2.1–2.3: Properties of Atoms, Molecules, and Water
2.1 Properties of Atoms
Elements & Atoms
Elements are pure substances, each with one type of atom. They cannot be broken down chemically. ( C, H, O, N )
Atoms:( nguyên tử) basic units of matter, composed of protons, neutrons, and electrons.
Atomic Structure & Identity
Nucleus: dense center with protons
—have mass—(positive charge) and neutrons (neutral).Electrons: negatively charged, move in orbitals around the nucleus.
Atomic Number—số hiệu nguyên tử— (Z)=Number of protons, defines the element (e.g., hydrogen \text{Z}=1 , carbon \text{Z}=6 ).
In neutral atoms, protons = electrons.
Atomic Mass—khối lượng nguyên tử— determined by protons + neutrons
Isotopes & Ions
Isotopes—đồng vị—: atoms of the same element with same proton but different numbers of neutrons (e.g., ^{12} \text{C}, ^{13} \text{C}, ^{14} \text{C} ).
Ions: atoms that gain or lose electrons, becoming charged. Nhường hoặc cho electron
Cation: positively charged (loses electrons, e.g., \text{H}^+ ).
Anion: negatively charged (gains electrons).
Electron Arrangement & Octet Rule
Orbitals: regions where electrons are likely to be found (e.g., 1s, 2s, 2p).
Thứ tự sắp xếp các orbital theo năng lượng tăng dần: 1s, 2s, 2p, 3s, 3p, 4s, 3d, 4p, \dots
s - first shell, p- 2 shell → the different is the shape
2 electron per orbital, and 1 per orbital until every orbital has 1 electron
Electron Shells: energy levels for orbitals.
First shell (1s) holds up to 2 electrons.
Subsequent shells can hold up to 8 electrons (octet rule).
Valence Electrons: Hoá trị
electrons in the outermost shell, determine chemical behavior and bonding.
Atoms tend to achieve very stable outer shell with 8 electrons (octet)
only it participate in chemical bond ( liên kết hoá học )
Periodic Table: Organizes elements by recurring chemical properties and increasing atomic number.
2.2 Molecules and Chemical Bonds
Molecule—phân tử—Groups of two or more atoms held together by chemical bonds.
Chemical Bonds: Attractions that hold atoms together in molecules
Covalent Bonds—liên kết cộng hoá trị—
