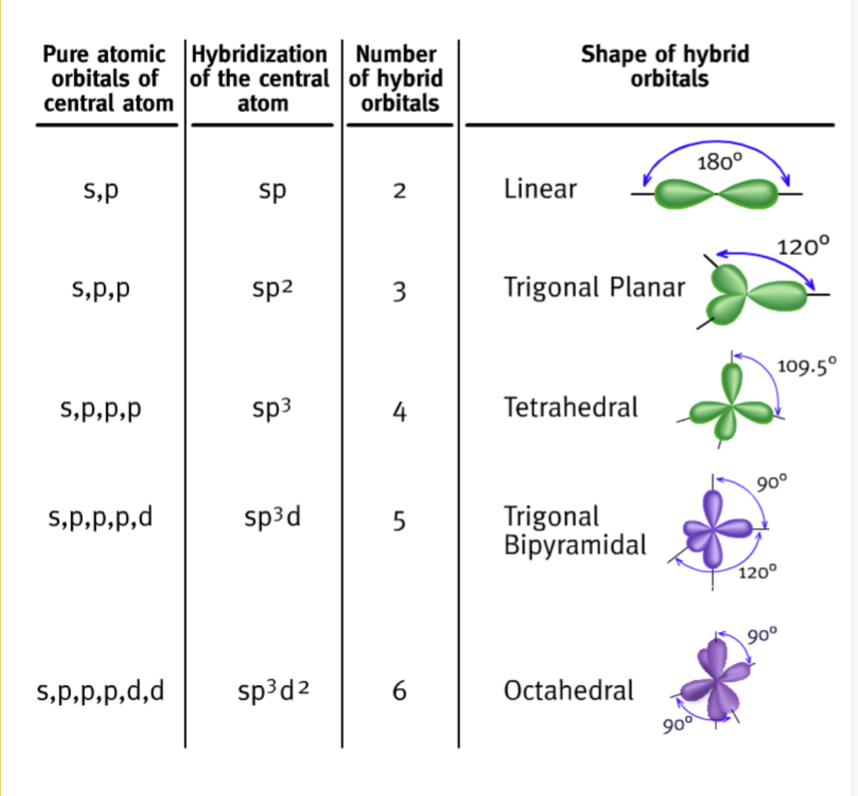
4.6 Hybrid Orbitals
- Orbitals overlap, may form hybrid orbitals of equal length.
- Form to satisfy VSEPR geometry.
- Hybrid orbitals form by combining orbitals with different shapes in a process called hybridization.
Types of Hybrid Orbitals
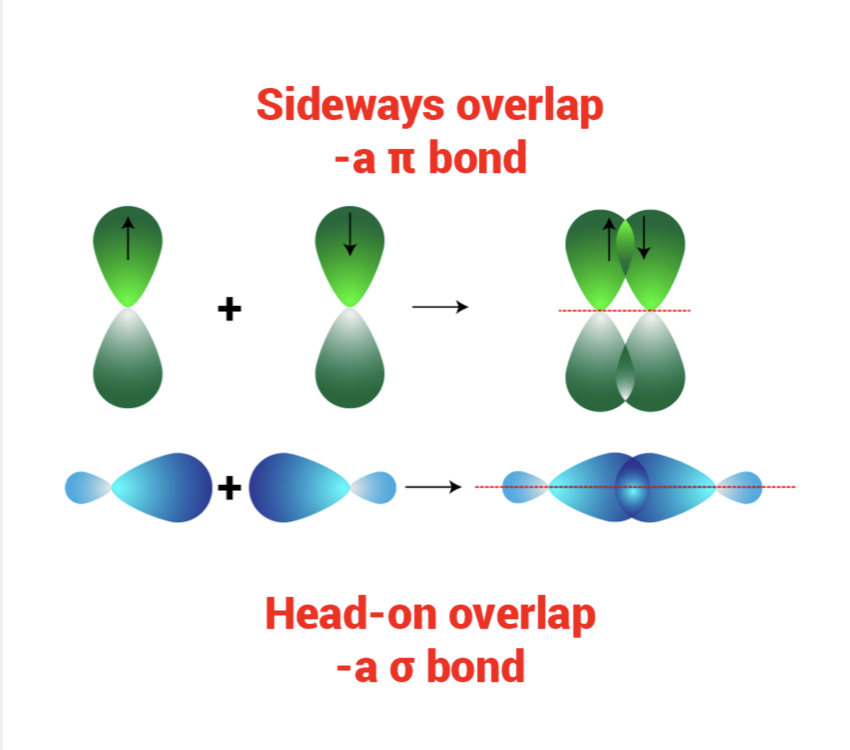
Sigma vs. Pi Bonds
Sigma bonds
- Bonds that are formed when 2 orbitals overlap end to end
- (s, p, sp hybrid)
Pi Bonds
- Bonds that form when two parallel orbitals overlap
- Occur during double/triple bonds.
Example Test Questions
- For the molecule NH3:
- Draw the Lewis Structure, identify sigma and pi bonds (consider formal charge)
- Draw the VSEPR diagram
- List the AXE #, steric #, basic geometry, molecular geometry
- Determine the hybrid orbitals, identify sigma and pi bonds
- Must match basic geometry (tetrahedral is sp3, trigonal planar is sp2, linear is sp, etc)
- Bonds require unpaired electron
- Pi bonds must be p orbitals (not hybridized)
- Draw orbital diagram
Steps:
Write electron configuration
Draw diagram showing valence electrons
Determine is any promotion is required, if so, draw another diagram
- keep in mind sigma and pi bonds
Hybridize by moving electrons, draw a full orbital diagram showcasing the bonding and hybridization