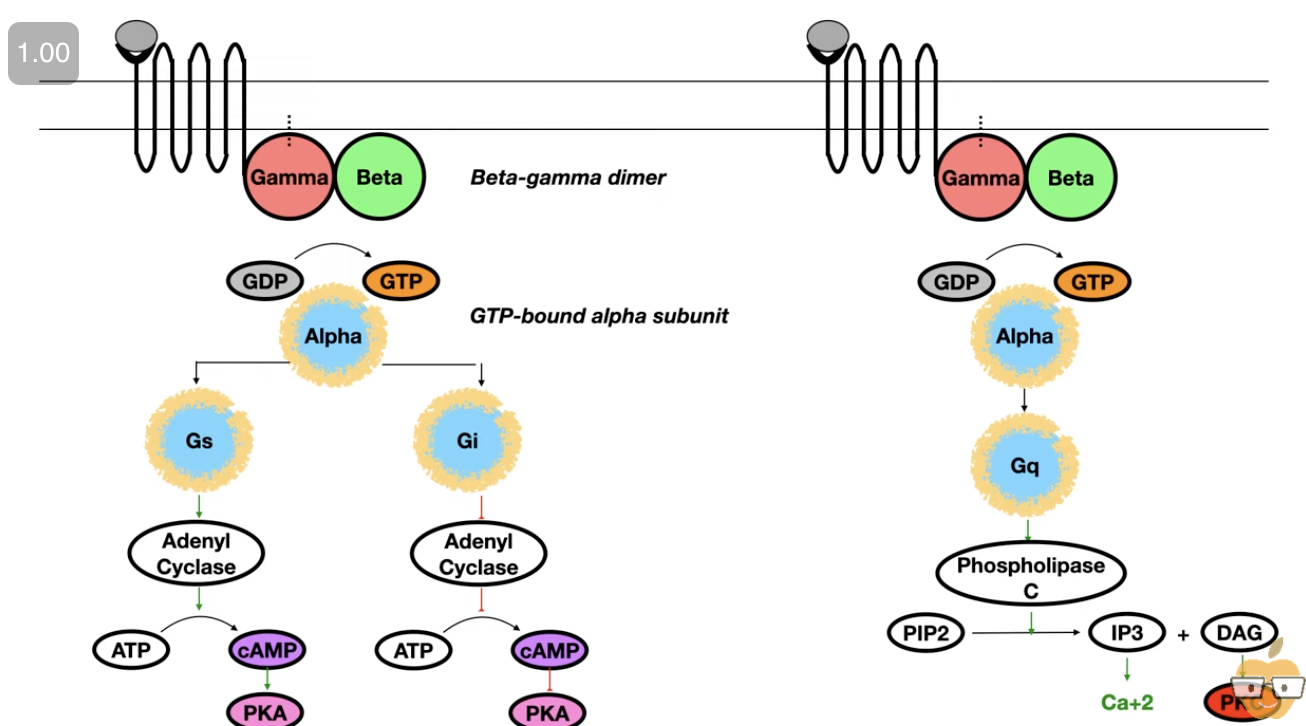
Glucagon Signal-Transduction Cascade (Classical G_s Pathway)
In essence, signal transduction allows glucagon's message to quickly and efficiently turn off glucose-consuming processes and turn on glucose-producing processes like gluconeogenesis in the liver, helping to maintain stable blood sugar levels
1° messenger: Glucagon (peptide) binds GPCR (7-TM receptor) on hepatocyte membrane.
Receptor undergoes conformational change ➝ activates heterotrimeric G_s protein (GDP → GTP exchange on G_\alpha).
G_\alpha(GTP) dissociates and stimulates Adenylyl cyclase (AC).
AC converts ATP → cAMP (2° messenger). 1 receptor → many G_proteins → many cAMP (AMPLIFICATION).
Protein kinase A (PKA) binds 4 cAMP → releases catalytic subunits → broad phosphorylation program.
Termination: cAMP → AMP by phosphodiesterase; intrinsic GTPase activity resets G_\alpha.
Enzyme Regulation by Phosphorylation
Residues modified: Ser, Thr, Tyr (hydroxyl-containing).
Phosphorylation can either activate OR inhibit a target enzyme (conformational/charge effects).
Added negative charge alters electrostatic & H-bond network.
Kinase ⇆ phosphatase opponent pairs allow rapid reversibility.
