Option D
D1: Human Nutrition
Nutrient
A chemical substance found in foods that is used in the human body
Classes of nutrients
carbohydrates, proteins, lipids, vitamins, minerals and water
Essential nutrients
Cannot be synthesised by the body and must be ingested as part of the diet
Non-essential nutrients
Can be made by the body or have a replacement nutrient which serves the same dietary purpose
Carbohydrates are not considered essential nutrients as human diets can obtain energy from other sources without ill effect
Malnutrition
Health condition caused by a deficiency, imbalance or excess of nutrients in the diet
Can be caused by an improper dietary intake of nutrients – e.g. overnutrition (too much) or undernutrition (not enough)
Or by the inadequate utilisation of nutrients by the body – e.g. due to illness or disease
Determining Energy Content
The energy content of food can be estimated by burning a sample of known mass and measuring the energy released via calorimetry
Combustion of the food source causes the stored energy to be released as heat, which raises the temperature of water
Energy content (joules) = Mass of water (g) × 4.2 (J/gºC) × Temperature increase (ºC)
Comparing Energy Content
Carbohydrates are preferentially used as an energy source because they are easier to digest and transport
Lipids can store more energy per gram but are harder to digest and transport (hence are used for long-term storage)
Protein metabolism produces nitrogenous waste products which must be removed from cells
Essential amino acids
Cannot be produced by the body and must be present in the diet
Non-essential amino acids
Can be produced by the body and are therefore not required as part of the diet
Conditionally non-essential amino acids
Can be produced by the body, but at rates lower than certain conditional requirements (e.g. during pregnancy or infancy) – they are essential at certain times only
Shortage of amino acids
Prevents the production of proteins (protein deficiency malnutrition)
Cause of phenylketonuria
Phenylketonuria (PKU) is a genetic condition that results in the impaired metabolism of the amino acid phenylalanine
It is an autosomal recessive disease caused by a mutation to the gene encoding the enzyme phenylalanine hydroxylase (PAH)
PAH normally converts excess phenylalanine within the body into tyrosine
In people with PKU, the excess phenylalanine results in a toxic build up of phenylketone in the blood and urine (hence phenylketonuria)
Treatment of phenylketonuria
PKU is treated by enforcing a strict diet that restricts the intake of phenylalanine to prevent its build up within the body
This low-protein diet should include certain types of fruits, grains, vegetables and special formula milk
This diet should be supplemented with a medical formula that contains precise quantities of essential amino acids
Patients who are diagnosed early and maintain this strict diet can have a normal life span without damaging symptoms
Essential Fatty acids
Alpha-linolenic acid (an omega-3 fatty acid) and linoleic acid (an omega-6 fatty acid) cannot be synthesised by the body
This is because humans lack the enzyme required to introduce double bonds at the required position of the carbon chain
Essential fatty acids are modified by the body to make important lipid-based compounds (such as signalling molecules)
Low density lipoproteins (LDLs)
Carry cholesterol from the liver to the body (hence raise blood cholesterol levels)
High density lipoproteins (HDLs)
Carry excess cholesterol back to the liver for disposal (hence lower blood cholesterol levels)
High cholesterol levels in the bloodstream
Lead to the hardening and narrowing of arteries (atherosclerosis)
When there are high levels of LDL in the bloodstream, the LDL particles will form deposits in the walls of the arteries
The accumulation of fat within the arterial wall leads to the development of plaques which restrict blood flow
If coronary arteries become blocked, coronary heart disease (CHD) will result – this includes heart attacks and strokes
Vitamins
Chemically diverse carbon compounds that cannot be synthesised by the body
Ascorbic acid
Form of vitamin C required for metabolic activities in animals and plants
Made internally by most mammals from monosaccharides BUT NOT HUMANS
