Organic Chemistry
CONTENT 10.1 Fundamentals of Organic Chemistry 10.1.1 The big picture 10.1.2 Homologous series 10.1.3 Molecular and structural formulae 10.1.4 Structural isomerism 10.1.5 Naming organic compounds 10.1.6 Trends in physical properties 10.1.7 Classification of organic compounds 10.1.8 Aromatic hydrocarbons 10.2 Functional Group Chemistry 10.2.1 The big picture 10.2.1 Alkanes 10.2.1 Alkenes 10.2.1 Alcohols 10.2.1 Halogenoalkanes 10.2.1 Benzene |
|---|
10.1 Fundamentals of Organic Chemistry
10.1.2 Homologous Series
Carbon has the ability to form covalent bonds with itself, producing long chains and rings; this is known as catenation.
Homologous series: is a series of closely-related compounds in organic chemistry.
Alkanes are hydrocarbons — compounds composed of carbon and hydrogen atoms only.
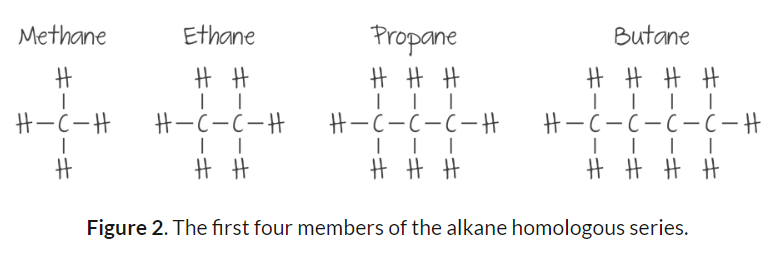
Members of the same homologous series also have the same general formula and functional group.
For the alkanes, the general formula is CnH2n+2
This allows us to determine the molecular formula of any alkane given the number of carbon atoms.
The functional group of the alkanes is the alkyl group, which contains carbon-carbon single bonds.
Exam Tip
Make sure that you are able to explain the trend in the boiling points of the members of a homologous series. The boiling points increase with the number of carbon atoms in the molecule due to the increase in molar mass and stronger intermolecular forces.
Intermolecular Forces: is the force that mediates interaction between molecules.
In summary, a homologous series is a group of organic compounds that:
contain the same functional group,
have the same general formula,
differ by a CH2 group
have similar chemical properties
show a gradation in physical properties such as boiling points
10.1.3 Molecular and Structural Formulae
Organic compounds can be represented by three main different types of formula: empirical formulae, molecular formulae, and structural formulae.
Structural formulae can be divided into full structural formulae, condensed structural formulae, and skeletal formulae.
Empirical and Molecular Formulae
For some alkanes, methane, and propane, for example the empirical formula is the same as the molecular formula.
Though for compounds, this is not that case.
ex. the e.f. of ethane (C2H6) is CH3.
The molecular formula represents the actual number of each type of atom in a compound.
Structural Formulae
In structural formulae, all atoms and the bonds between the atoms are shown.
In full structural formulae, a single line represents a single covalent bond, a double line a double covalent bond and a triple line a triple covalent bond.
Sometimes it is preferable to use condensed structural formulae, in which the bonds between atoms are omitted.
In skeletal formulae, all atoms are omitted leaving only the backbone of the molecule.
In this type of formula, carbon atoms are assumed to be at the intersection of each line and at the end of each line.
In addition, hydrogen atoms bonded to carbon atoms are not shown.
If hydrogen is part of a functional group (such as hydroxyl group, OH) it is shown.
Represented Formulae in 3-D
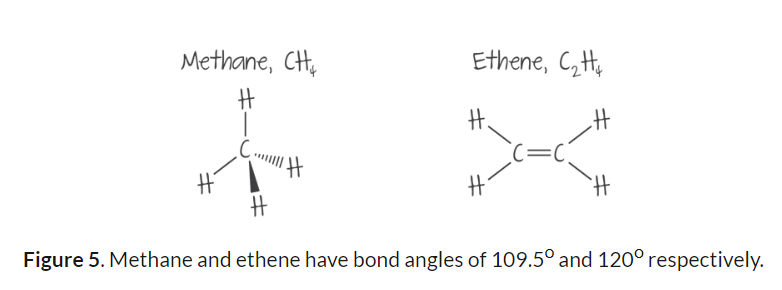
When representing organic molecules in two-dimensions, we use structural formulae with 90 degree and 180 bond angles.
Though this does not represent the actual molecular geometry of the molecule.
When carbon forms for single bonds, the molecular geometry is tetrahedral with the bond at 109.5 degree to each other.
When it for forms a double bond, as in ethene, the arrangement is trigonal planar with a bond angle of 120 degree.
For certain molecules, it is useful to show the relative three-dimensional positions of atoms or groups around a selected carbon atom, known as the stereochemical formula.
In a stereochemical formula, a bond sticking out from the plane of the page is shown as a solid wedge, whereas a bond directed behind the page is shown as a broken line.
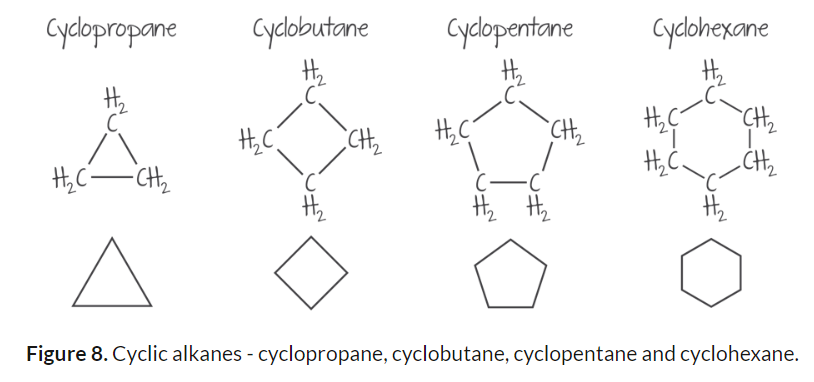
Branched and Cyclic Structures
Carbon atoms can also be joined together in rings, forming cyclic alkanes.
The structures of these cyclic hydrocarbon molecules are often represented by polygons.
The corners of the polygon represent a carbon atom together with the hydrogen atoms that are bonded to the carbon.
10.1.4 Structural Isomerism
Structural Isomers: they are compounds with the same molecular formula but different arrangements of atoms.
—