Carboxylic Acid Derivatives 3/21
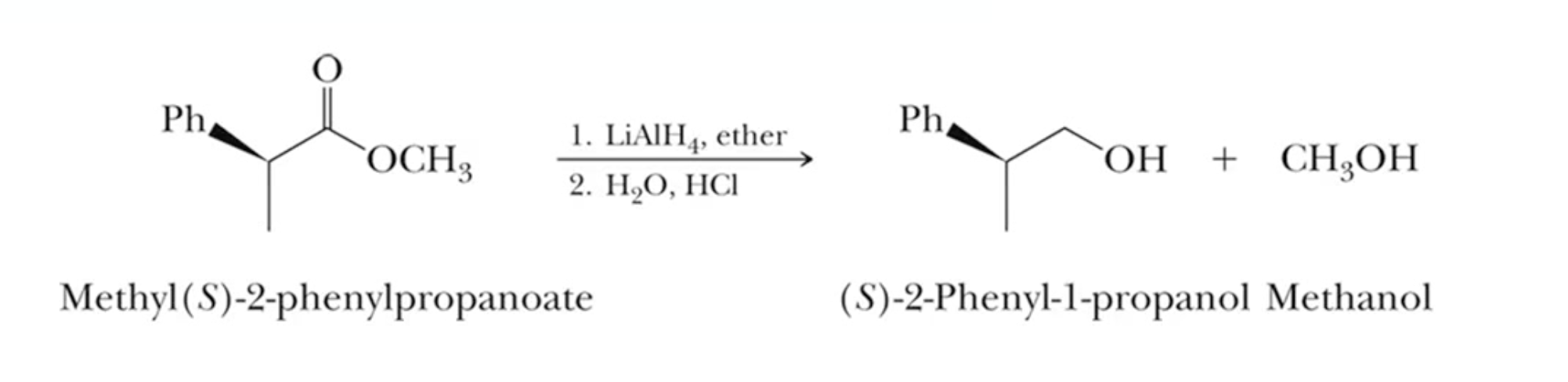
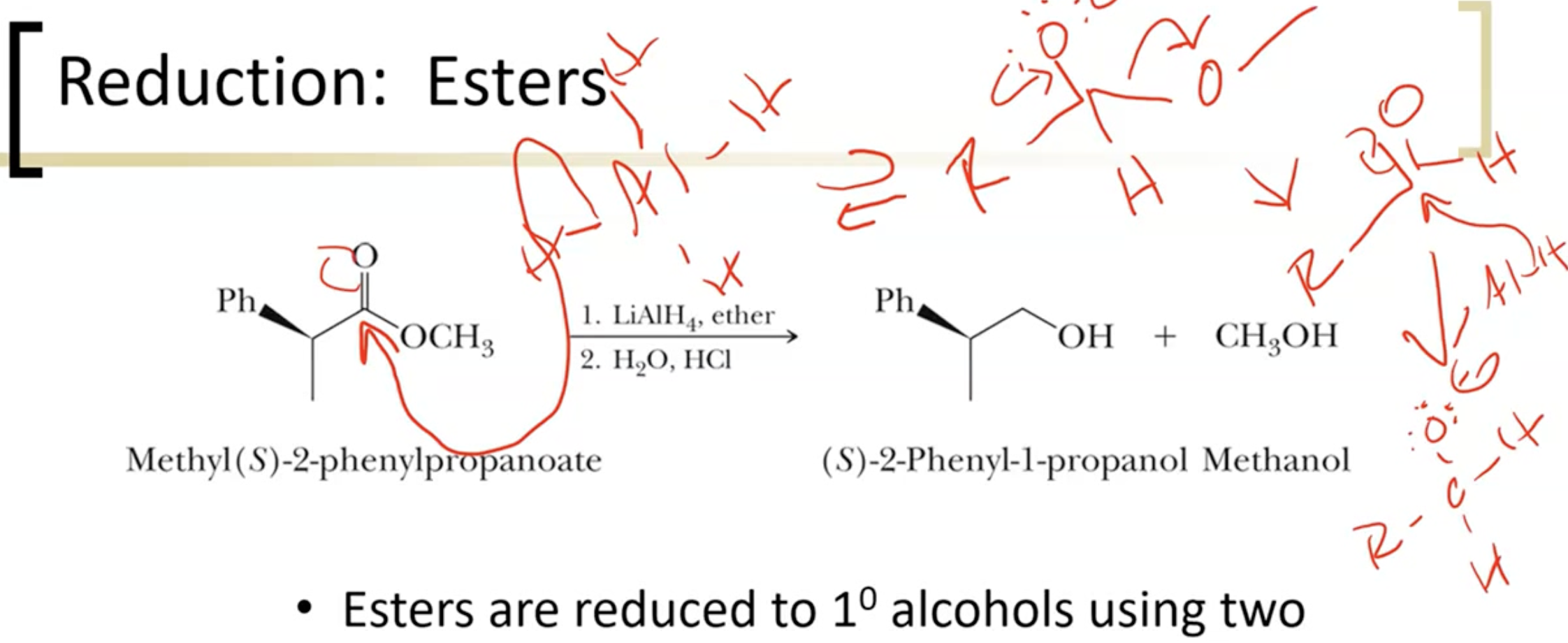
Reducing Esters
esters reduced to primary