Humans must ingest as part of their dietary intake
Vitamin D
Involved in the absorption of calcium and phosphorus by the body (bone mineralisation)
Naturally synthesised in the presence of UV light
Lack of Vitamin D/Calcium
Lack of vitamin D will reduce calcium absorption
Calcium maintains the strength and rigidity of bones
This can lead to osteomalacia (softening of bones due to inadequate mineralisation of bone tissue)
Dietary minerals
Essential chemical elements required as essential nutrients by organisms
Humans: Ca, P, Mg, Na, K, Cl, Fe, I
Plants: Mg, K, O
Appetite
Appetite is controlled by hormones produced in the pancreas, stomach, intestines and adipose tissue
These hormones send messages to the appetite control centre of the brain (within the hypothalamus)
Hormonal signals will either trigger a feeling of hunger (promote feasting) or satiety (promote fasting)
Triggering hormones for appetite
Stretch receptors in the stomach and intestine become activated when ingested food distends these organs
Adipose tissue releases hormones in response to fat storage
The pancreas will release hormones in response to changes in blood sugar concentrations
Triggering a hunger response
ghrelin (from stomach)
glucagon (from pancreas)
Triggereing a satiety response
leptin (from adipose tissue)
CCK (from intestine)
Obesity
Clinical obesity (BMI > 30) describes a significant excess in body fat and is caused by a combination of two factors:
Increased energy intake (i.e. overeating or an increased reliance on diets rich in fats and sugars)
Decreased energy expenditure (i.e. less exercise resulting from an increasingly sedentary lifestyle)
Hypertension
Individuals who are overweight/obese are more likely to suffer from hypertension
hypertension = abnormally high blood pressure
Excess weight places more strain on the heart to pump blood, leading to a faster heart rate and higher blood pressure
High cholesterol diets will lead to atherosclerosis, narrowing the blood vessels which contributes to raised blood pressure
Hypertension is a common precursor to CHD
Type 2 diabetes
Individuals who are overweight/obese are more likely to suffer from type 2 diabetes
Type II diabetes occurs when fat, liver and muscle cells become unresponsive to insulin (insulin insensitivity)
This typically results from a diet rich in sugars causing the progressive overstimulation of these cells by insulin
Hence overweight individuals who have a high sugar intake are more likely to develop type II diabetes
Starvation
Starvation describes the severe restriction of daily energy intake, leading to a significant loss of weight
As the body is not receiving a sufficient energy supply from the diet, body tissue is broken down as an energy source
This leads to muscle loss (as muscle proteins are metabolised for food) and eventually organ damage (and death)
Effects of severe anorexia
The body begins to break down heart muscle, making heart disease the most common cause of death
Blood flow is reduced and blood pressure may drop as heart tissue begins to starve
The heart may also develop dangerous arrhythmias and become physically diminished in size
RDI
Recommended daily intake for a nutrient
The recommendations are based on a daily energy intake of 8400 kJ (2000 kcal) for healthy adults
On food packages, this information is usually presented as a percentage of a daily total (based on identified serving size)
D2: Digestion
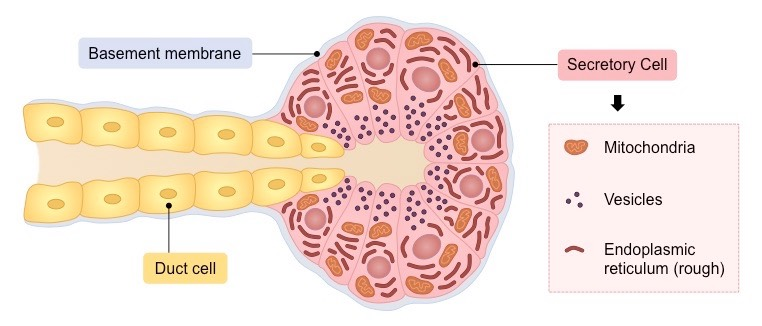
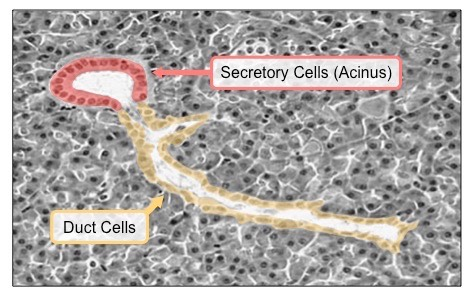
Exocrine glands
Produce and secrete substances via a duct onto an epithelial surface
Surface of the body (sweat glands, sebaceous glands)
Lumen of the digestive tract (digestive glands)
Structure of a Typical Exocrine Gland
Electron Micrograph of an Exocrine Gland
Nervous control of gastric secretion
The sight and smell of food triggers an immediate response by which gastric juice is secreted by the stomach pre-ingestion
When food enters the stomach it causes distension, which is detected by stretch receptors in the stomach lining
Signals are sent to the brain, which triggers the release of digestive hormones to achieve sustained gastric stimulation
Hormonal control of gastric secretion
Gastrin is secreted into the bloodstream from the gastric pits of the stomach and stimulates the release of stomach acids
