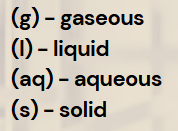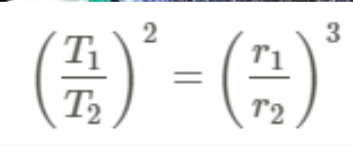PHYSICAL SCIENCE
Module 6: Chemical Reactions
Laws of Conservation of Energy
Atoms can neither be created nor destroyed in a given chemical reaction
The number of atoms that are in the reactants has to balance the number of atoms in the products.
Proust’s Law of Definite Proportions
The elements are in a fixed ratio
Elements in a compound are always in the same mass ratio—if the ratio changes, it is an entirely different compound.
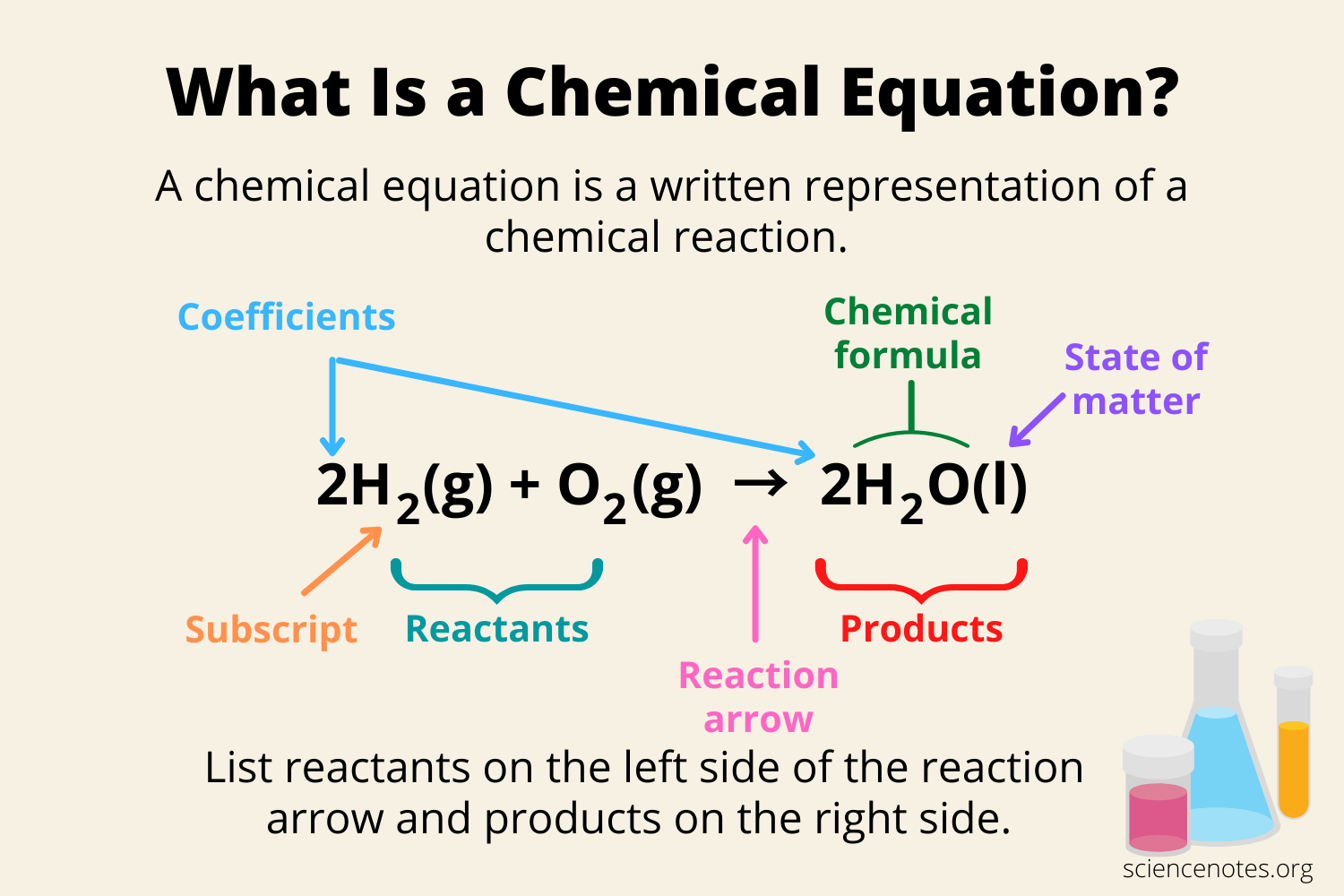
Chemical reaction
Chemicals interact to form new chemicals with different compositions
Reactants - Starting materials (either elements or compounds)
Products - Result of the chemical reaction and have different chemical properties from the reactants
Chemical change - breaking old and making new chemical bonds to rearrange atoms from the reactants
Types of Chemical Reactions
Synthesis Reaction (Combination reaction)
Two or more simple substances combine to form a more complex product
A + B → AB
Decomposition Reaction
Opposite of synthesis reaction
The breakdown of a single complex compound to simpler compounds/elements
AB → A + B
Single Replacement Reaction
One element replaces another element in a compound
A + BC → AC + B
Double Replacement Reaction
Compounds undergo exchange of their elements with each other
AB + CD → AD + CB
Combustion Reaction
A rapid chemical reaction between a fuel and an oxidant (usually O) that releases heat and light
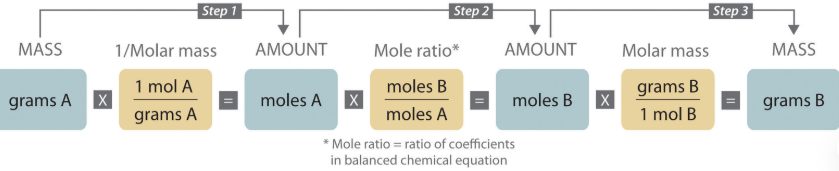
Stoichiometry
Stoikhein (element) and metron (measure) = measure of elements
Given gram x 1 mol/molar mass (gram) of element a = moles a x moles B/moles A (mole ratio) = moles b x molar mass (grams) of element b/1 mol = grams of element b
Mole
