M6L1 Hypoxia targeted therapies

The definition of hypoxia varies for different tissues
Hypoxia causes - tumour growth/survival, angiogenesis, Warburg effect and low pH, metastasis, genetic changes, immune evasion, stem-like phenotypes
More hypoxia = worse response of tumours, regardless of the type of treatment
Chemotherapies are less effective
Tumour is more likely to have spread at time of surgery
x3 less DNA damage during radiotherapy
Resistance to immunotherapies
Strategies to target hypoxia:
Hyperbaric oxygen - increased radiotherapy toxiciuty, oxygen seizures, difficult to provide for all patients/cumbersome
Oxygen mimics
Electron affinic chemicals that mimic oxygen in fixing free-radical damage
Not metabolised by tumour cells through which they diffused
Can penetrate further than oxygen
Matronidazole - more active, toxic, benefit in some cancer sub groups
Etanidazole - equal activity to miso, less toxic, no clinical benefit
Nimorazole - less active, much less toxic, benefit in HNSCC (Denmark, no change found in combination with RT in UK studies)
CARGOGEN (95% O2, 5% CO2) - thought to influence chronic (diffusion limited) hypoxia only, limited benefit unless combined with radiosensitiser (nicotinamide)
Comparable toxicity
Positive improvements in regional contral
More effective in highly hypoxic tumours, confirmed by 26-gene hypoxia signature
Hypoxia activated prodrugs/cytotoxins
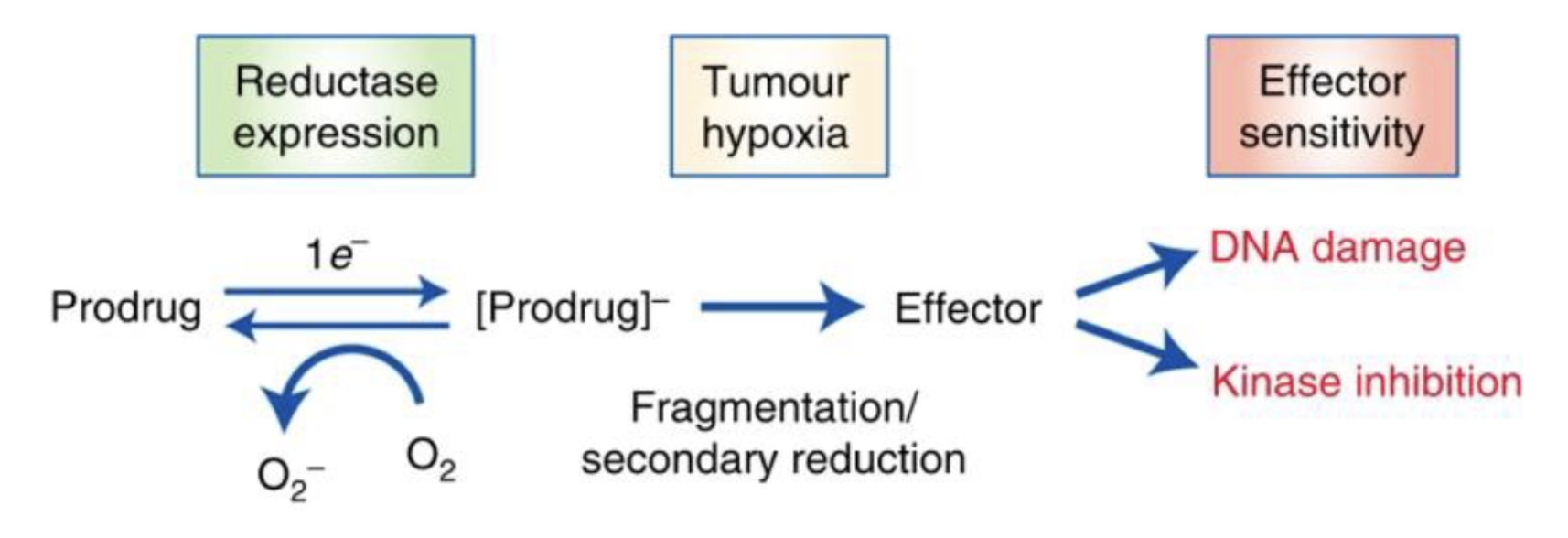
In hypoxic conditions, prodrug is reduced and fragmented to produce the active agent
Drug delivery issues
Some may require very high hypioxia
Relevant enzymes must be expressed, and there is high level of reductase in liver which may cause side effects if this is not the site of interest
Tirapazamine - no evidence of benefit in HNSCC, poor radiotherapy delivery, drug toxicity, no selection of patients based on tumour hypoxia in trials
Evofosamide - little/no advantage in pancreatic and soft tissue sarcoma, no selection of patients based on tumour hypoxia in trials
Molecularly targeted hypoxia activated prodrugs - inhibits a molecular target only under hypoxia (conventional HAPs release a cytotoxic agent/chemotherapy)
Proteolysis targeting chimeras (PROTACs)/hypoxia activated PROTAC (HAP-TAC) - allows degradation of protein of interest in hypoxic regions specifically
Modifying existing therapies (eg oxaliplatin) for use in hypoxia
Modulating oxygen consumption
Atovaquone - antimalarial drug which inhibits complex III in ETC
Papaverine - phosphodiesterase 10A inhibitor, inhibits complex I of ETC
Targeting hypoxia induced biological response, eg DDR
None of these approaches have made it to clinic
In clinical trials, there is rarely stratification for hypoxic tumours
We need to pick the right patients - needle electrodes, hypoxia signatures, tissue based biomarkers, serological markers, imaging
However this is difficult to do as there is different levels of hypoxia at different parts of the tumour
Need to come up with a method to quickly and cheaply provide a picture of the full tumour landscape of hypoxia, not just the few parts being sampled
Preclinical testing has not been good enough, for example only choosing highly hypoxic cell lines and not others which is not representative of patients in clinic