ELECTRIC FIELD AND CHARGES
ELECTRIC CHARGE
Amber a fossilized tree resin, when rubbed with wool or silk cloth attracts light weighted objects. This was discovered by Thales of Miletus, Greece, around 600 BC.
The name electricity is coined from the Greek word elektron meaning amber. Many such pairs of materials were known which on rubbing could attract light objects like straw, pith balls and bits of papers.
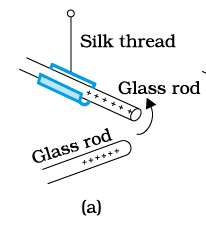
So in (a) part
Glass rods will repel each other
Silk thread will repel each other
Glass rod and silk thread will attract each other
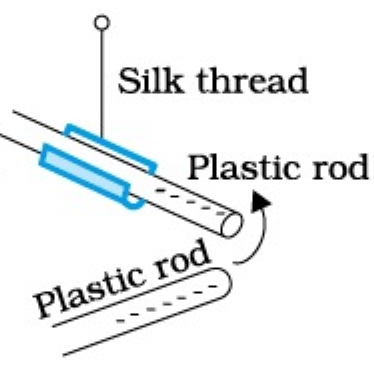
Same as (a) part
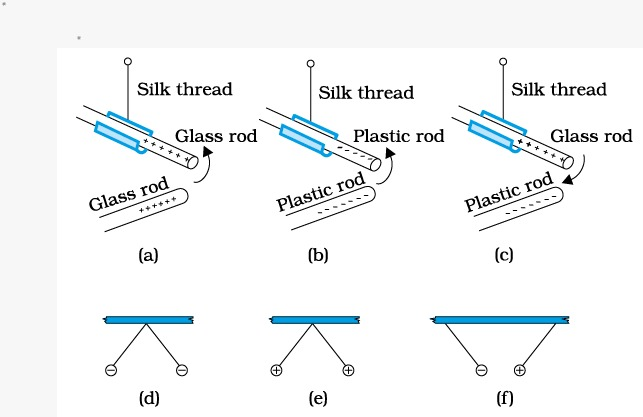
In ( c) part,
1. Plastic rod and glass rod attract each other
2. Plastic rod repels silk thread
In (d) part,
Both the balls are touched by plastic rods which are rubbed with silk repel each other
In (e) part,
Both the balls are touched with glass rod which are rubbed with silk repel each other
In (f) part,
One ball is touched with glass rod whereas other one is touched with plastic rod , so they both attract each other
The experiments on pith balls suggested that there are two kinds of electrification and we find that
(i) like charges repel and
(ii) unlike charges attract each other.
The experiments also demonstrated that the charges are transferred from the rods to the pith balls on contact. It is said that the pith balls are electrified or are charged by contact. The property which differentiates the two kinds of charges is called the polarity of charge.
When a glass rod is rubbed with silk, the rod acquires one kind of charge and the silk acquires the second kind of charge. This is true for any pair of objects that are rubbed to be electrified. Now if the electrified glass rod is brought in contact with silk, with which it was rubbed, .they no longer attract each other They also do not attract or repel other light objects as they did on being electrified.
Thus, the charges acquired after rubbing are lost when the charged bodies are brought in contact.
What can you conclude from these observations?
It just tells us that unlike charges acquired by the objects neutralise or nullify each other’s effect.
Therefore, the charges were named as positive and negative by the American scientist Benjamin Franklin. We know that when we add a positive number to a negative number of the same magnitude, the sum is zero. This might have been the philosophy in naming the charges as positive and negative. By convention, the charge on glass rod or cat’s fur is called positive and that on plastic rod or silk is termed negative. If an object possesses an electric charge, it is said to be electrified or charged. When it has no charge it is said to be electrically neutral.
Gold leaf electrosope
A simple apparatus to detect charge on a body is the gold-leaf electroscope [Fig. 1.2(a)]. It consists of a vertical metal rod housed in a box, with two thin gold leaves attached to its bottom end. When a charged object touches the metal knob at the top of the rod, charge flows on to the leaves and they diverge. The degree of divergance is an indicator of the amount of charge.

Unification of electricity and magnetism
Electricity and magnetism were once separate subjects, but discoveries by Oersted, Ampere, and Faraday showed they are linked. Maxwell and Lorentz unified them into electromagnetism, a fundamental force behind most natural and technological phenomena. Maxwell's equations describe this field and show that light is electromagnetic. Electromagnetism powers modern life—electricity, communication, and appliances—and is much stronger than gravity.Electromagnetic force is one of the fundamental forces of nature.
Working of gold leaf electroscope
To understand how the electroscope works, use the white paper strips we used or seeing the attraction of charged bodies. Fold the strips into half so that you make a mark of fold. Open the strip and iron it lightly with the mountain fold up, as shown in Fig. 1.3. Hold the strip by pinching it at the fold. You would notice that the two halves move apart. This shows that the strip has acquired charge on ironing. When you fold it into half, both the halves have the same charge. Hence they repel each other.
