BIOL 200 7.2 CDK-Cyclin Regulation
Cyclins & CDKs
-these work together to control the cell cycle
Frog Egg Experiment
-extracted cytosol from frog eggs that were at a known stage of division
injected it into a cell that was stalled at the end of interphase
compared with cells at the same stage that had been injected with cytosol from cells also in interphase
active agent that promotes next stage of cell cycle is in cytosol of cell
control of DNA synthesis and mitosis is positive → active agent turns on the process in the recipient cell (capable to respond but hasn’t initiated process itself)
cells can be advanced into next stage by appropriate factors
-after this discovery → replicated using cytosol from mitotic cells from different species and taking cytosol from one species and using it to induce mitosis in another species
-’factor’ in cytosol was universal and helped to promote the maturation of cell → maturation promoting factor (MPF)
-MPF = Activated Cyclin-CDK Complex
active agent is a protein (Cyclin-Dependant Kinase — CDK)
regulates activity of a large number of other proteins required for cell cycle progression by phosphorylating them
second protein (cyclin)
interacts with CDK
acts as a regulatory unit
CDK can only perform its function as a kinase when cyclin is bound
if cyclin removed from CDK → CDK inactivated
these 2 proteins combine to produce a single enzyme → cyclin-CDK complex
Cyclin-CDK Complex
-CDK concentrations in cell are more or less constant throughout the cell cycle
enzymatic activity becomes activated and deactivated throughout
-cyclin shows a cyclical pattern of increasing concentrations
coincides with increasing enzymatic activity of CDK
concentrations decrease to almost nothing
change in cyclin concentration coincides with end of CDK enzymatic activity
cycling of cyclin concentration and CDK activity that allow the cell to progress through checkpoints and the cell cycle
Four Major Classes of Cyclins

-increasing cyclin concentrations always precedes the passage through a checkpoint
cyclin-CDK activity must hit threshold before checkpoint is passed
M-cyclin: activity of CDK decreases rapidly not long after checkpoint has ben passed
S-cyclin: activity of CDK is activated at one checkpoint and stays high throughout rest of cycle
Phosphorylation Controls Cyclin-CDK Enzymatic Activity
-while the binding of cyclin to CDK is necessary for CDK activity, not sufficient on its own
-cyclin-CDK complex must be phosphorylated by other kinases
-activity of CDKs are tightly controlled by cell
cell moving into next stage of cell cycle before it is ready can be disastrous
-cyclin-CDKs are at the heart of a complex signalling pathway
involving hundreds of enzymes fighting each other to activate or deactivate cyclin-CDK complex
-balance between activators and deactivators occurs when conditions are just right
correct phosphates are in place on the cyclin-CDK complex
kinase is able to function
ex
addition of phosphate group to M-cyclin-CDK by kinase Wee1 results in inhibition of complex
Wee1 thought to respond to intracellular cues to ensure cell doesn’t progress through G2/M checkpoint unless they are big enough to divide
once cell has reached appropriate size → Wee1 protein is degraded (can’t phosphorylate M-CDK) → another protein Cdc25 removes phosphates from M-cyclin-CDK complex → activating complex
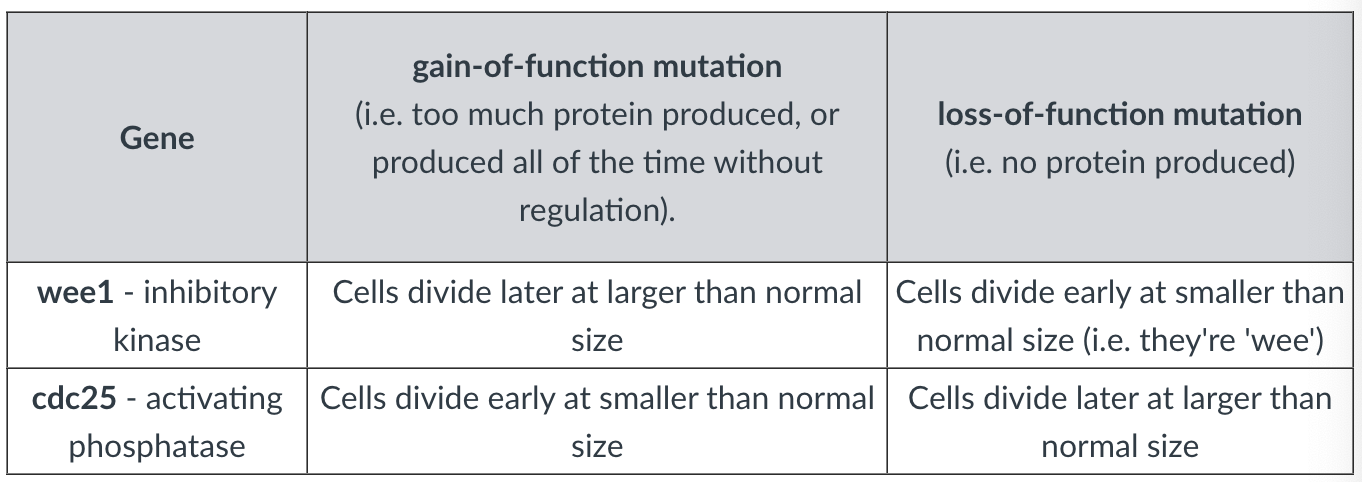
-in yeast → Wee1 and Cdc25 are key regulators of M-CDK
antagonistic relation discovered using genetic experiments where the dosage of genes was experimentally manipulated
increasing gene dosage (gof) → increases enzyme concentration
decreasing gene dosage (lof) → decreases enzyme concentration
-relationship between amount of protein and difference in when they go through mitosis is evidence of the complexity of the control that cyclin-CDK complexes are under
Specific Cyclins & their Role in the Cell Cycle
M-CDK Controls the G2/M Checkpoint and Re-entry into G1
-transition from G2 phase into M phase is complex
-requires complete rearrangement of cytoplasm
shutting down transcription & translation
preparing all organelles for separation → shut down
building a second MTOC for mitotic spindle
completely rearranging cytoskeleton