Formed by sharing valence electrons between atoms. ( chia sẻ electron với nhau đạt trạng thái ổn định)
Stability: Atoms share electrons to complete their outer shells (octet rule )
Polar vs. Nonpolar Covalent Bonds— Nhóm nhỏ trong Covalent Bonds
Electronegativity—độ âm điện—: atom's attraction for shared electrons.—khả năng hút electron—
CH → low electronegavity
ON- high electronegavity
If it in the same group then it non-polar , EX: C-C, C-H, H-H
If they go different then it polar
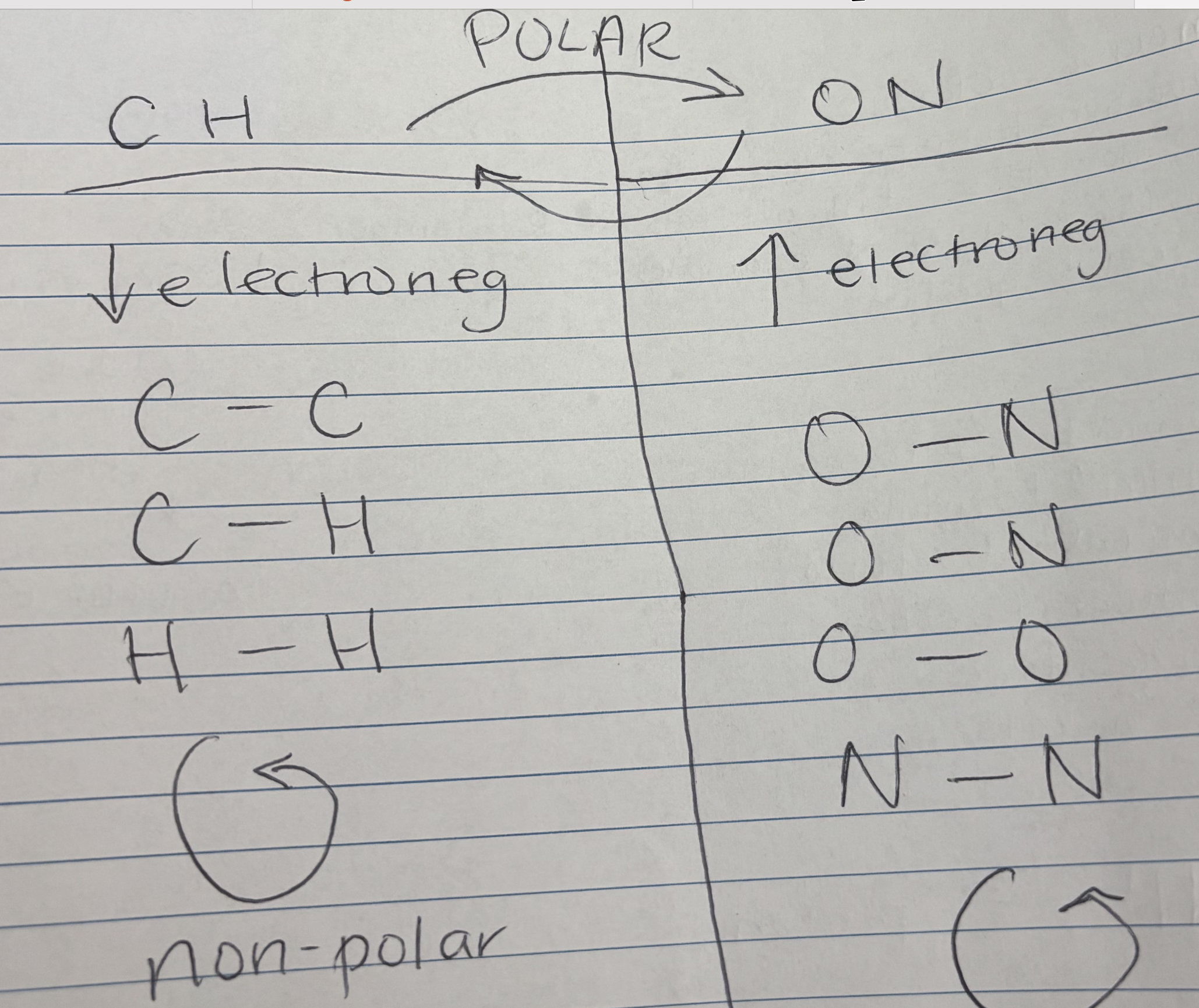
Polar Covalent—liên kết hoá trị có cực— Unequal sharing of electrons due to electronegativity differences—nguyên tử nào hút mạnh hơn thì mang điện tích âm , yếu thì dương
Nonpolar Covalent—liên kết hoá trị ko cực—: Equal or nearly equal sharing of electrons. Less soluble in water.
Cách phân biệt giữa polar và nonpolar : là dựa vào electronegativity
Nếu gần bằng nhau → nonpolar
Nếu có sự chênh lệch lớn → polar
Ionic Bonds— liên kết ion—sự nhường hoặc nhận electron để trái dấu và hút nhau—fully stole
Formed by the transfer of electrons, creating oppositely charged ions.
Example: Sodium Chloride (NaCl) - Na loses electron ( \text{Na}^+ ), Cl gains electron ( \text{Cl}^- ).
Dissolution in Water (Hydration): Water molecules surround and separate ions due to their polarity.
Chemical Reactions: Involve breaking and forming of chemical bonds; atoms are conserved but rearrange (Reactants \rightarrow Products).
2.3 Water
Polarity & Solubility—Tính phân cực và khả năng hoà tan —
Water is a polar molecule due to polar covalent bonds and bent shape.
Hydrophilic—yêu nước—: "Water-loving" substances; polar molecules that dissolve in water.
Hydrophobic—kị nước—: "Water-fearing" substances; nonpolar molecules that avoid water
If it has H-bonds → always polar
Hydrogen Bonds—liên kết hydro—very weak
Weak electrostatic attraction between a hydrogen and another electronegative atom (e.g., O or N). —Nguyên tử Hydrogen (H) phải đã gắn trực tiếp với một nguyên tử có độ âm điện cao → thường là O, N, hoặc F.— they not sharing
pH and Proton Concentration
pH: A measure of proton ( \text{H}^+ ) concentration in solution.
