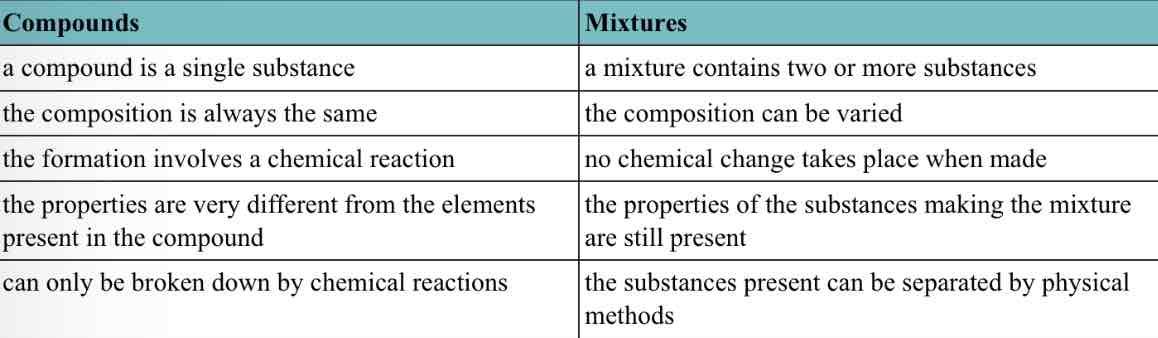
C2: Elements, compounds, mixtures
Elements, compounds and mixtures
Element: substance made up of just one type of atom
Compounds: different types of atoms are chemically bonded
Mixtures: compounds, elements, molecules can mix without chemically bonding
Atomic structure and the periodic table
The atom has a central nucleus containing neutrons and protons, surrounded by electrons in shells
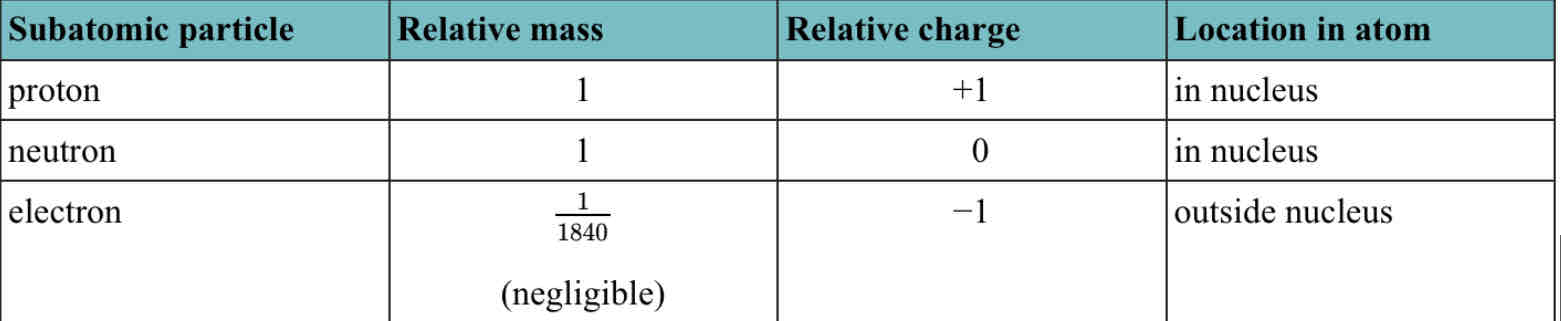
Atomic number = number of protons in the nucleus
Nucleon number = protons + neutrons in the nucleus
Electronic configuration
group number = outer shell electrons
period number = number of shells
Noble gas (VIII) electronic configuration
Unreactive gases
Full outer shell (8 electrons)
Do not make chemical bonds with atoms of other elements
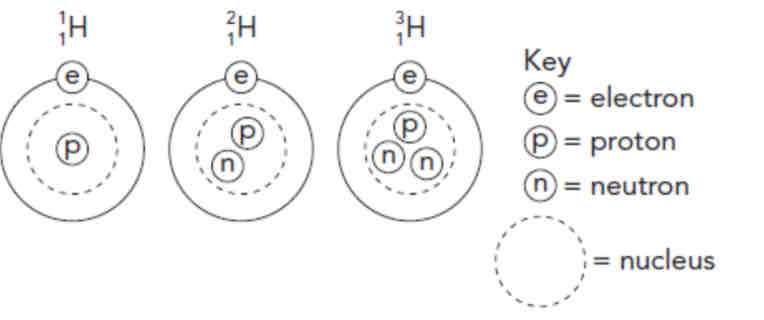
Isotopes
Pure elements with atoms of different masses
Same number of protons & electrons, different number of neutrons.