The same effect is seen in the leaf electroscope. On charging the curtain rod by touching the ball end with an electrified body, charge is transferred to the curtain rod and the attached aluminium foil. Both the halves of the foil get similar charge and therefore repel each other. The divergence in the leaves depends on the amount of charge on them.
You know that all matter is made up of atoms and/or molecules. Although normally the materials are electrically neutral, they do contain charges; but their charges are exactly balanced. Forces that hold the molecules together, forces that hold atoms together in a solid, the adhesive force of glue, forces associated with surface tension, all are basically electrical in nature, arising from the forces between charged particles. Thus the electric force is all pervasive and it encompasses almost each and every field associated with our life. It is therefore essential that we learn more about such a force
To electrify a neutral body, we need to add or remove one kind of charge. When we say that a body is charged, we always refer to this excess charge or deficit of charge. In solids, some of the electrons, being less tightly bound in the atom, are the charges which are transferred from one body to the other. A body can thus be charged positively by losing some of its electrons. Similarly, a body can be charged negatively by gaining electrons.
When we rub a glass rod with silk, some of the electrons from the rod are transferred to the silk cloth. Thus the rod gets positively charged and the silk gets negatively charged.
No new charge is created in the process of rubbing. Also the number of electrons, that are transferred, is a very small fraction of the total number of electrons in the material body. Also only the less tightly bound electrons in a material body can be transferred from it to another by rubbing. Therefore, when a body is rubbed with another, the bodies get charged and that is why we have to stick to certain pairs of materials to notice charging on rubbing the bodies.
CONDUCTORS & INSULATORS
A metal rod held in hand and rubbed with wool will not show any sign of being charged. However, if a metal rod with a wooden or plastic handle is rubbed without touching its metal part, it shows signs of charging. Suppose we connect one end of a copper wire to a neutral pith ball and the other end to a negatively charged plastic rod. We will find that the pith ball acquires a negative charge. If a similar experiment is repeated with a nylon thread or a rubber band, no transfer of charge will take place from the plastic rod to the pith ball. Why does the transfer of charge not take place from the rod to the ball?
Some substances readily allow passage of electricity through them, others do not. Those which allow electricity to pass through them easily are called conductors. They have electric charges (electrons) that are comparatively free to move inside the material. Metals, human and animal bodies and earth are conductors. Most of the non-metals like glass, porcelain, plastic, nylon, wood offer high resistance to the passage of electricity through them. They are called insulators. Most substances fall into one of the two classes stated above*.
When some charge is transferred to a conductor, it readily gets distributed over the entire surface of the conductor. In contrast, if some charge is put on an insulator, it stays at the same place
This property of the materials tells you why a nylon or plastic comb gets electrified on combing dry hair or on rubbing, but a metal article like spoon does not. The charges on metal leak through our body to the ground as both are conductors of electricity.
When we bring a charged body in contact with the earth, all the excess charge on the body disappears by causing a momentary current to pass to the ground through the connecting conductor (such as our body). This process of sharing the charges with the earth is called grounding or earthing. Earthing provides a safety measure for electrical circuits and appliances. A thick metal plate is buried deep into the earth and thick wires are drawn from this plate; these are used in buildings for the purpose of earthing near the mains supply. The electric wiring in our houses has three wires: live, neutral and earth. The first two carry electric current from the power station and the third is earthed by connecting it to the buried metal plate. Metallic bodies of the electric appliances such as electric iron, refrigerator, TV are connected to the earth wire. When any fault occurs or live wire touches the metallic body, the charge flows to the earth without damaging the appliance and without causing any injury to the humans; this would have otherwise been unavoidable since the human body is a conductor of electricity.
There is a third category called semiconductors, which offer resistance to the movement of charges which is intermediate between the conductors and insulators
SUMMARY
Amber, when rubbed, attracts light objects, leading to the term 'electricity' from the Greek word 'elektron.' Experiments show two types of electrification: like charges repel and unlike charges attract. Charges transfer via contact, defining charge polarity. Benjamin Franklin named charges positive and negative; convention dictates glass rods rubbed with silk are positive, while plastic rods are negative.
A gold-leaf electroscope detects charge; leaves diverge with charge presence. Electricity and magnetism are unified into electromagnetism, governing various phenomena. Electromagnetism is one of nature's fundamental forces, seen in everyday life.
Materials are neutral with balanced charges. Electrifying a body involves adding or removing charges, typically electrons. Rubbing a glass rod with silk transfers electrons, charging them oppositely.
Conductors allow electricity to pass easily, while insulators resist. Conductors distribute charge across their surface, whereas insulators hold charge in place. Grounding shares charges
CHARGING BY INDUCTION
(i) Bring two metal spheres, A and B, supported on insulating stands, in contact as shown in Fig. 1.4(a).