^+]
Scale: 0-14 ; Neutral: pH = 7
Acids—pH<7— Release \text{H}^+ , proton donor , hydrogen without electron then —> only proton left
Bases—pH>7—: Accept \text{H}^+ $$

Important from the slide that ask on test :
label C
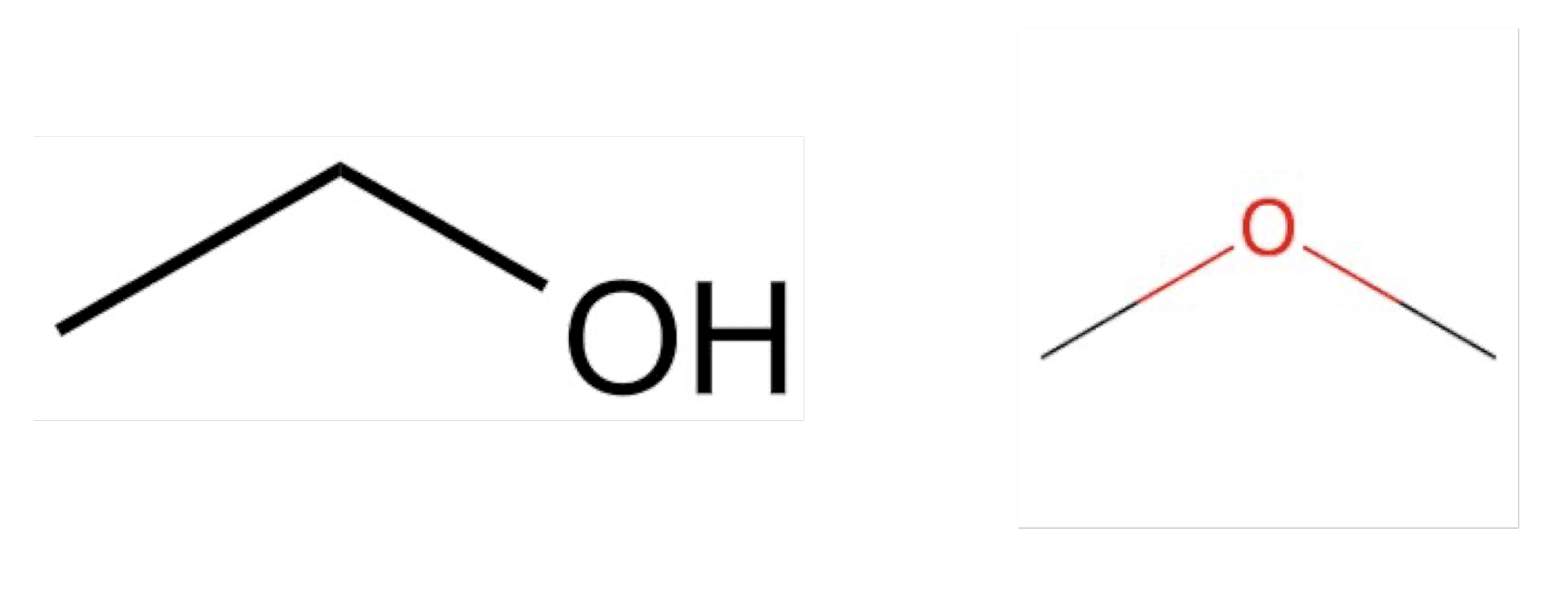
How to tell it polar and non-polar activity:
Solid line → covalent bonds
Dot line → H-bonds
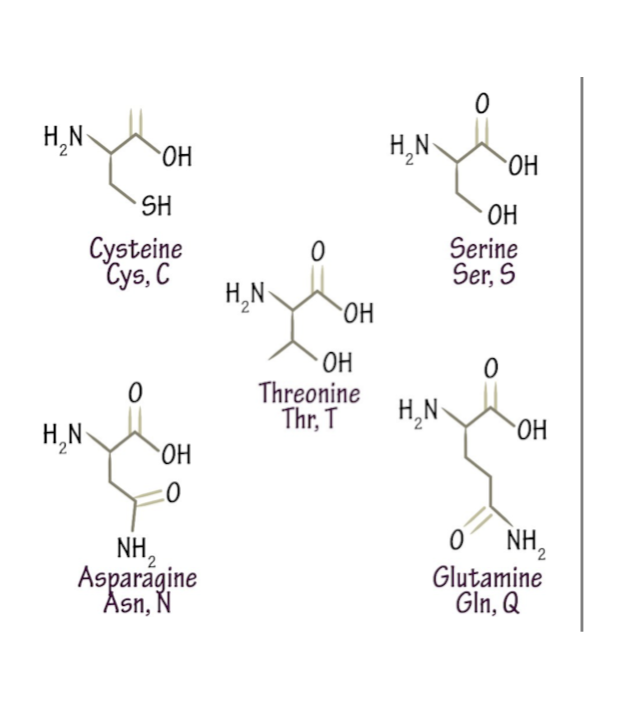
→ Polar , have H-bonds
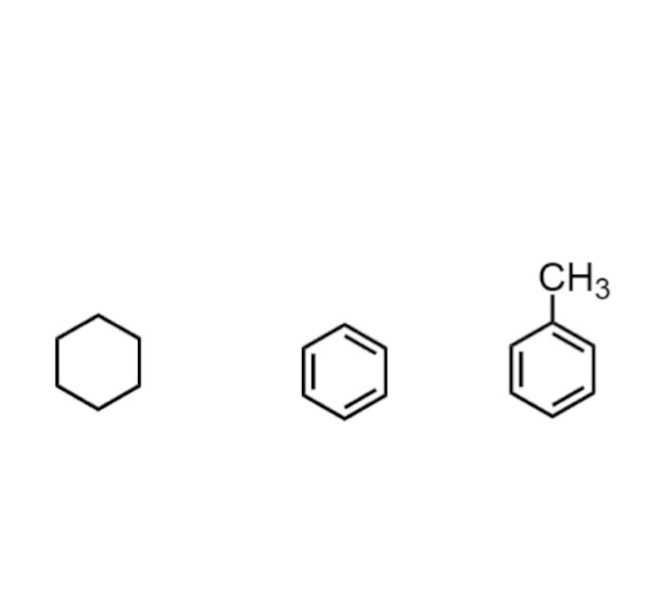
→ Covalent bonds