If stomach pH drops too low, gastrin secretion is inhibited by gut hormones (secretin and somatostatin)
When digested food (chyme) passes into the small intestine, the duodenum also releases digestive hormones:
Secretin and cholecystokinin (CCK) stimulate the pancreas and liver to release digestive juices
Pancreatic juices contain bicarbonate ions which neutralise stomach acids, while the liver produces bile to emulsify fats
Stomach acid
Between optimum of pH 1-3
Assists in the digestion of food (by dissolving chemical bonds within food molecules)
Activates stomach proteases (e.g. pepsin is activated when pepsinogen is proteolytically cleaved in acid conditions)
Prevents pathogenic infection (stomach acids destroy microorganisms in ingested food)
Proton pump inhibitor drugs
PPIs irreversibly bind to the proton pumps and prevent H+ ion secretion in gastric pits and hence reduces stomach acid secretion
This effectively raises the pH in the stomach to prevent gastric discomfort caused by high acidity (e.g. acid reflux)
Individuals taking PPIs may have increased susceptibility to gastric infections due to the reduction of acid secretion
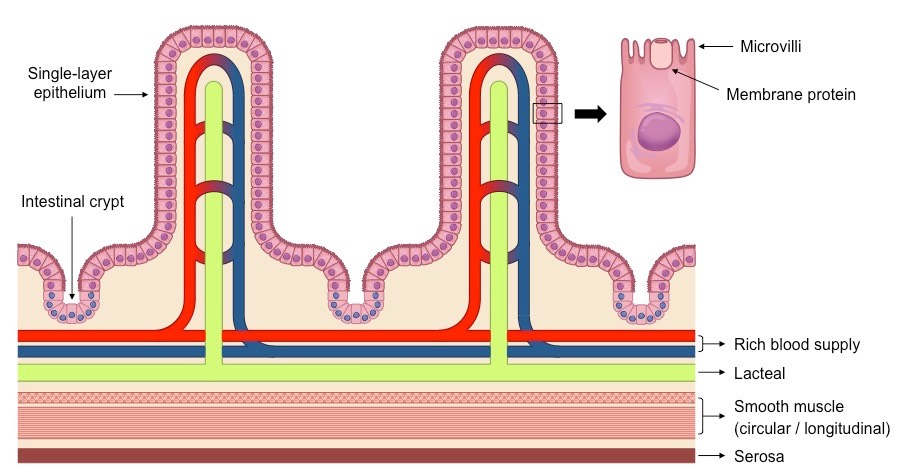
Features of Villi
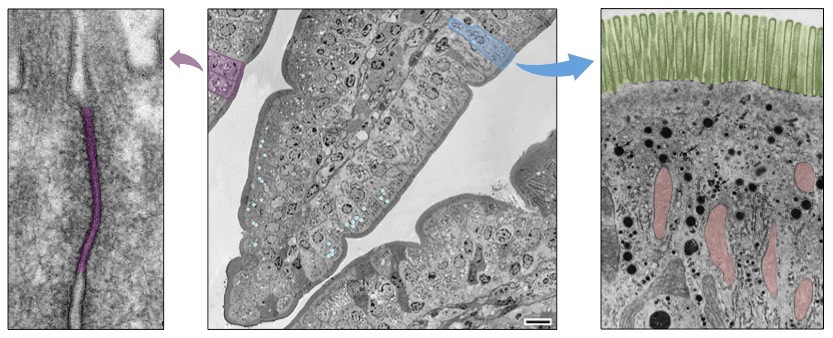
Microvilli – Ruffling of epithelial membrane further increases surface area
Rich blood supply – Dense capillary network rapidly transports absorbed products
Single layer epithelium – Minimises diffusion distance between lumen and blood
Lacteals – Absorbs lipids from the intestine into the lymphatic system
Intestinal glands – Exocrine pits (crypts of Lieberkuhn) release digestive juices
Membrane proteins – Facilitates transport of digested materials into epithelial cells
Diagram of Intestinal Villi
 Electron Micrograph of Villus Epithelium
Electron Micrograph of Villus Epithelium
Dietary Fibre
The indigestible portion of food derived from plants and fungi
The rate of transit of materials through the large intestine is positively correlated with their fibre content
Fibre provides bulk in the intestines to help keep materials moving in the gut and absorbs water for easier bowel movements
Egestion
Materials that are not absorbed by the small and large intestines are ultimately egested from the body as faeces
A large portion of human faeces consists of dietary fibre, such as cellulose and lignin
Also present in faeces are the remains of intestinal epithelial cells, bile pigments and human flora (intestinal bacteria)
Stomach ulcers
Inflamed and damaged areas in the stomach wall, typically caused by exposure to gastric acids
Helicobacter pylori infection
Helicobacter pylori is a bacterium that can survive the acid conditions of the stomach
H. pylori penetrates the mucus layer lining the stomach
The bacteria then damages the goblet cells responsible for mucus production
The loss of mucus exposes cells in the stomach wall to gastric acids and causes ulcers
Vibrio cholerae
A bacterial pathogen that infects the intestines and causes acute diarrhoea and dehydration
The associated disease – cholera – can kill within hours unless treated with oral rehydration therapies
Cholera toxin
V. cholerae releases a toxin that binds to receptors on the surface of intestinal epithelium cells
This toxin is internalised by endocytosis and triggers the production of cyclic AMP (a second messenger) within the cell
Cyclic AMP (cAMP) activates specific ion channels within the cell membrane, causing an efflux of ions from the cell
The build up of ions in the intestinal lumen draws water from cells and tissues via osmosis – causing acute diarrhoea
As water is being removed from body tissues, dehydration will result if left untreated
D3: Functions of the liver
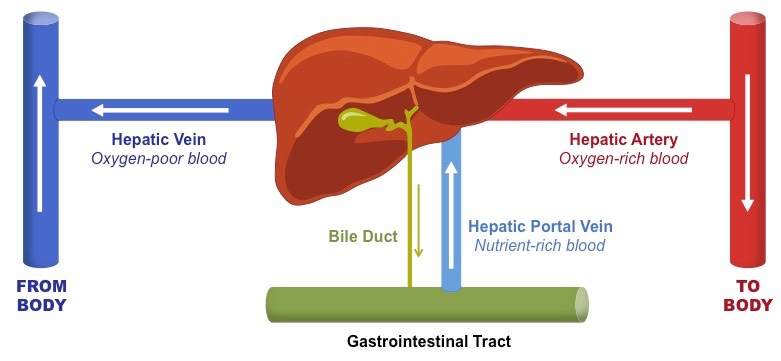
Liver blood flow
The liver is a lobed organ located below the diaphragm that functions to regulate the chemical composition of blood
It receives oxygenated blood via the hepatic artery, which is used to sustain liver cells (hepatocytes)
It also receives nutrient rich blood from the gut via the portal vein
Deoxygenated blood is transported from the liver via the hepatic vein
Liver functions
The liver functions to process the nutrients absorbed from the gut and hence regulates the body’s metabolic processes
