stem cells and gene therapy
Human Body Composition:
Approximately 50-100 trillion cells, varying in size, shape, and function
More than 200 different types of cells exist in the body
Definition of Stem Cells:
Unique cells with capabilities for self-renewal and differentiation into specialized cells
hCharacteristics of Stem Cells
Definition:
Stem cells can 'self-renew' and give rise to specialized cells within the body and can give rise to all or a subset of specialised cells of the body.
Types of Stem Cells:
Embryonic Stem Cells (ESCs): Derived from the early stages of embryo development.
Adult Stem Cells: Found in adult organs or peripheral tissues.
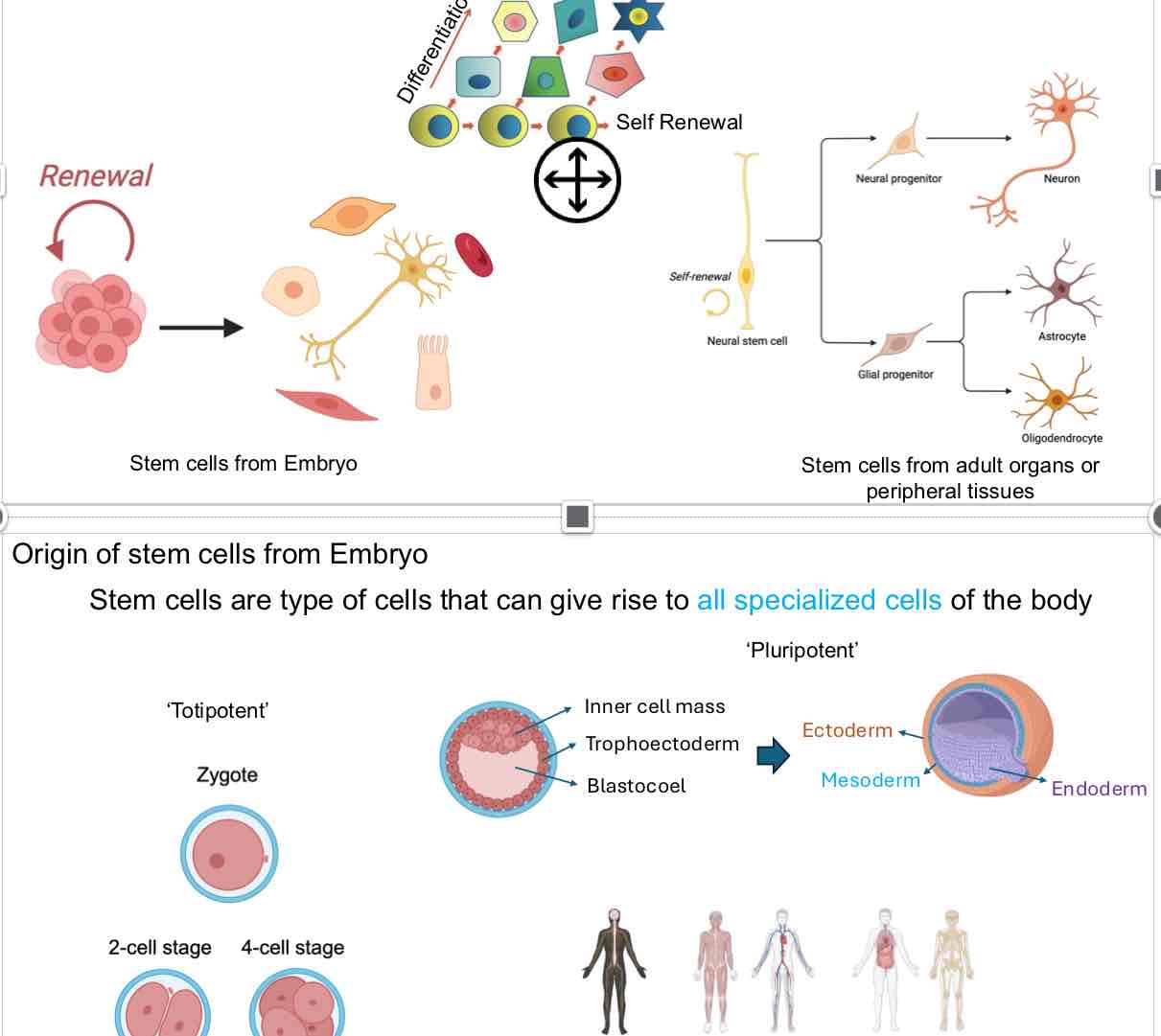
Origin of Stem Cells
Embryonic Stem Cells: Can become all types of specialized cells (Totipotent).
