1.1 Alkanes
Organic Chemistry
- Organic Chemistry is the study of carbon-based compounds.
- Except CO, CO2, HCN. These are inorganic compounds.
- Organic compounds all contain: CHNOPS
- ^^Carbon^^
- ^^Hydrogen^^
- Nitrogen
- Oxygen
- Phosphorus
- Sulfure
Alkanes
- Called saturated hydrocarbons - they cannot fit any more hydrogens
- All bonds are single bonds
- General formula is CnH2n + 2
- An alkane with 4 carbons: C4H10 (Butane)
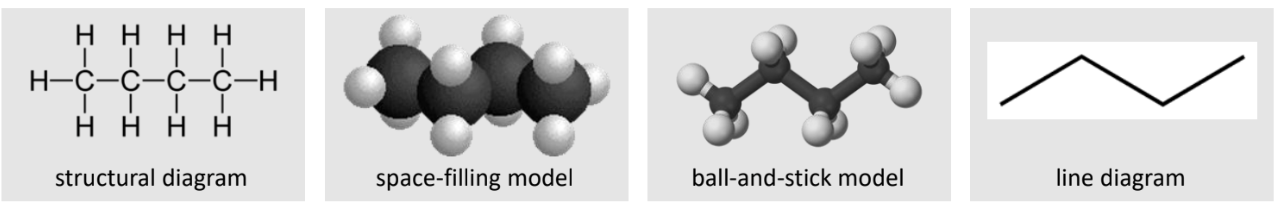
Representation of Molecules
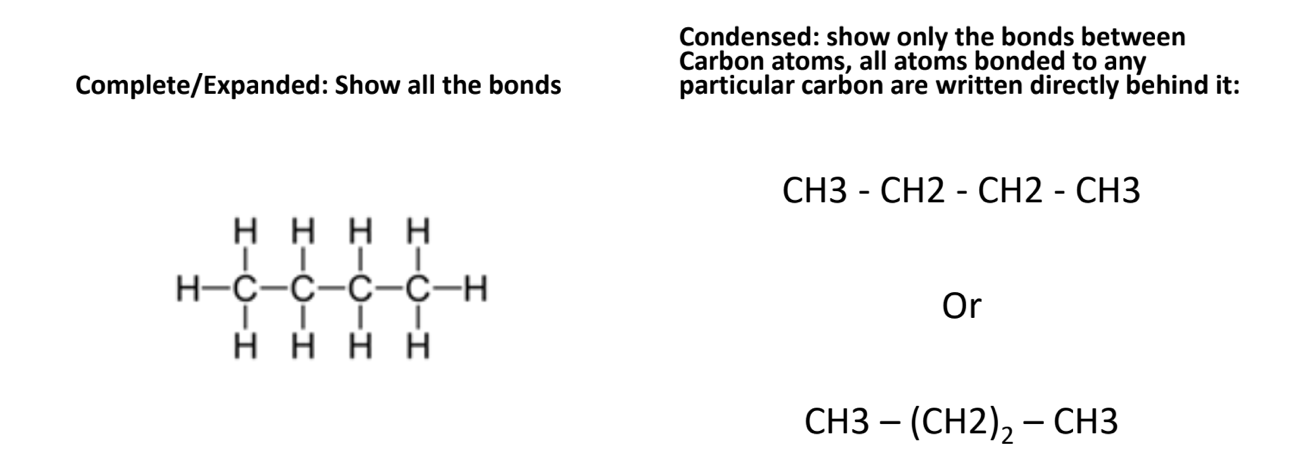
Structural diagram
- Complete/Extended: shows all bonds
- Condensed: show only the bonds between carbon atoms, all atoms bonded to any particular are written directly behind it
Space-filled model
Ball-and-stick model
Lined diagram
Molecular formula
Expended molecular formula
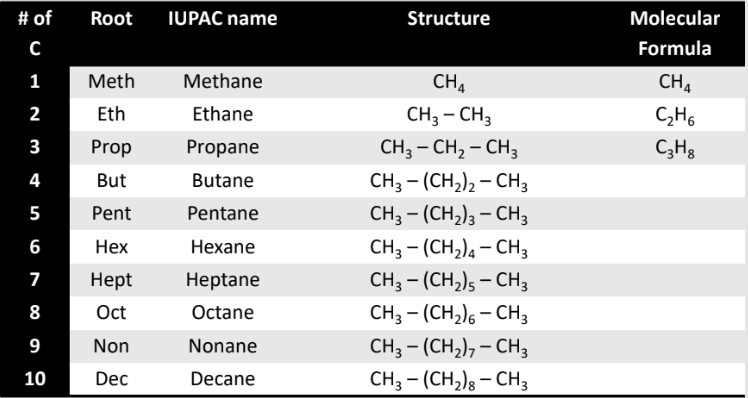
Naming Straight Chain Alkanes
The “root” of the name describes the number of carbons in the chain.