-M-CDK-cyclin enzymatically active at the end of G2 phase → primary control for this translation
-M-cyclin concentrations peak in metaphase → crashes and deactivates M-CDK
Kinase
-manages transition by phosphorylating several other proteins
-phosphate group changes conformation of protein → affect its function
-some targets of activated M-cyclin-CDK complex
histone H1
changes in chromatin configuration and in conjunction with other proteins leads to chromatin condensation
condensins
class of DNA binding proteins that bind to chromatin and help with chromosome condensation
nuclear lamins
phosphorylated lamins have a lower affinity for each other → nuclear lamins fall apart
disassembly of nuclear lamina results in break up of nuclear envelope into ER
structural proteins of the nucleolus
dispersion of nucleolar proteins
disintegration of nucleolus
vital for condensation of chromosomes
variety of protein kinases that regulate the cytoskeleton
arrangement of cytoskeleton as the cell disassembles the interphase microtubule network and forms the mitotic spindle
cdc25
M-CDK activating phosphate
creates a positive-feedback loop → further activation of M-CDK-cyclin → M-CDK activity rises increasingly rapidly as more M-CDK becomes active
anaphase promoting complex (APC)
activated
at the beginning of anaphase → protein complex degrades cohesin proteins that bind sister chromatids together → releases 2 separate chromosomes
APC also activates enzymes that tag M-cyclin for degradation
ensures that M-CDK will be properly deactivated at the end of its usefulness
cell can’t complete mitosis and return to cytoplasm to its interphase state unless MCDK is inactive
Control of G1/S Checkpoint and S-Phase Progression
-requires 2 cyclin classes
-transition from G1 to S-phase controlled by G1/S cyclin-CDK complexes
checkpoint passed → G1/S cyclin degraded and S-cyclin takes over
in some cases → 2 cyclins can bind to same CDKs → re-activated when new cyclin binds (and enzyme is properly phosphorylated)
-in order for cell to pass into S-phase (and replicate DNA) → must pass through G1/S checkpoint
there must be no DNA damage
protein that checks for damage is a transcription regulator → p53
protein that is constantly translated and degraded as long as it remains inactive
when DNA damage detected → p53 becomes phosphorylated → stops degradation → binds to promoter sequence for a CDK inhibitor (p21) → activating transcription & subsequent translation
p21 blocks the activity of cyclin-CDK complex
-once checkpoint has passed → G1/S cyclin helps to initiate S-phase via phosphorylation of replication machinery to help it assembly
-G1/S cyclin degraded and S-cyclin takes over
-S-cyclin-CDK complex activates DNA helicase and promotes assembly of the rest of the replication machinery required
-as it is activating replication → prevents replication from being able to happen more than once a cell cycle
does this by phosphorylating key enzymes in replication (cdc6)
phosphorylation of cdc6 result in it being tagged for degradation
without cdc6 → replication can’t be initiated
-before replication, the chromosome consists of a single (double-stranded) DNA molecule (chromatid)
-after replication, the chromosome consists of 2 DNA molecules (sister chromatids)
connected at centromere
complete with full complement of histones and any other proteins required for chromosome packing
-2 sister chromatids are held together along their length via a protein (cohesin)
must be degraded just prior to chromatid separation during anaphase
-S-CDK remains active right until start of mitosis despite S-phase being long over before that
proves that different cyclins are influenced by each other
Deactivation of Cyclin-CDK Complex
-once cyclin-CDK complex has completed its job → deactivated
via shutting down transcription of cyclin genes and degrading the cyclin proteins that already exist
-negative feedback loops are built in so that shutting down CDKs is possible
-one of phosphorylation targets of M-CDK is anaphase promoting complex (APC)
tags proteins for ubiquitination → marks them for degradation by proteasome
concentration of cyclin then drops rapidly
cyclin degraded → CDK deactivated → next step of cell cycle can begin
APC is deactivated in G1
one of the phosphorylation targets of activated G1/S-cyclin CDK complex