It is responsible for the storage and controlled release of key nutrients (e.g. glycogen, cholesterol, triglycerides)
It is responsible for the detoxification of potentially harmful ingested substances (e.g. amino acids, medications, alcohol)
It produces plasma proteins that function to maintain sustainable osmotic conditions within the bloodstream
It is responsible for the breakdown of red blood cells and the production of bile salts
Overview of Hepatic Circulation
Hepatic Lobules
The liver is composed of smaller histological structures called lobules, which are roughly hexagonal in shape
Each lobule is surrounded by branches of the hepatic artery (provide oxygen) and the portal vein (provide nutrients)
These vessels drain into capillary-like structures called sinusoids, which exchange materials directly with the hepatocytes
The sinusoids drain into a central vein, which feeds deoxygenated blood into the hepatic vein
Hepatocytes also produce bile, which is transported by vessels called canaliculi to bile ducts, which surround the lobule
Sinusoids
Sinusoids are a type of small blood vessel found in the liver that perform a similar function to capillaries (material exchange)
Sinusoids have increased permeability, allowing larger molecules (e.g. plasma proteins) to enter and leave the bloodstream
Structure of sinusoids
The surrounding diaphragm (basement membrane) is incomplete or discontinuous in sinusoids (but not in capillaries)
The endothelial layer contains large intercellular gaps and fewer tight junctions (allowing for the passage of larger molecules)
Liver regulating nutrient levels
Nutrients absorbed by the small intestine are transported by the hepatic portal vein to the liver for metabolism
The liver converts these nutrients into forms that can be stored or used and mediates their transport to various tissues
Nutrients stored within the liver include glycogen, iron, vitamin A and vitamin D
Carbohydrate Metabolism
Excess glucose in the bloodstream is taken up by the liver and stored as glycogen
When blood glucose levels drop, the liver breaks down glycogen into glucose and exports it to body tissues
When hepatic glycogen reserves become exhausted, the liver synthesises glucose from other sources (e.g. fats)
These metabolic processes are coordinated by the pancreatic hormones – insulin and glucagon
Protein Metabolism
The body can not store amino acids, they must be broken down in excess
Amino acid breakdown releases an amine group (NH2), which cannot be used by the body and is potentially toxic
The liver is responsible for the removal of the amine group (deamination) and its conversion into a harmless product
The amine group is converted into urea by the liver, which is excreted within urine by the kidneys
The liver can also synthesise non-essential amino acids from surplus stock (via transamination)
Fat Metabolism
The liver is the major site for converting excess carbohydrates and proteins into fatty acids and triglycerides
It is also responsible for the synthesis of large quantities of phospholipids and cholesterol
These compounds are then stored by the liver or exported to cells by different types of lipoproteins (LDL and HDL)
Surplus cholesterol is converted by the liver into bile salts, which can be eliminated from the body via the bowels
Liver detoxification
Toxins are converted into less harmful chemicals by oxidation, reduction and hydrolysis reactions
These reactions are mediated by the cytochrome P450 enzyme group
These conversions produce damaging free radicals, which are neutralised by antioxidants within the liver
The converted chemical is then attached to another substance (e.g. cysteine) via a conjugation reaction
This renders the compound even less harmful and also makes it water soluble
The compounds can now be excreted from the body within urine by the kidneys
Plasma proteins
Proteins present in the blood plasma and are produced by the liver (except for immunoglobulins)
The proteins are produced by the rough ER in hepatocytes and exported into the blood via the Golgi complex
Types of plasma proteins
Albumins regulate the osmotic pressure of the blood (and hence moderate the osmotic pressure of body fluids)
Globulins participate in the immune system (i.e. immunoglobulins) and act as transport proteins