alcohols using two equivalents of LiAlH4, NaBH4 is not strong enough
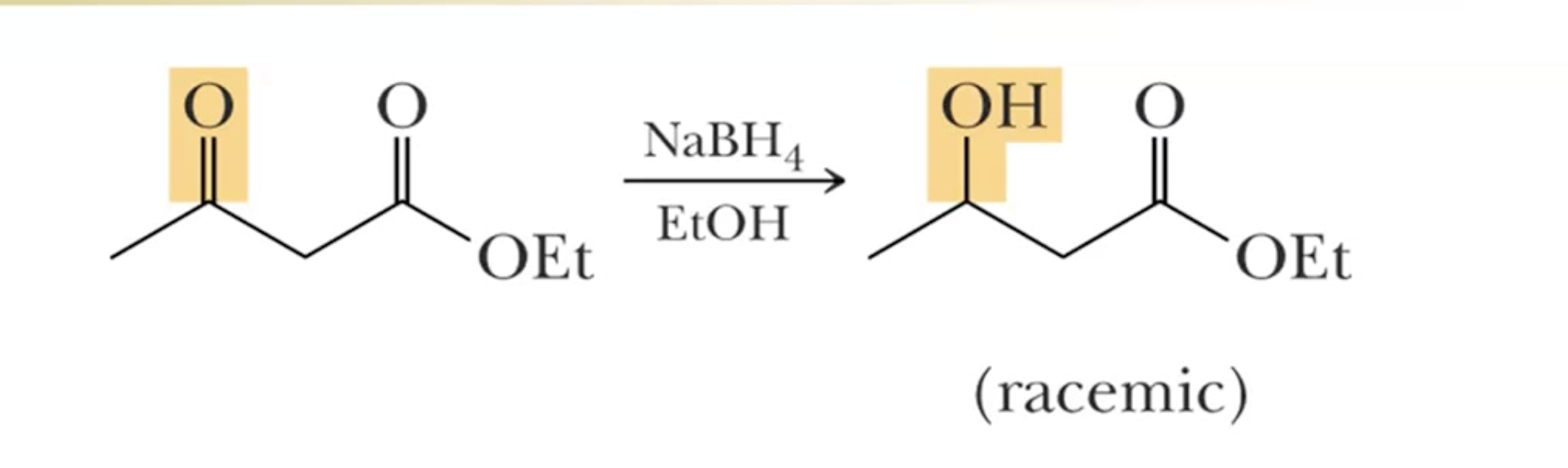
NaBH4 is used to reduce an aldehyde or ketone