The amount of substance containing the same number of discrete entities like atom or molecules
Number of molecules can be calculated with moles using Avogadro’s number (6.022 × 1023)
Represents the number of molecules in one gram of a compound (or element)
Mole ratio
Ratio between any two substances in a balanced reaction
Molar mass
Mass of one mole of a substance
g/mol or kg/mol
Gases in Stoichiometry
1 mole of gas = 22.4 L
Limiting reactant
Consumed first in a chemical reaction among other reactants
Limits the amount of products that can be formed
Excess reactant
Not fully or entirely consumed/reacted in a chemical reaction
Does not limit the amount of products that can be formed
Module 7: Models of the Universe and the Early Astronomy
Orlando Ferguson’s Map of the Square and Stationary Earth
Pythagoras - First to originate a spherical earth by north-south constellations, sinking ships and lunar eclipse
Plato - Earth forms a globe and is equidistant from the center: it is the center of the universe, perfect and eternal, has four elements (WAFE)
Aristotle - Idea of geocentric universe with fixed spherical earth as center
Rounded shadow of the earth in the moon
Change in constellations
Indirect idea on gravity
Eratosthenes - gave the most accurate size (non-scientific calculation)
Figures for guidance:
Geocentric theorists
Plato
Aristotle
Ptolemy
Tycho Brahe
Heliocentric theorist
Aristarchus of Samos
Nicolaus Copernicus
Galileo Galilei
Johannes Kepler
Ancient Astronomy
Plato - Universe is perfect and unchanging. Questionned the circular paths of planets
Eudoxus of Cnidus - There are 27 interconnected geocentric spheres
Aristotle - Added 3 spheres to jupiter and mars and 4 sphere to sun, moon, venus and mercury (3 JM, 4 SMOMEV)
Claudius Ptolemy - Earth is spherical, stars are fixed, planets move independently and planet can reverse their motion.