Fibrinogens are involved in the clotting process (soluble fibrinogen can form an insoluble fibrin clot)
Low levels of other plasma proteins have various functions (e.g. α-1-antitrypsin neutralises digestive trypsin)
Red blood cell recycling
In humans, red blood cells possess minimal organelles and no nucleus in order to carry more haemoglobin
Consequently, red blood cells have a short lifespan (~120 days) and must be constantly replaced
The liver is responsible for the break down of red blood cells and recycling of its components
These components are used to make either new red blood cells or other important compounds (e.g. bile)
Process of RBC recycling
Kupffer cells are specialised phagocytes within the liver which engulf red blood cells and break them down
Kupffer cells break down haemoglobin into globin and iron-containing heme groups
Globin is digested by peptidases to produce amino acids (which are either recycled or metabolised by the liver)
Heme groups are broken down into iron and bilirubin (bile pigment)
The released iron must be complexed within a protein in order to avoid oxidation to a ferric state
Iron can be stored by the liver within a protein shell of ferritin
Iron can be transported to the bone marrow (where new haemoglobin is produced) within the protein transferrin
Process of Erythrocyte and Haemoglobin Recycling
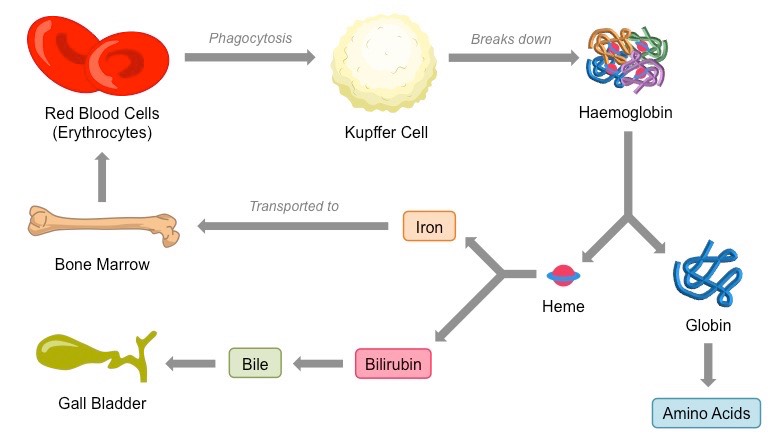
Cause of jaundice
An excess of bile pigment – bilirubin – within the body
Bilirubin is produced as part of the natural breakdown of haemoglobin by the liver
Normally, the liver conjugates this bilirubin to other chemicals and then secretes it in bile
When there is an excess of bilirubin, it may leak out into surrounding tissue fluids
Conditions leading to jaundice
Liver disease – impaired removal of bilirubin by the liver may cause levels to build within the body
Obstruction of the gall bladder – preventing the secretion of bile will cause bilirubin levels to accumulate
Damage to red blood cells – increased destruction of erythrocytes (e.g. anemia) will cause bilirubin levels to rise
Consequences of jaundice
The main consequence of jaundice is a yellowish discoloration of the skin and whites of the eyes (sclera)
Other common symptoms include itchiness, paler than usual stools and darkened urine
D4: The Heart
Cardiac muscle cell features
Contract without stimulation by the central nervous system (contraction is myogenic)
Branched, allowing for faster signal propagation and contraction in three dimensions
Not fused together, but are connected by gap junctions at intercalated discs
More mitochondria, as they are more reliant on aerobic respiration than skeletal muscle
Cardiac muscle properties
Longer period of contraction and refraction, to maintain a viable heart beat
The heart tissue does not become fatigued (unlike skeletal muscle), allowing for continuous, life long contractions
The interconnected network of cells is separated between atria and ventricles, allowing them to contract separately
Cardiac conduction
Cardiac muscle cells are not fused together but are instead connected via gap junctions at intercalated discs
This means that while electrical signals can pass between cells, each cell is capable of independent contraction
The coordinated contraction of cardiac muscle cells is controlled by specialised autorhythmic cells (‘pace makers’)
Atrial Contraction (systole)
Right atrium walls contain cardiomyocytes which directs the contraction of heart tissue
This cluster of cells the sinoatrial node (SA node or SAN)
SAN acts as a primary pacemaker, controlling the rate at which the heart beats
It sends out electrical signals which are propagated throughout the entire atria via gap junctions in the intercalated discs
In response, the cardiac muscle within the atrial walls contract simultaneously (atrial systole)
Connective tissue
The atria and ventricles of the heart are separated by a fibrous cardiac skeleton composed of connective tissue
This connective tissue anchors the heart valves and cannot conduct electrical signals
The signals from the SAN must instead be relayed through the atrioventricular node (or AV node) located within this cardiac skeleton
AV node separates atrial and ventricular contractions