to an alcohol in presence of an ester
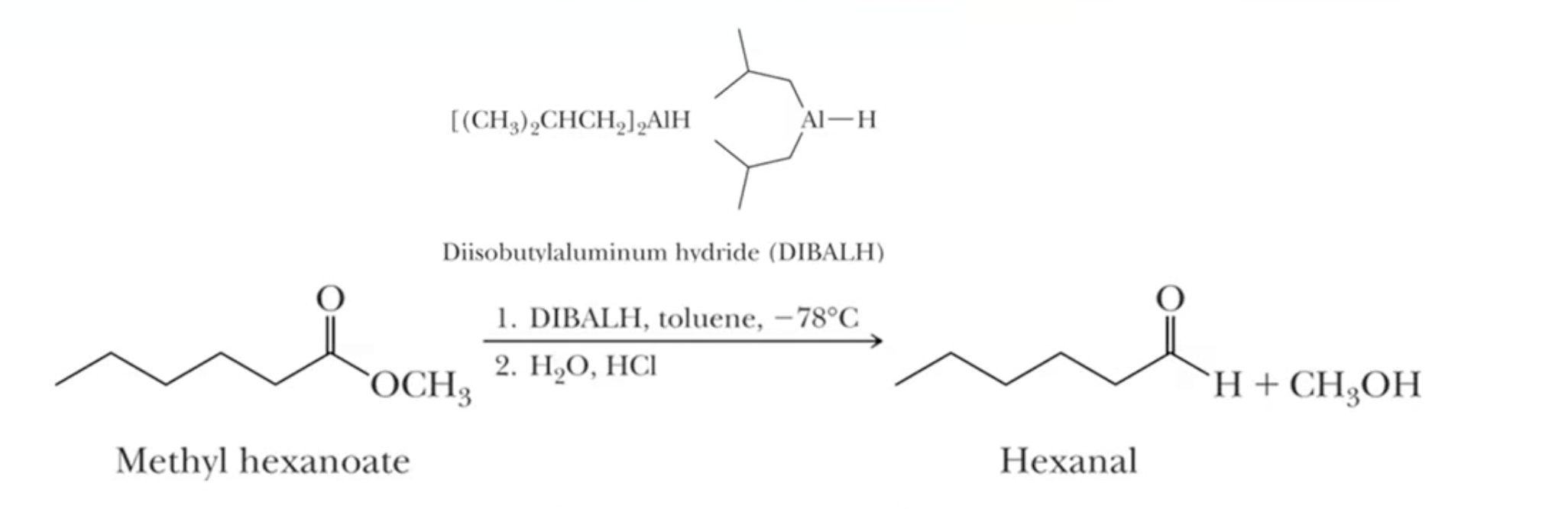
Reducing esters to aldehydes= use DIBALH, temperature-dependent