Aristarchus of Samos - Sun and stars are fixed and estimated the size of sun and moon through earth’s size and estimated the distance of earth to the sun and moon
Modern astronomy
Observations became more quantitative
Theories based on logical and mathematical concepts
Nicolaus copernicus - Proposed the heliocentric theory, tilting of the earth, and the four seasons
Earth as only one of the planets
Earth rotates in axis daily and around the sun once a year
Annual tilting of axis
Retrogade motion explained by earth’s motion
Distance of earth to sun is smaller than earth to stars
Tycho Brahe
Modified the geocentric model
Made instruments for precise measurements for positions of planets
Discovered supernova in constellation cassiopeia
Tychonic system
Galileo Galilei
Used the telescope to observe
Discovered sunspots and rough surface of the moon
Discovered the 4 famous moons of jupiter
Venus has phases like moon
Johannes Kepler
First law- Orbits as ellipses and sun at focus
Second law - Imaginary line between sun to a planet where it is in equal areas in equal time. Periphelion near to sun and Aphelion far to sun
Third Law - Cube of radius of the planet’s orbit is proportional to the square of its period of revolution
Module 8: Kinematics and Theories about Motion
Aristotle’s Theory of Motion
Object are made of 4 primordial elements (WAFE)
2 types of motion: Natural and Violent
Natural motion - motion naturally without forcing motion to happen
1st theory - Object made of earth falls toward the center of the Earth to seek it natural place
2nd theory - Heavy object fall faster than lighter ones
3rd theory - objects fall faster in air than water
4th theory - objects sometimes move away from natural places
Violent motion - a motion requires a force to make an object move in unnatural manner
Longevity of aristotle’s theory of motion: it was consistent in common sense, no other theories were made that time, quantitative method was not yet developed
Galileo Galilei’s Finding about Motion
First finding - 2 objects (light and heavy) fell and struck the earth at the same time **Acceleration due to gravity
Second finding - A marble on a flat surface will infinitely roll **Law of inertia
Third finding - When a marble is rolled in an inclined plane at a fixed angle, the distance covered and the square of time was constant **velocity (d/t)
Fourth finding - On a free falling object, gravity causes object to be uniformly accelerated **UAM Uniformly Accelerated Motion
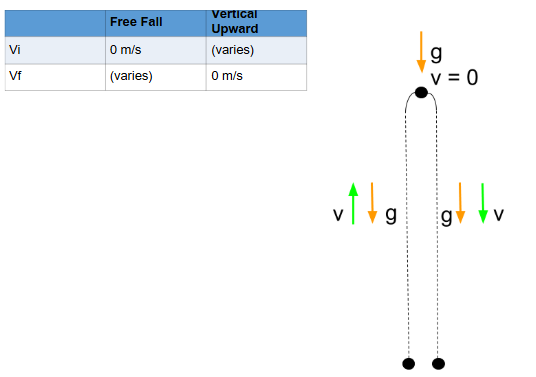
Free fall motion
Type of motion under the sole influence of gravity
Vi is 0
Vertical Upward Motion
Type of motion that follows a vertical upward path resisting the effect of acceleration due to gravity
Vf is 0
Projectile Motion
Type of motion along two dimensions acted upon by gravity. Combines the constant velocity (x) and constant acceleration (y)
Object is called projectile and the path is the trajectory
Motion of object vertically upward into the air moving dependently on gravity and horizontally independently of gravity. Vertical = dependent on gravity, Horizontally = independent on gravity
MODULE 9: Newton’s Laws of Motions
The earth is attracted to apple and apple is attracted to earth → laws of universal gravitation and laws of motion
Gravitational theory - observing an apple fall to the ground
First Law: Law of Inertia
An object at rest remains at rest; an object in motion remains in motion → it changes when it is acted on by an unbalanced force
Force - a push or pull motion that changes an object’s state of motion
Balanced Force -