The AV node propagates electrical signals more slowly than the SA node, creating a delay in the passing on of the signal
The delay in time following atrial systole allows for blood to fill the ventricles before the atrioventricular valves close
Ventricular Contraction
Ventricular contraction occurs following excitation of the AV node (located at the atrial and ventricular junction)
The AV node sends signals down the septum via a specialised bundle of cardiomyocytes called the Bundle of His
The Bundle of His innervates Purkinje fibres in the ventricular wall, which causes the cardiac muscle to contract
This sequence of events ensures contractions begin at the apex (bottom), forcing blood up towards the arteries
Heart Relaxation / Diastole
After every contraction of the heart, there is a period of insensitivity to stimulation (i.e. a refractory period)
This recovery period (diastole) is relatively long, and allows the heart to passively refill with blood between beats
This long recovery period also helps prevent heart tissue becoming fatigued, allowing contractions to continue for life
Valves in heart
Atrioventricular valves (tricuspid and bicuspid) prevent blood in the ventricles from flowing back into the atria
Semilunar valves (pulmonary and aortic) prevent blood in the arteries from flowing back into the ventricles
Heart sounds
Heart sounds are made when the two sets of valves close in response to pressure changes within the heart
The first heart sound is caused by the closure of the atrioventricular valves at the start of ventricular systole
The second heart sound is caused by the closure of the semilunar valves at the start of ventricular diastole
Electrocardiography
The cardiac cycle can be mapped by recording the electrical activity of the heart with each contraction
Activity is measured using a machine called an electrocardiograph to generate data called an electrocardiogram
Electrical Activity of the Heart
The P wave represents depolarisation of the atria in response to signalling from the sinoatrial node (i.e. atrial contraction)
The QRS complex represents depolarisation of the ventricles (i.e. ventricular contraction), triggered by signals from the AV node
The T wave represents repolarisation of the ventricles (i.e. ventricular relaxation) and the completion of a standard heart beat
Between these periods of electrical activity are intervals allowing for blood flow (PR interval and ST segment)
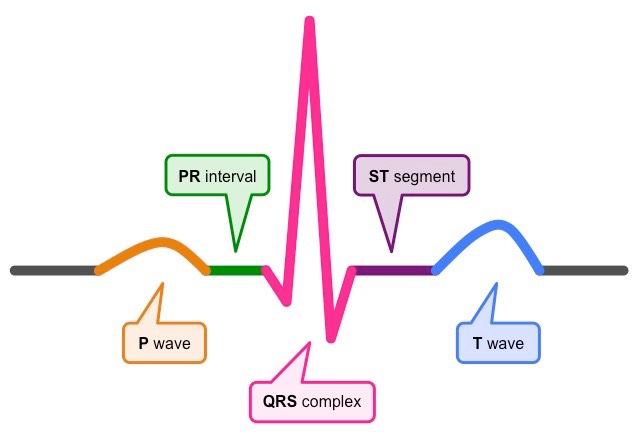
Cardiac output
The amount of blood the heart pumps through the circulatory system in one minute
It is an important medical indicator of how efficiently the heart can meet the demands of the body
Equation: Cardiac Output (CO) = Heart Rate (HR) × Stroke Volume (SV)
Heart Rate
The speed at which the heart beats, measured by the number of contractions per minute (or bpm)
Each ventricular contraction forces a wave of blood through the arteries which can be detected as a pulse
Heart rate can be affected by a number of conditions – including exercise, age, disease, temperature and emotional state
Additionally, the body will attempt to compensate for any changes to stroke volume with a corrective alteration to heart rate
Controlling heart rate
Increased by the sympathetic nervous system and decreased by parasympathetic stimulation (vagus nerve)
Heart rate can also be increased hormonally via the action of adrenaline / epinephrine
Stroke volume
The amount of blood pumped to the body (from the left ventricle) with each beat of the heart
It is affected by the volume of blood in the body, the contractility of the heart and the level of resistance from blood vessels
Blood pressure readings
Systolic blood pressure is higher, as it represents the pressure of the blood following the contraction of the heart
Diastolic blood pressure is lower, as it represents the pressure of the blood while the heart is relaxing between beats
A typical adult is expected to have an approximate blood pressure in their brachial artery of 120/80 mmHg to 140/90 mmHg
Hypertension
Hypertension is defined as an abnormally high blood pressure – either systolic, diastolic or both (e.g. > 140/90 mmHg)
Common causes of hypertension include a sedentary lifestyle, salt or fat-rich diets and excessive alcohol or tobacco use