If tried to do it at room temperature, the ester converts to the alcohol
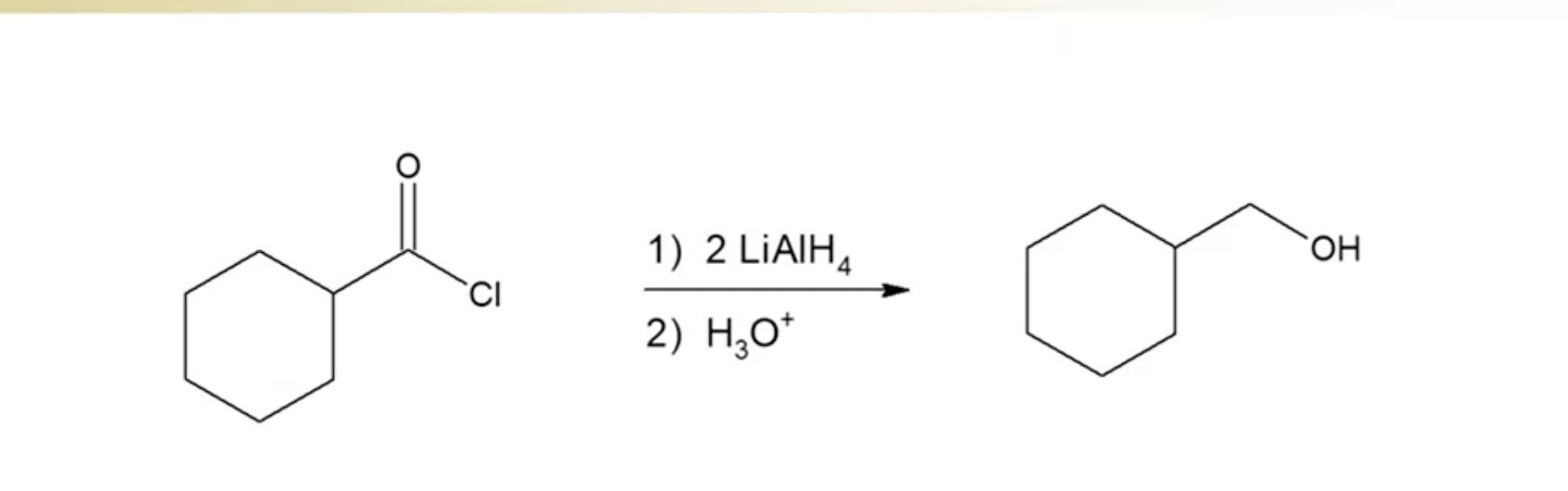
Reducing Acid Halides
Reduced to primary alcohols using