Chemistry Unit 3
Transverse Wave
- Amplitude- the height of the wave; measured from highest to the center point
- Wavelength- length of the wave; distance between two consecutive points on the wave
- Frequency- number of waves that pass a given point every second
- Speed- how fast the wave is moving
Electromagnetic radiation- a type of energy or wave that actually consists of two waves at 90-degree angles
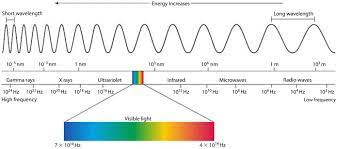
Electromagnetic Spectrum- the entire range of all the different types of electromagnetic waves arranged from lowest to highest energy
 Scientific Notation
Scientific Notation
- negative exponent= a small number
- positive exponent= large number
has to be one digit, between 1 and 9
Calculating frequency and wavelength
“c = 𝞴*𝞶”
c = speed of light = 3.0 x 108 m/s
𝞴 = wavelength (m or nm)
𝞶 = frequency (waves/sec or Hz)
1 nm = 10-9 m
Conversion Example:
570 nm x 10^-9__m__ = 5.7x10-^7 m \n 1 nm
Calculating the energy of one photon
E = h𝝼
The energy of a wave is directly proportional to the frequency (𝞶) of the wave.
h = Planck’s Constant = 6.626 x 10-34 J⋅s
𝞶 = frequency (waves/sec or Hz)
Atomic Emissions
- When electrons transition away from the nucleus, they absorb energy
- When electrons transition to the nucleus, they release energy
- more energy= farther away from the nucleus
- When electrons transition closer to the nucleus, they emit energy, producing colored light
- the unique patterns of emitted energy from atoms allow us to identify them
Electron Configurations
-- maximum number of electrons in a shell is given by the formula of 2n^2, where n represents the shell number
Orbitals- subdivisions of the main shells, a three-dimensional region that contains a maximum of two electrons; not real physical barriers, they are just mathematical predictions of where the electrons are likely 90% of the time
-- All orbitals can only hold 2 electrons
-- most orbitals exist in a variety of different shapes/orientations
Kinds of orbitals:
S- subshell has one “s” orbital, spherical, located on all four shells
P- subshell has three “p” orbitals, dumbell shaped, located on n=2, n=3 and n=4
D- subshell has five “d” orbitals, double dumbell, located on n=3, and n=4
F- subshell has seven “f” orbitals, located on n=4+
Periodic Trends
Periods- rows on the table
Groups- columns on the table
Atom Size-
- Atoms get smaller as you go up and to the right
- metal ions will be smaller than the atom as they lose electrons
nonmetals ions will be bigger than the atom as they gain electrons
Chemical Reactivity
- bigger atoms =more chemical reactivity
Ionization energry
ionization energy- the energy required to remove a valence electron
The smaller the atom, the more ionization energy required
Ionization energy increases when you move up and to the right
Electronegativity- the attraction an atom has for shared electrons in a bond
Increases as you move up and to the right
doesn’t include noble gases