Sources:
Inner Cell Mass of the blastocyst:
Develops into Ectoderm, Mesoderm, and Endoderm layers.
Definition of Terms:
Totipotent: Ability to form all cell types.
Pluripotent: Ability to form nearly all cell types.
Page 6: Pluripotent Stem Cells
Characteristics:
Embryonic Stem Cells (ESCs)
Capable of self-renewal and differentiation into various specialized cells such as:
Germ cells, Muscle cells, Neuronal cells, Blood cells, etc.
Differentiation Factors & Renewal Factors: Key to their functionality.
Page 7: Therapeutic Applications of ESCs
Diseases Treated Using ESCs:
Spinal Cord Injuries: Transplantation of ESCs to injury site.
Eye Diseases: ESCs directed to form cone and retinal ganglion cells.
Cardiovascular Diseases: ESCs differentiated into cardiomyocytes.
Liver Injuries: ESCs differentiated to hepatocytes.
Diabetes: ESCs to pancreatic islet cells.
Osteoarthritis: Chondrocyte organoids used.
Page 8: Ethical Considerations of ESC Research
Key Debates:
Ethical implications of when life begins.
UK Regulations:
Use requires authority from the Human Fertilisation and Embryology Authority (HFEA).
Research allowed only under strict ethical guidelines; typically, using surplus embryos from IVF treatments.
Licensed research can only take place on embryos up to 14 days. Stem cells are isolated from the blastocyst (at 5 to 6 days).
Page 9: Induced Pluripotent Stem Cells (iPSCs)
Development of iPSCs:
Adult fibroblast cells are reprogrammed with transcription factors (e.g. Oct4, Sox2).
Significant potential for disease modeling and development studies.
•Epigenetic landscape of the chromatin is remodelled
•iPSCs can be used to reprogram to different lineages.
Cell from patients are used the issues of immune reaction can be avoided.
Page 10: Limitations of Stem Cell Therapies
Challenges include:
Tumor formation potential.
Poor differentiation or survival rates within transplanted sites.
Page 11: Multipotent Stem Cells
Definition and Sources:
Mesenchymal Stem Cells (MSCs): Found in bone marrow, umbilical cord blood, adipose tissue.
Can differentiate into multiple cell types across more than one germ layer.
Page 12: Functions of Multipotent Stem Cells
MSCs contribute to:
Tissue repair and regeneration.
Homing to injury sites following systemic injection.
Differentiation into various cell types, reducing inflammation, and promoting efficient wound healing.
Page 13: Hematopoietic Stem Cells (HSCs)
Role and Types:
HSCs commit to various blood cell lineages, such as lymphoid and myeloid.
They can be classified as oligo-potent or unipotent based on their differentiation potential.