(ii) Bring a positively charged rod near one of the spheres, say A, taking care that it does not touch the sphere.
The free electrons in the spheres are attracted towards the rod. This leaves an excess of positive charge on the rear surface of sphere B. Both kinds of charges are bound in the metal spheres and cannot escape. They, therefore, reside on the surfaces, as shown in Fig. 1.4(b).
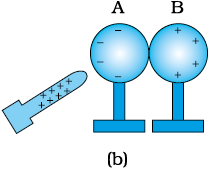
The left surface of sphere A, has an excess of negative charge and the right surface of sphere B, has an excess of positive charge. However, not all of the electrons in the spheres have accumulated on the left surface of A. As the negative charge starts building up at the left surface of A, other electrons are repelled by these. In a short time, equilibrium is reached under the action of force of attraction of the rod and the force of repulsion due to the accumulated charges. Fig. 1.4(b) shows the equilibrium situation. The process is called induction of charge and happens almost instantly. The accumulated charges remain on the surface, as shown, till the glass rod is held near the sphere. If the rod is removed, the charges are not acted by any outside force and they redistribute to their original neutral state.
(iii) Separate the spheres by a small distance while the glass rod is still held near sphere A, as shown in Fig. 1.4(c). The two spheres are found to be oppositely charged and attract each other.
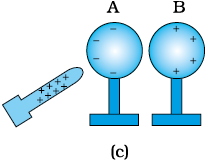
(iv) Remove the rod. The charges on spheres rearrange themselves as shown in Fig. 1.4(d).
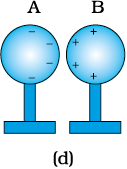
Now, separate the spheres quite apart. The charges on them get uniformly distributed over them, as shown in
Fig. 1.4(e).
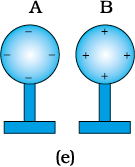
In this process, the metal spheres will each be equal and oppositely charged. This is charging by induction. The positively charged glass rod does not lose any of its charge, contrary to the process of charging by contact.
When electrified rods are brought near light objects, a similar effect takes place. The rods induce opposite charges on the near surfaces of the objects and similar charges move to the farther side of the object. The centres of the two types of charges are slightly separated. We know that opposite charges attract while similar charges repel. However, the magnitude of force depends on the distance between the charges and in this case the force of attraction overweighs the force of repulsion. As a result the particles like bits of paper or pith balls, being light, are pulled towards the rods.
COULOMB’S LAW
Coulomb’s law is a quantitative statement about the force between two point charges.
When the linear size of charged bodies are much smaller than the distance separating them, the size may be ignored and the charged bodies are treated as point charges.
Coulomb measured the force between two point charges and found that it varied inversely as the square of the distance between the charges and was directly proportional to the product of the magnitude of the two charges and acted along the line joining the two charges.
Thus, if two point charges q1, q2 are separated by a distance r in vacuum, the magnitude of the force (F) between them is given by
F=\frac{kq1q2}{r^2}
How did Coulomb arrive at this law from his experiments?
Coulomb used a torsion balance* for measuring the force between two charged metallic spheres. When the separation between two spheres is much larger than the radius of each sphere, the charged spheres may be regarded as point charges. However, the charges on the spheres were unknown, to begin with. How then could he discover a relation like Eq. (1.1)?
Coulomb thought of the following simple way: Suppose the charge on a metallic sphere is q. If the sphere is put in contact with an identical uncharged sphere, the charge will spread over the two spheres. By symmetry, the charge on each sphere will be q/2*. Repeating this process, we can get charges q/2, q/4, etc.
Coulomb varied the distance for a fixed pair of charges and measured the force for different separations. He then varied the charges in pairs, keeping the distance fixed for each pair. Comparing forces for different pairs of charges at different distances, Coulomb arrived at the relation, Eq. (1.1).
A torsion balance is a sensitive device to measure force. It was also used later by Cavendish to measure the very feeble gravitational force between two objects, to verify Newton’s Law of Gravitation.

1 C is the charge that when placed at a distance of 1 m from another charge of the same magnitude in vacuum experiences an electrical force of repulsion of magnitude 9 × 109 N. One coulomb is evidently too big a unit to be used. In practice, in electrostatics, one uses smaller units like 1 mC or 1 µC.
The constant k in Eq. (1.1) is usually put as
k = \frac{1}{4\pi\epsilon}

F12 - It is force on q1 due to q2
F21 - It is force on q2 due to q1
r21 = r2 – r1
r12 = r1 – r2 = – r21
Coulomb’s force law between two point charges q1 and q2 located at r1 and r2 is then expressed as
F=\frac{1}{4\pi\epsilon0}\frac{q1q2}{r^221}r21
Negative sign means that force is attractive and vice versa
gravitational force is always attractive