lithium aluminum hydride and an acid wash
first hydride forms aldehyde, second hydride forms the alcohol
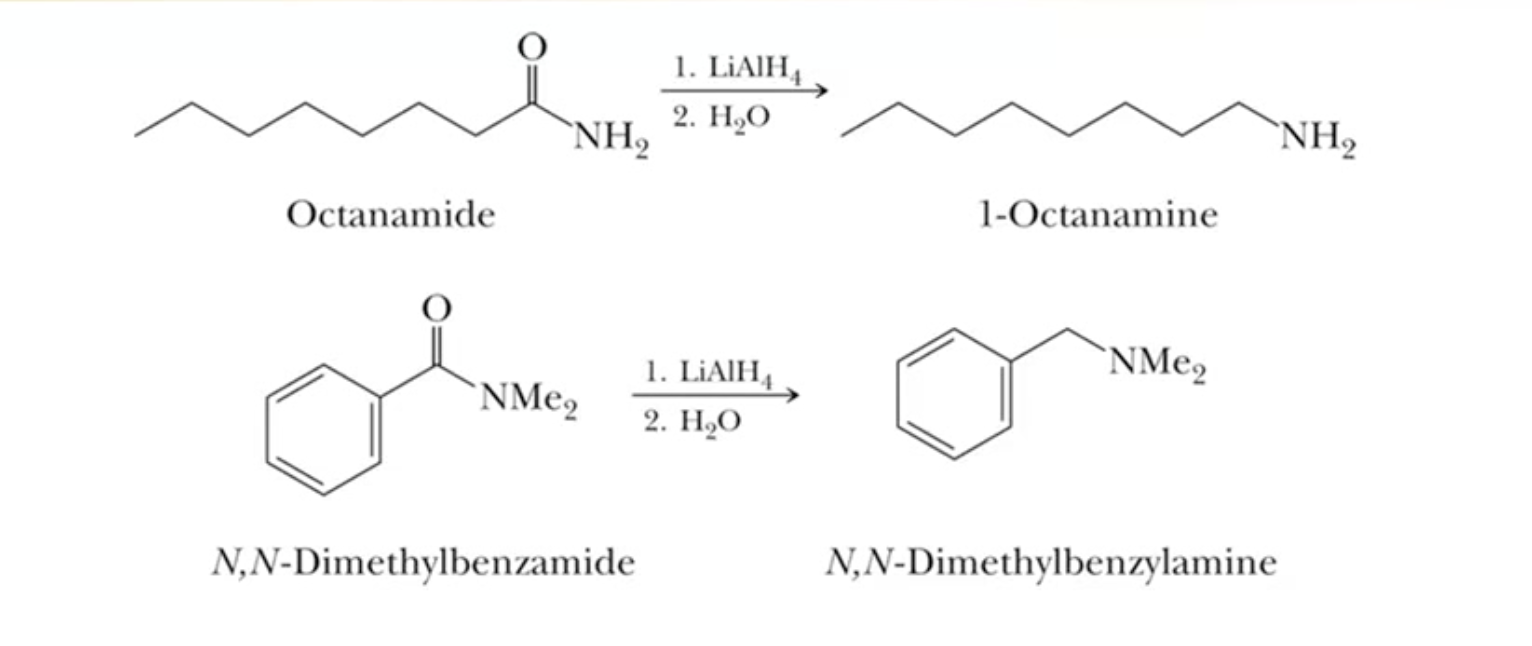