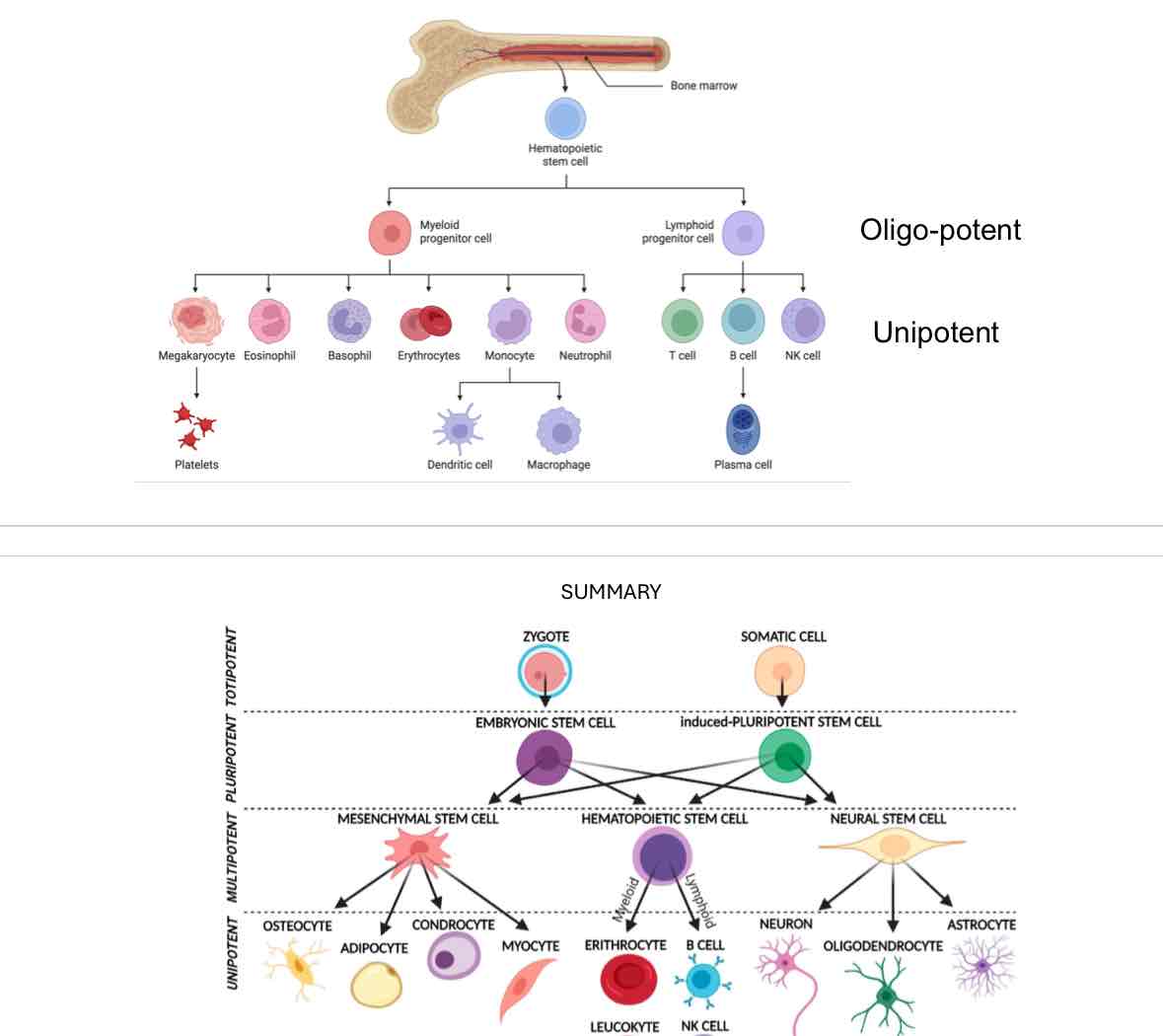
Summary of Stem Cells
Types of Stem Cells:
Zygote, Somatic cell, Embryonic stem cell, Induced pluripotent stem cell, Mesenchymal stem cell, Hematopoietic stem cell, Neural stem cell, various differentiated cell types.
Page 17: Gene Therapy and Disease
Disease Classifications:
Monogenic: Caused by mutations in a single gene.
Chromosomal: Involves deletions or duplications of chromosome structures.
Multifactorial: Result from interactions of defective genes with environmental factors (e.g., Cancer, Diabetes).
Page 18: Gene Therapy Approaches
Key Strategies:
Gene Supplementation: Replacing non-functional gene with a functional copy.
Gene Silencing: Using siRNAs to inhibit gene expression by blocking mRNA.