High blood pressure can also be secondary to other conditions (e.g. kidney disease) or caused by some medications
Hypertension itself does not cause symptoms but in the long-term leads to consequences caused by narrowing blood vessels
Thrombosis
Thrombosis is the formation of a clot within a blood vessel
Thrombosis occurs in arteries when the vessels are damaged as a result of the deposition of cholesterol (atherosclerosis)
Atheromas (fat deposits) develop in the arteries and significantly reduce the diameter of the vessel (leading to hypertension)
The high blood pressure damages the arterial wall, forming lesions known as atherosclerotic plaques
If a plaque ruptures, blood clotting is triggered, forming a thrombus that restricts blood flow
If the thrombus becomes dislodged it becomes an embolus and can cause blockage at another site
Thrombosis in the coronary arteries leads to heart attacks, while thrombosis in the brain causes strokes
Coronary heart disease (CHD)
The condition caused by the build up of plaque within the coronary arteries
It is essentially the consequence of atherosclerosis in the blood vessels that supply and sustain heart tissue
The incidence of coronary heart disease will vary in different populations according to the occurrence of certain risk factors
Artificial pacemaker
An artificial pacemaker is a medical device that delivers electrical impulses to the heart in order to regulate heart rate
Modern pacemakers are externally programmable, allowing cardiologists to make adjustments as required
Artificial pacemakers are typically used to treat: Abnormally slow heart rates (bradycardia) and arrhythmias arising from blockages within the heart’s electrical conduction system
Fibrillation
The rapid, irregular and unsynchronised contraction of the heart muscle fibres
This causes heart muscle to convulse spasmodically rather than beat in concert, preventing the optimal flow of blood
Defibrillator
Fibrillation is treated by applying a controlled electrical current to the heart via a device called a defibrillator
This functions to depolarise the heart tissue in an effort to terminate unsynchronised contractions
Once heart tissue is depolarised, normal sinus rhythm should hopefully be re-established by the sinoatrial node
D5: Hormones and metabolism
Endocrine system
Comprised of ductless glands that release chemicals into the blood to regulate body functions
The endocrine system is slower to initiate, but has a more prolonged response when compared to the nervous system
Hormone
Chemical messenger that is transported indiscriminately via the bloodstream to act on distant target cells
Specific and will only activate cells or tissues that possess the appropriate target receptor
Endocrine Glands
Endocrine glands secrete their product (hormones) directly into the bloodstream, rather than through a duct (e.g. exocrine gland)
Major endocrine glands include the pancreas, adrenal gland, thyroid gland, pineal gland and the gonads (ovaries and testes)
The hypothalamus and pituitary gland are neuroendocrine glands and function to link the nervous and endocrine systems
Some organs may also secrete hormones despite not being endocrine glands (e.g. adipose tissue secretes leptin)
Steroid Hormones
Steroid hormones are lipophilic (fat-loving) – meaning they can freely diffuse across the plasma membrane of a cell
They bind to receptors in the cytoplasm/nucleus of the target cell, to form an active receptor-hormone complex
This activated complex will move into the nucleus and bind directly to DNA, acting as a transcription factor for gene expression
Examples of steroid hormones include those produced by the gonads (i.e. estrogen, progesterone and testosterone)
Peptide Hormones
Peptide hormones are hydrophllic and lipophobic, they cannot freely cross the plasma membrane
They bind to receptors on the surface of the cell, which are typically coupled to internally anchored proteins (e.g. G proteins)
The receptor complex activates a series of intracellular molecules called second messengers, which initiate cell activity - transduction
Examples of second messengers include cyclic AMP (cAMP), calcium ions (Ca2+), nitric oxide (NO) and protein kinases
The use of second messengers enables the amplification of the initial signal (as more molecules are activated)