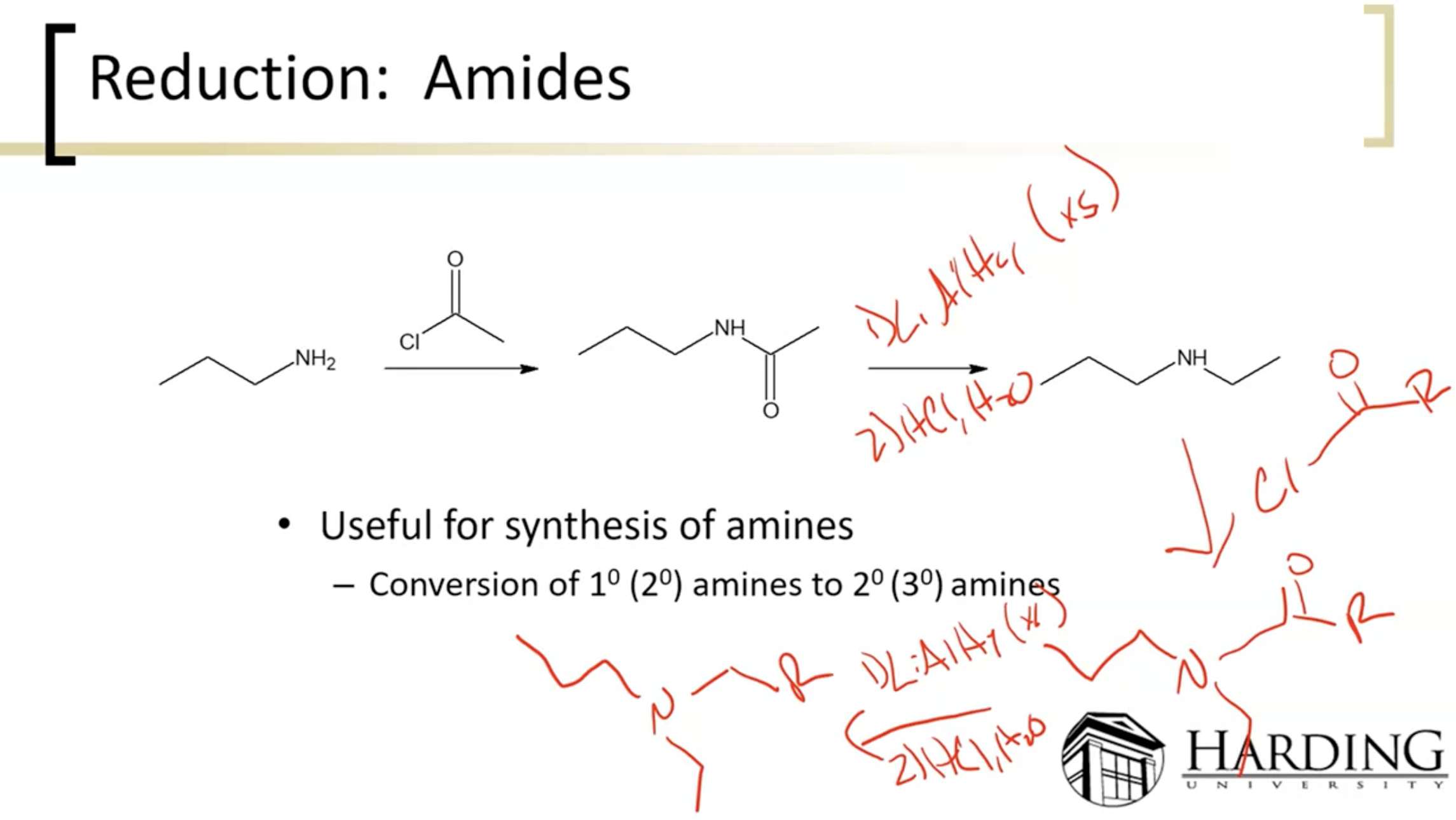
Reduction of Amide
using lithium aluminum hydride, you can make primary/secondary/tertiary amines
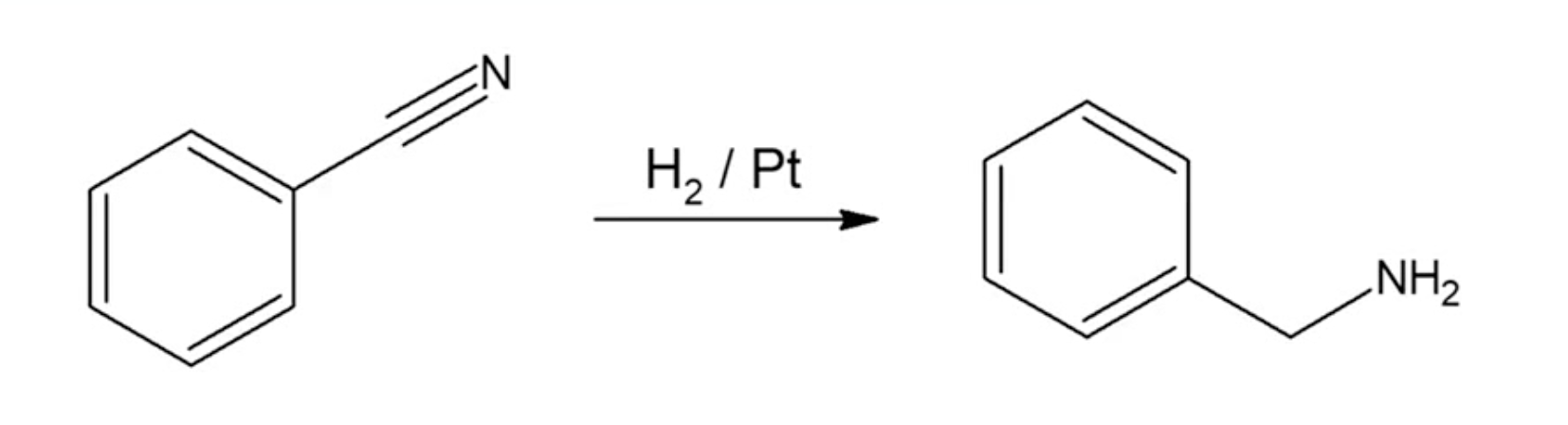
