M8L3 Data management for medical research
Data management objectives
Ensure integrity/confidentiality of research data
Facilitate data accessibility —> analysis + sharing
Challenges
Compliance with legal/ethical standards
Maintaining data security and patient privacy
Handling large volumes of data
Data management is central to the designing, conducting, and reporting of trials and critical for validity/reliability of results
MHRA GXP Data Integrity Guidance Document - GXP= good X practice (distribution, clinical, lab, manufacturing, pharmacovigilance)
Source data - original records/documentation of data
Metadata is ‘data about data’ - structured information that describes, explains, locates, or manages other data eg documenting context like creation date, creator, format, and usage rights for efficient cataloging, discovery, and retrieval of resources
Derived data - result of the data analysis
Research data lifecycle - all phases in the life of the data from generation/recording to processing, use, retention, archive, retrieval, and destruction
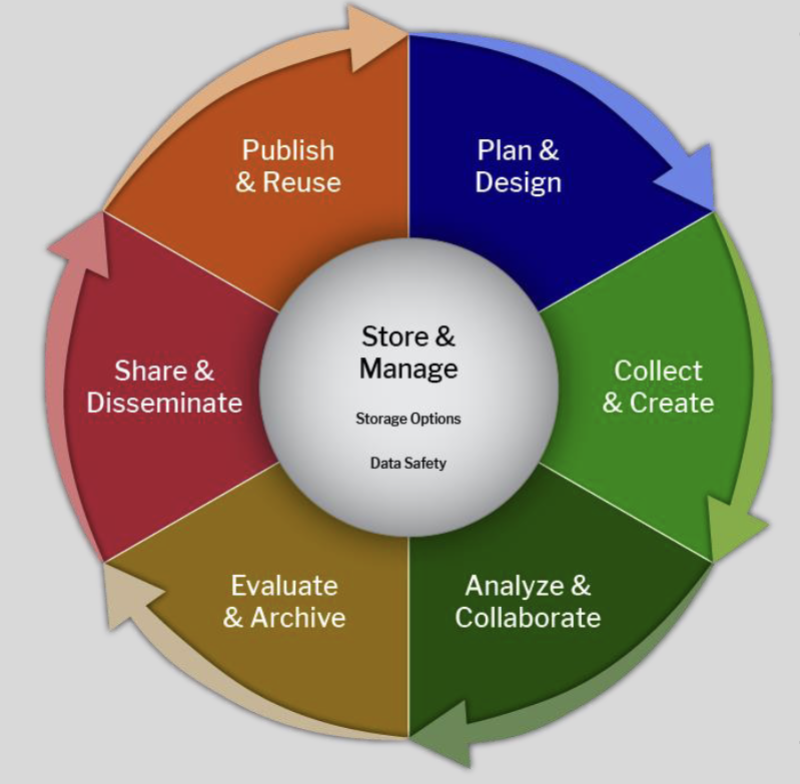
Data governance applies over the whole life cycle to provide assurance of data integrity
This is the arrangements to ensure data are recorded, processed, retained and used to ensure the record throughout the data lifecycle
Data integrity - degree to which data are complete, consistent, accurate, trustworthy, and reliable
Data should be ALCOA
A - attributable to the person generating it
L - legible and permanent
C - contemporaneous
O - original record or certified true copu
A - accurate
Data governance should ensure that data is complete, consistent, enduring, available
Legislative requirement - UK policy framework for health and social care research
Principle 14 - specific reference to transparent management of tissue/sample collection
Appendix 2: Laws - comprehensive list of regulations eg Medicines for Human Use (Clinical Trials) Regulations 2004, Human Tissue Act 2004, Data Protection Act 2018 (UK Implementation of GDPR)
Data management plan - structured tool outlining data handling during research which guides collection, processing, storage, and sharing of data