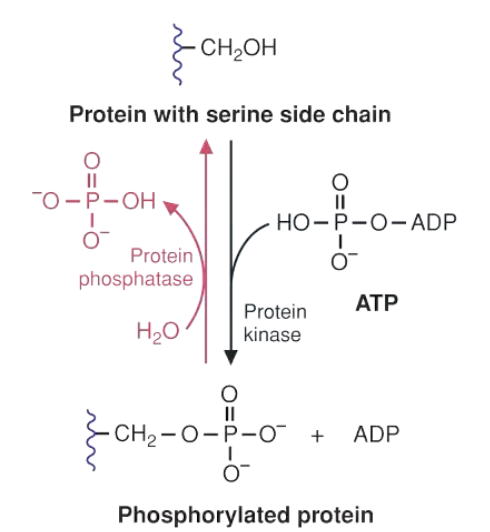
protein kinase: adds phosphate to amino acid side chain
protein phosphatase: removes phosphate from amino acid side chain
Key PKA Targets in Carbohydrate Metabolism
1. Pyruvate kinase (liver isoform)
PKA phosphorylation → inactive form.
Consequence: blocks final glycolytic step, diverts PEP toward GNG.
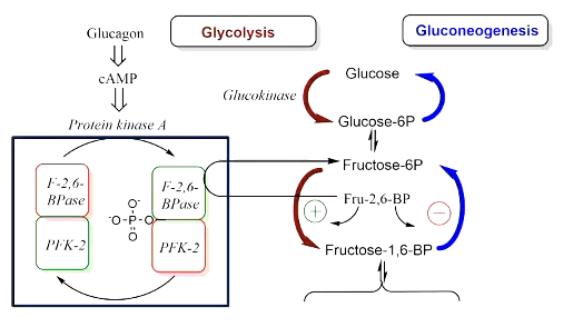
2. Bifunctional Enzyme PFK-2 / FBPase-2
Single polypeptide with kinase domain (makes Fructose-2,6-bis-P) & phosphatase domain (hydrolyses it).
Fructose-2,6-bis-phosphate (F2,6BP)
Potent allosteric activator of PFK-1 (↑ glycolysis).
Potent allosteric inhibitor of FBPase-1 (↓ GNG).
Phosphorylation by PKA switches OFF the kinase domain & ON the phosphatase domain → ↓ F2,6BP.
↓ PFK-1 activity + ↑ FBPase-1 activity → glycolysis slows, GNG accelerates.
3. Glycogen Metabolic Enzymes (covered briefly)
PKA cascade (via phosphorylase kinase) activates glycogen phosphorylase (breakdown) & inactivates glycogen synthase (synthesis).
Glycogen Metabolism
Absolutely, Raine — glycogen metabolism is like your body’s glucose savings account, with deposits and withdrawals happening based on energy needs. Let’s break it down into two main processes:
---
### 🏗 Glycogenesis (Glycogen Synthesis)
This is how your body stores excess glucose after a meal:
1. Glucose → Glucose-6-phosphate (via hexokinase/glucokinase)
2. G6P → Glucose-1-phosphate (via phosphoglucomutase)
3. G1P + UTP → UDP-glucose (activated form)
4. UDP-glucose → Glycogen (via glycogen synthase)
5. Branching enzyme adds α-1,6 linkages for compact storage
🧠 Triggered by: Insulin, high blood glucose, and low energy demand.
---
### 🔓 Glycogenolysis (Glycogen Breakdown)
This is how your body releases glucose when energy is needed:
1. Glycogen → Glucose-1-phosphate (via glycogen phosphorylase)
2. G1P → G6P (via phosphoglucomutase)
3. In liver: G6P → Glucose (via glucose-6-phosphatase) → enters bloodstream
In muscle: G6P enters glycolysis for local ATP production
🧠 Triggered by: Glucagon (liver), epinephrine (muscle), and low blood glucose.
---
### 🔄 Regulation: The Balancing Act
| Hormone | Effect |
|-------------|------------|
| Insulin | Activates glycogenesis, inhibits glycogenolysis |
| Glucagon | Activates glycogenolysis in liver |
| Epinephrine | Activates glycogenolysis in muscle and liver |
| AMP/ATP | Allosterically regulate enzymes based on energy status |
Phosphorylation plays a key role:
- Glycogen synthase is inactivated by phosphorylation
- Glycogen phosphorylase is activated by phosphorylation
---
Would you like a visual map of these pathways or a quick quiz to test your understanding?
Representative Equations (contextual)
Overall hepatic gluconeogenesis (from 2 pyruvate):
2pyruvate+ 4ATP + 2GTP + 2NADH + 6H2O →glucose+ 4ADP + 2GDP + 6Pi + 2NAD + 2HUDP-glucose formation:
\text{Glucose-1-P} + UTP \rightarrow UDP\text{–Glucose} + PP_iAdenylyl cyclase reaction:
ATP \xrightarrow{AC} cAMP + PP_i
Ethical / Practical Considerations
Understanding hormone-driven glucose homeostasis underpins treatment of diabetes & hypoglycaemia.