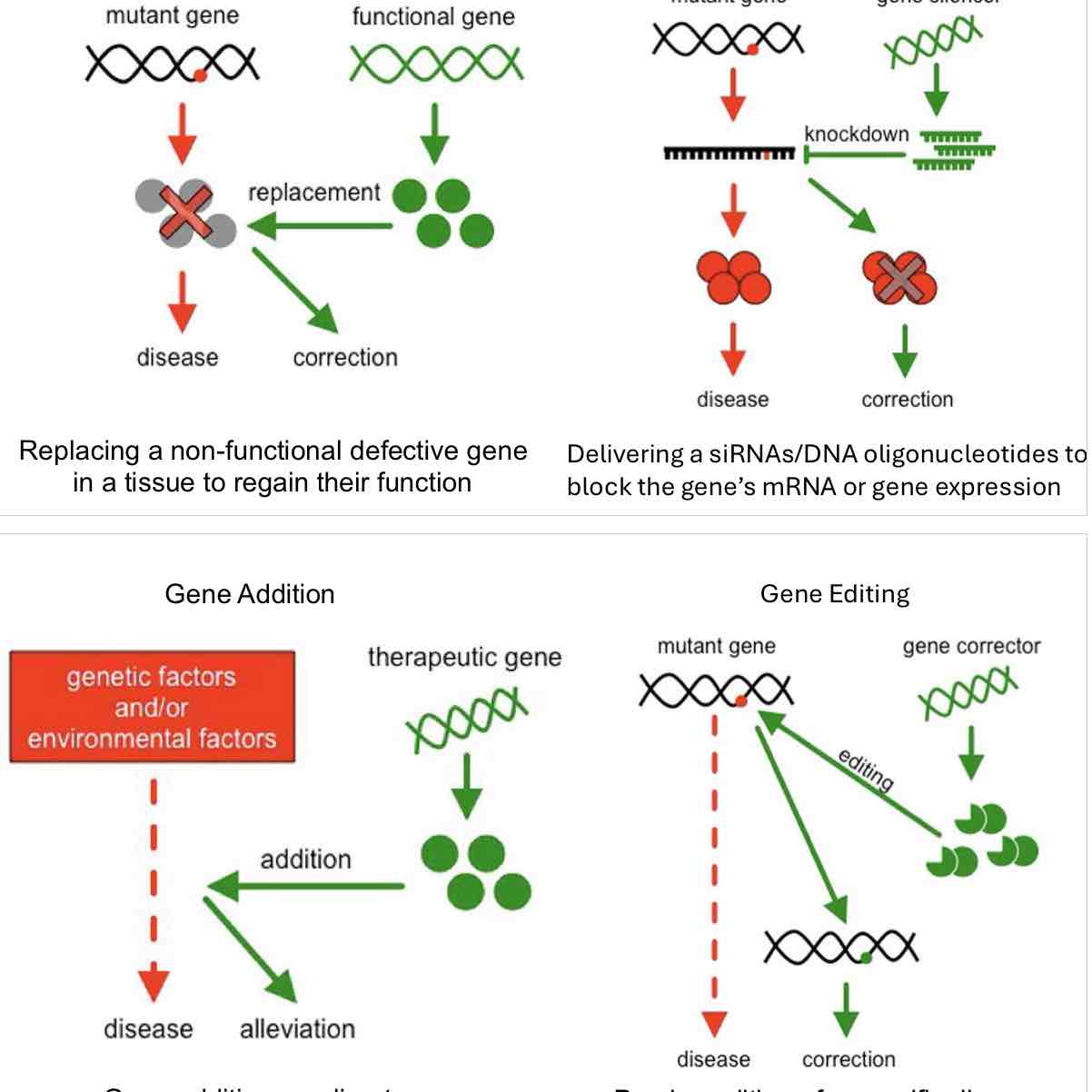
Page 19: Advanced Gene Therapy Techniques
Gene Addition: Adding functional genes to ameliorate complex diseases.
Gene Editing: Precise alterations in specific disease-causing mutations.
Page 20: Types of Gene Therapy
Classifications:
Based on cell target (Somatic vs. Germ cells).
Based on vector type and mode of delivery.
Page 21: Practical Considerations in Gene Therapy
Key Requirements:
Effective target tissue identification.
•The therapeutic gene should express in ample amount and to sustain the expression for long period of time.
Avoidance of immune reactions and disruption of normal cellular functions.
