Tissue distribution of drugs
Intended learning outcomes:
- Understand the role of drug binding to tissue components and its impact on drug distribution
- Understand the role of drug transporters on distribution and drug safety/efficacy
- Understand subcellular drug distribution
- Understand principles of in silico tools used to predict drug distribution
Tissue distribution of drugs:
- Tissue binding is often inferred from plasma data
- At equilibrium the tissue distribution of a drug within the body depends upon binding to both plasma proteins and tissue components
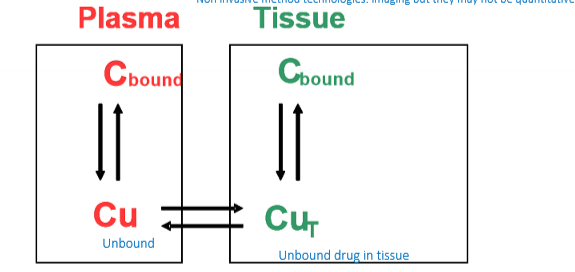
- The tissue- plasma partition coefficient (Kp) gives an idea of what extent drug may be potentially accumulating in the tissue


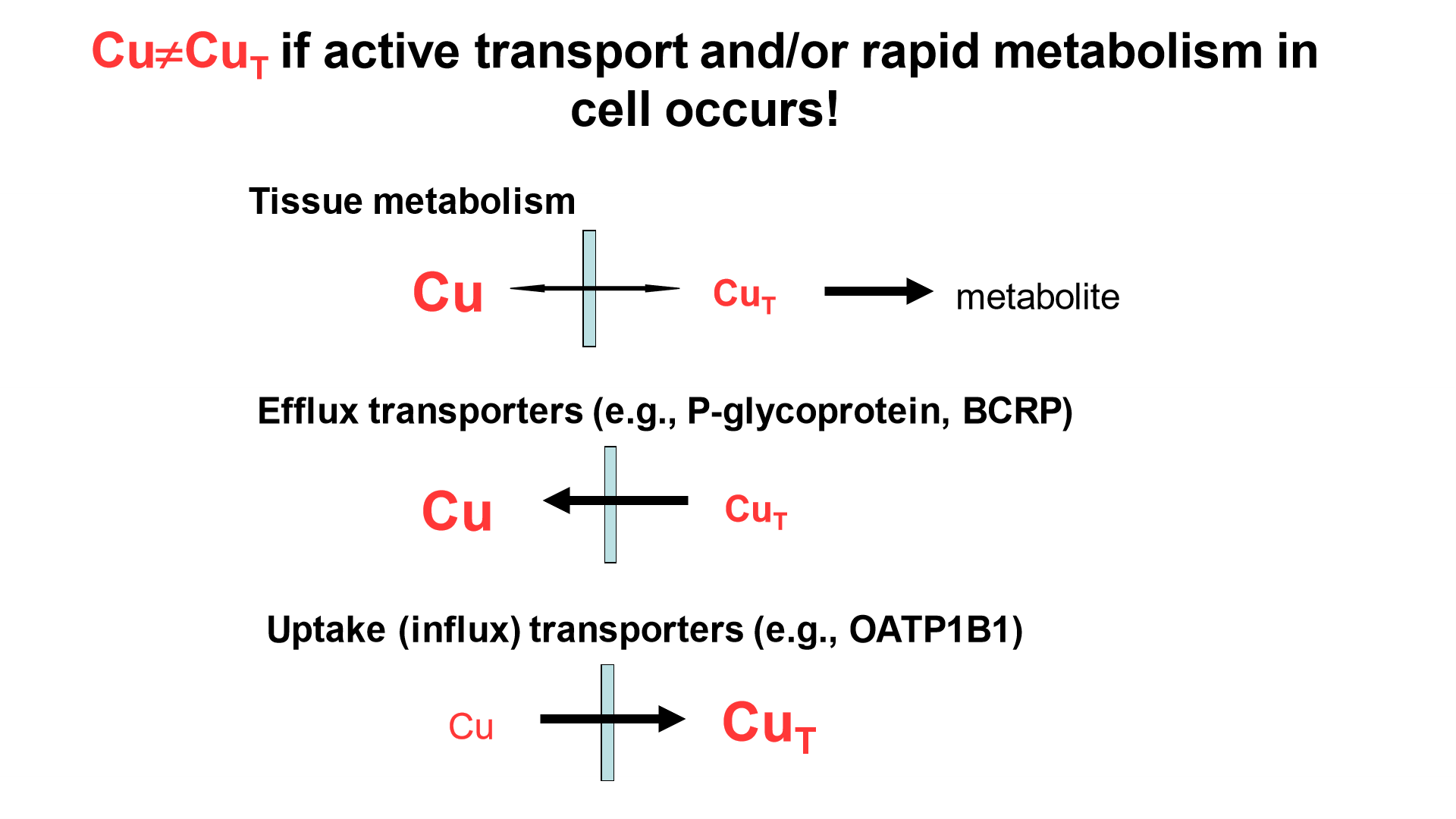
Role of transporters in ADME processes:
- For many of the new drugs, they are ionised and had poor permeability and rely on transporter proteins
- SLC - involved in uptake of drug from blood to particular tissue
- ABC - mediate efflux of drug from tissue back into systemic circulation
OATP:
- Located in membrane of hepatocytes
- Only anion drugs will be substrates for this transporter
- OATP1B1:
- Uptake of therapeutic drugs into hepatocytes
- OATP1B3:
- Active uptake of glutathione into hepatocytes
- Can be expressed in some cancer tissues
Clinical relevance of OATP1B1 transporter:
- Co-administration:
- Co administration with OATP1B1 inhibitors can lead to decrease in active uptake of drug into hepatocytes
- This leads to an increase in plasma concentration of drugs so there is an increase in adverse effects
- Variation in expression:
- SLOC1B1 c521T>C is the most relevant snp. This leads to decreased ability of OAT1P1B1 leading to elevated blood concentration resulting in increased myopathy
- 521 CC variation leads to decreased OATP1B1 activity, reduced uptake into the liver and increased myopathy when taking statin - so concentration of statin is higher
Effect of active transport and/ or rapid metabolism in cell