Pharmacological manipulation (e.g., cAMP-phosphodiesterase inhibitors, PKA modulators) can affect metabolism & are active areas of drug discovery.
Key Points for Exam Study
Physiological Context & Glucose Homeostasis
Maintaining blood glucose at "\approx4–5\,mM" is critically important.
Understand the consequences of severe hypoglycaemia (<3 mM): poor judgement, disorientation, coma, death.
Two Core Strategies for Glucose Maintenance
Glycogenolysis: Rapid, short-term glucose release from stored glycogen.
Gluconeogenesis (GNG): Slower, long-term de-novo synthesis of glucose from non-carbohydrate precursors.
Major Pathway Directions & Locations
Glycolysis: Glucose "\longrightarrow" Pyruvate (cytosolic, universal, energy-yielding).
Gluconeogenesis: Pyruvate "\longrightarrow" Glucose (mainly liver, minor kidney contribution, energy-consuming).
Key Organ Roles
Liver: Central for both glycogen turnover and GNG; exports glucose to blood.
Kidney: Provides backup GNG (≈10% during prolonged fast).
Skeletal Muscle: Stores glycogen for local use only; lacks glucagon receptors and glucose-6-phosphatase, so cannot directly contribute glucose to blood.
Non-Carbohydrate Precursors for GNG
Lactate: Via Cori cycle from anaerobic glycolysis.
Amino acids: Especially alanine and glutamine from muscle proteolysis (Alanine cycle).
Glycerol: From adipose triglyceride breakdown (fatty acid carbon cannot yield net glucose).
Irreversible Glycolytic Steps and GNG Bypass Reactions
Memorize the three irreversible glycolytic steps and their corresponding GNG bypass enzymes:
Pyruvate kinase: Bypassed by Pyruvate carboxylase (mitochondrial) and PEP-carboxy-kinase (cytosolic/mitochondrial).
PFK-1: Bypassed by Fructose-1,6-bisphosphatase-1.
Hexokinase/Glucokinase: Bypassed by Glucose-6-phosphatase (liver, kidney).
Hormonal Control (Insulin vs. Glucagon)
Fed state: High insulin, low glucagon "\longrightarrow" favors glycogen synthesis & glycolysis, suppresses GNG.
Fasting state: Low insulin, high glucagon "\longrightarrow" activates glycogenolysis, GNG, lipolysis.
Crucial detail: Glucagon acts on liver & adipose, NOT skeletal muscle.
Adrenaline (epinephrine) plays a glucagon-like role in muscle during exercise.
Glucagon Signal-Transduction Cascade (Classical G"_s" Pathway)
Understand the steps: Glucagon (peptide) "\longrightarrow" GPCR "\longrightarrow" G"_s" protein activation "\longrightarrow" Adenylyl cyclase "\longrightarrow" cAMP (amplification) "\longrightarrow" Protein kinase A (PKA) activation "\longrightarrow" phosphorylation program.
Enzyme Regulation by Phosphorylation (PKA Targets)
Pyruvate kinase (liver): Phosphorylation by PKA inactivates it, shunting PEP towards GNG.
Bifunctional Enzyme PFK-2 / FBPase-2: Phosphorylation by PKA switches OFF kinase domain & ON phosphatase domain "\longrightarrow" "\downarrow F"2,6BP. This "\downarrow" PFK-1 (glycolysis) and "\uparrow" FBPase-1 (GNG).
Glycogen Metabolic Enzymes: PKA activates glycogen phosphorylase (breakdown) & inactivates glycogen synthase (synthesis) via phosphorylase kinase.
Glycogen Metabolism Key Steps
Synthesis: UDP-Glucose as precursor, Glycogen synthase, branching enzyme.
Breakdown: Glycogen phosphorylase, debranching enzyme.
Distinguish glucose fate: Liver G1P "\longrightarrow" Glucose (exported); Muscle G1P "\longrightarrow" Glycolysis (local use).
Integrated Cycles
Cori cycle: Lactate (muscle) "\leftrightarrow" Glucose (liver).
Alanine cycle: Transports nitrogen & carbon from muscle to liver for GNG.
Key Equations (Contextual)
Overall hepatic gluconeogenesis (from 2 pyruvate):2\,\text{Pyruvate} + 4\,ATP + 2\,GTP + 2\,NADH + 6\,H2O \rightarrow \text{Glucose} + 4\,ADP + 2\,GDP + 6\,Pi + 2\,NAD^+ + 2\,H^+