Defined by their difference in mass number. Eg. carbon-12, carbon-13
Characteristics
Same elements → same properties (same number of electrons and electronic configuration)
Chemical bonding
positive ions: cations
negative ions: anions
Ionic bonds: strong electrostatic attraction between oppositely charged ions
involves the outer electron shells
Molecular compounds: atoms bonded by shared electrons. Eg. water, methane,…
Ionic compounds: ions (charged atoms) are held together in a regular structure. Eg. Sodium chloride
Ions and ionic bonds (metal)
Ionic compounds
Have a formation of positive and negative ions
Metal atoms lose their electrons to become positive ions
Non-metals gains electrons to become negative ions
Ions formed achieve stable electron arrangement of the nearest noble gas to them
Alkali metals + halogens → greater number of electrons that get transferred
Physical properties
Solid at room temperature
High m.p and b.p
Often soluble in water
Conduct electricity in water when liquid/dissolved
Reason:
Regular lattice structure (each ion is surrounded by opposite charge ions)
Ions attracted by strong electrostatic forces → large amounts of energy is needed to break them
Water is attracted to charged ions
Liquid/solution electrons are attracted to free ions moving about
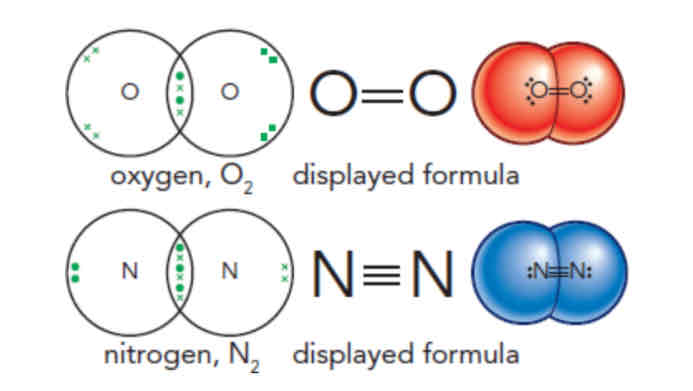
Simple molecules and covalent bonds (non-metal)
A pair/multiple pairs of electrons are shared between atoms
Simple molecular elements (diatomic molecules)
Held together by the forces of attraction between shared negative electrons and the nuclei involved
Main features:
formed by sharing a pair of electrons → noble gas configuration
each atom contributes an equal number of electrons to each bond
Group 7 halogens exist as diatomic particles (covalent molecules)
Hydrogen and halogens are held together by a single covalent bond.
Uses 2 electrons, one from each atom
Physical properties
Liquids/gas at room temp → made of simple molecules
low m.p and b.p → intermolecular forces are very weak
Do not conduct electricity→ no ions to carry a charge
Giant covalent structures
Macromolecules: giant molecular crystals held together by strong covalent bonds
Structures
Diamond: 1 carbon atom is attached to 4 others
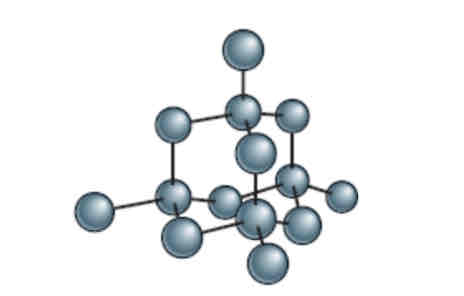
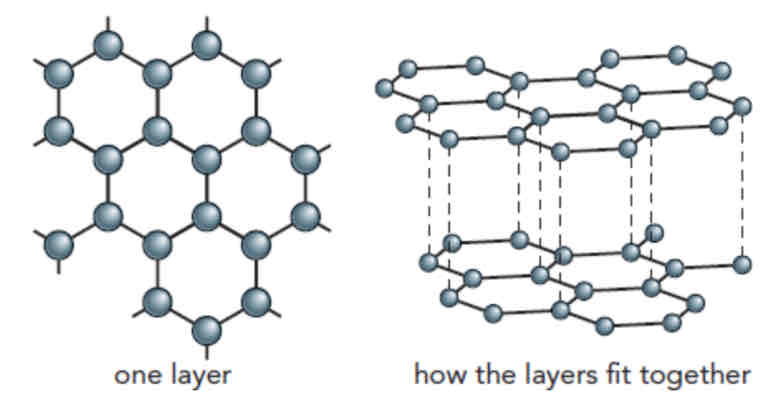
Graphite:
Does not conduct electricity
Arranged in flat layers of linked hexagons
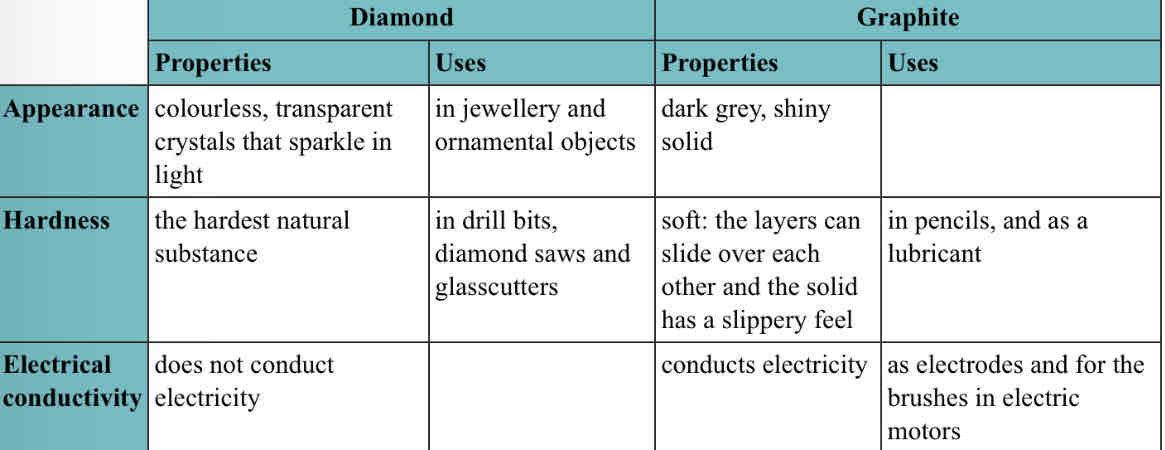
Uses of diamond and graphite
Metallic bonding
positive ions in a giant metallic lattice surrounded by a sea of delocalized electrons
Physical properties of metals:
Good conductors of electricity → delocalized electrons can carry a charge
Malleable → positive ions are arranged in layers that slide over each other