Reduction of Nitriles
use LiAlH4 or H2/Pt
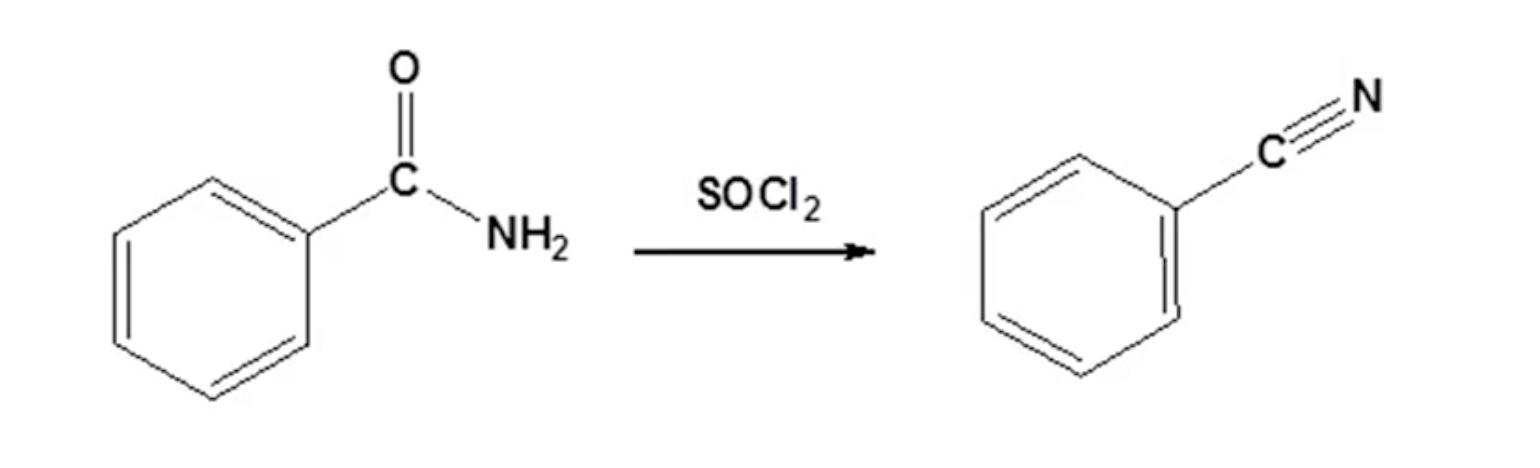
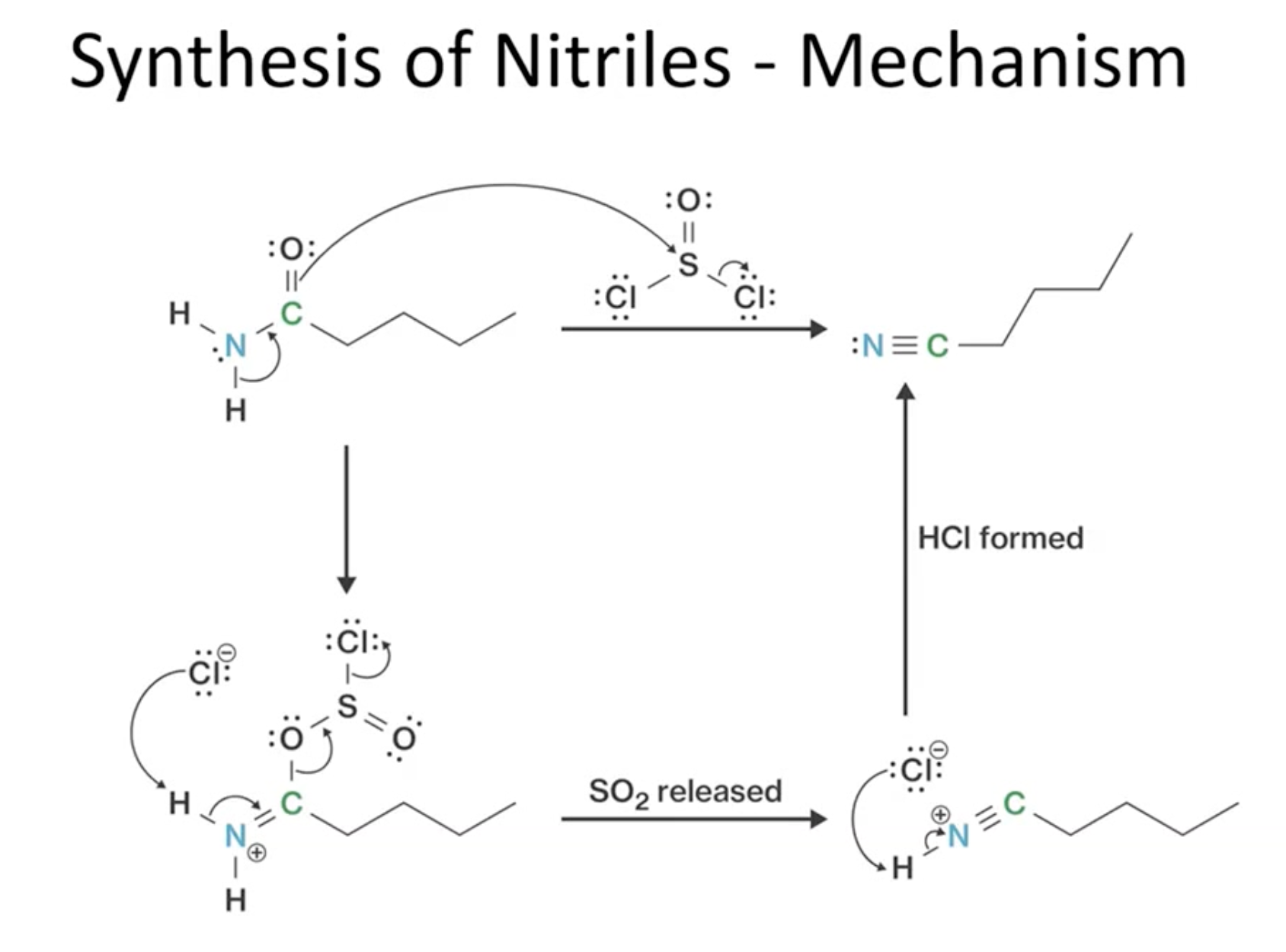
Synthesis of Nitriles