Page 22: Steps in Viral Vector Gene Therapy
Process Overview:
Identification of affected genes and generation of therapeutic genes
Targeting and delivering vector into affected tissues
Integration of viral DNA into the host chromosome or maintaining as an episome
Continuous monitoring of therapy progress
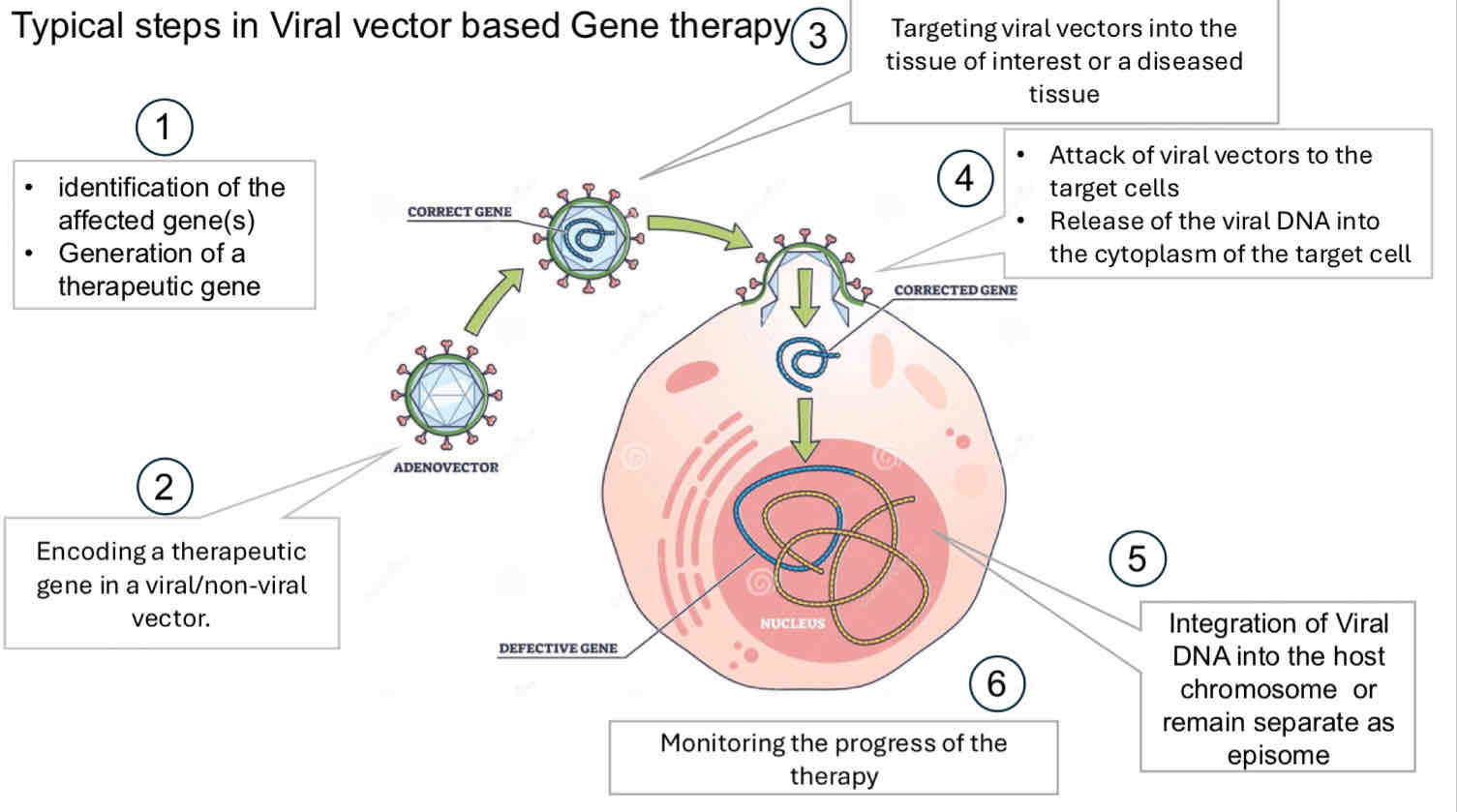
Page 23: Viral Vectors in Gene Therapy
Functions:
Capsid properties determine tissue specificity.
Major viral vectors include Adenovirus, AAV, Lentivirus.
Page 24: Gene Delivery Methods
Viral Vectors:
Capable of efficient gene delivery and optimized for specific targeting.
Non-Viral Gene Delivery:
Includes natural or synthetic polymers, less efficient but lower immunogenicity.
Page 25: Success Stories in Gene Therapy
Notable Treatments:
Gendicine: First approved gene therapy (2003) for head and neck squamous cell carcinoma affecting TP53.
Luxturna: Corrects vision loss due to RPE65 mutations with an AAV vector, costing $425,000 per eye.
Page 26: Risks Associated with Gene Therapy
Potential Challenges:
Immune responses to viral vectors, targeting issues, potential infections, and tumorigenesis.