Cyclic Alkanes
- The chain is linked to form a closed loop.
- Add “cycle” as a prefix.
- Example: cyclobutane
Structural Isomers
compounds that have the same molecular formula, but different structure
A straight butane has the same molecular formula as a propane carbon branch
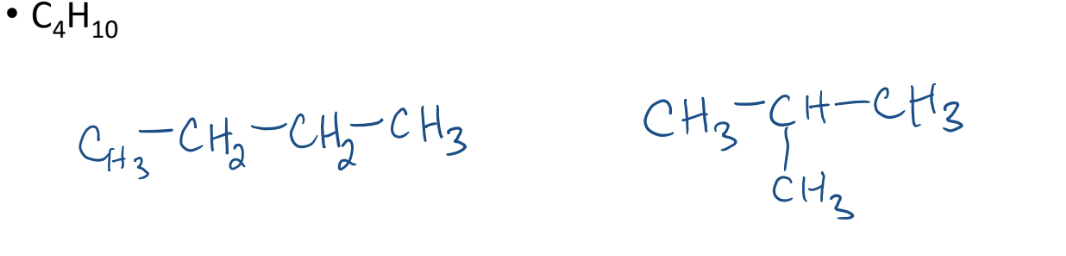
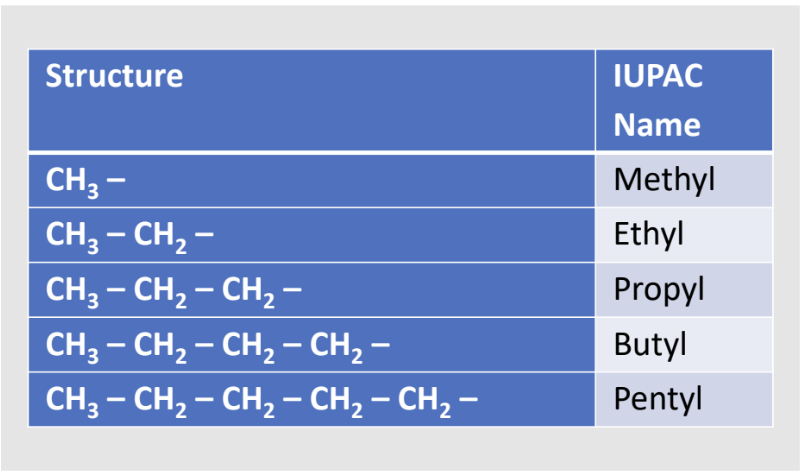
Substituent & Alkyl Groups
Substituent group: an atom or group of atoms that replaces a hydrogen in an organic compound.
Alkyl group: one or more carbon atoms that form a branch off the main chain of a hydrocarbon.
- If the group is attached to the main chain via a middle carbon, a number is inserted:
How to name alkanes
- Find the longest carbon chain (called the parent chain)
- Identify the substituent groups
- Number the parent chain so that the substituents are attached to the carbon atom with the smallest number. In case of a tie, the group that comes first alphabetically gets the lowest number
- If the same substituent is present more than once, use a prefix (di, tri, tetra) and include the number to indicate the substituent’s location
- List the substituents in alpha order (NOT including the di, tri tetra). Separate words by hyphens, numbers by commas.
Prefix + Root + Suffix
- Prefix: name and location of substituents
- Root: # of carbons in parent chain
- Suffix: type of hydrocarbon