Peptide hormones include insulin, glucagon, leptin, ADH and oxytocin
Hypothalamus
The hypothalamus is the section of the brain that links the nervous and endocrine systems in order to maintain homeostasis
It receives information from nerves throughout the body and other parts of the brain and initiates endocrine responses
It secretes neurochemicals (called releasing factors) into a portal system which target the anterior lobe of the pituitary gland
It also secretes hormones directly into the blood via neurosecretory cells that extend into the posterior pituitary lobe
Pituitary Gland
The pituitary gland lies adjacent to the hypothalamus and is in direct contact due to a portal blood system
The pituitary gland receives instructions from the hypothalamus and consists of two lobes (anterior and posterior lobe)
Anterior Lobe
The hypothalamus produces releasing factors, which are released into portal vessels by neurosecretory cells
The releasing factors cause endocrine cells in the anterior pituitary to release specific hormones into the bloodstream
An example of a releasing factor is GnRH, which triggers the release of LH and FSH from the anterior pituitary
Posterior Lobe
The posterior lobe releases hormones produced by the hypothalamus itself (via neurosecretory cells)
These neurosecretory cells extend into the posterior lobe from the hypothalamus and release hormones into the blood
Pituitary hormones hence control many vital body processes, including:
Metabolism (e.g. TSH activates thyroxin)
Adult Development (e.g. LH / FSH trigger puberty)
Reproduction (e.g. LH / FSH control menstruation)
Growth (e.g. growth hormone promotes growth)
Equilibrium / Homeostasis (e.g. ADH and water balance)
Growth hormone
Also known as somatotropin is an anabolic peptide hormone that stimulates growth
It acts directly to reduce the formation of adipose cells (i.e. less nutrients stored as fat)
It acts indirectly via insulin growth factor (IGF) – produced by the liver – to increase muscle mass and bone size
Growth hormone abuse
Due to its role in promoting growth and regeneration, it is used by some athletes as a performance enhancer
The use of human growth hormone is banned in sports, with proven cases of doping strictly punished
Lactation
The production and secretion of milk by maternal mammary glands following birth
It is predominantly controlled and regulated by oxytocin and prolactin
Prolactin
Responsible for the development of the mammary glands and the production of milk
It is secreted by the anterior pituitary in response to the release of PRH (prolactin releasing hormone) from the hypothalamus
The effects of prolactin are inhibited by progesterone, which prevents milk production from occurring prior to birth
Oxytocin
Responsible for the release of milk from the mammary glands (milk ejection reflex)
It is produced in the hypothalamus and secreted by neurosecretory cells that extend into the posterior pituitary
Oxytocin release is triggered by stimulation of sensory receptors in the breast tissue by the suckling infant
This creates a positive feedback loop that will result in continuous oxytocin secretion until the infant stops feeding
D6 Transport of Respiratory Gases
Capillary endothelium cells
Alveolar air spaces are surrounded by a dense network of capillaries, which transport respiratory gases to and from the lungs
The capillaries are located close to the pneumocytes and are composed of a very thin, single-layer endothelium
Diagrammatic Representation of Lung Tissue
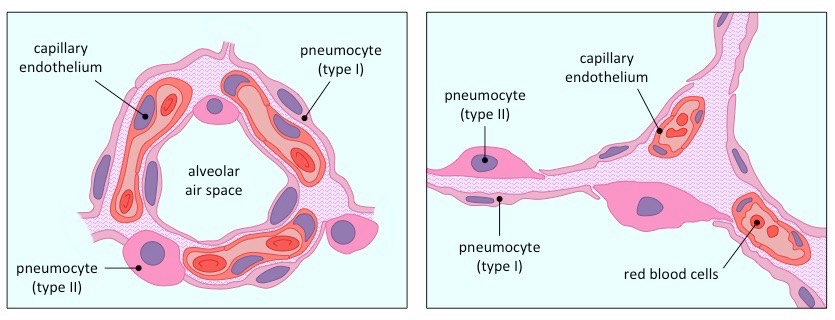
Light Micrograph of Lung Tissue

Electron Micrograph of Lung Tissue

Oxygen transport in the body
Oxygen is transported throughout the body in red blood cells, which contain an oxygen-binding protein called haemoglobin
Haemoglobin is composed of four polypeptide chains, each with an iron-containing heme group that reversibly binds oxygen
As such, each haemoglobin can reversibly bind up to four oxygen molecules (Hb + 4O2 = HbO8)
Cooperative binding
As each O2 molecule binds, it alters the conformation of haemoglobin, making subsequent binding easier (cooperative binding)
This means haemoglobin will have a higher affinity for O2 in oxygen-rich areas (like the lung), promoting oxygen loading
Conversely, haemoglobin will have a lower affinity for O2 in oxygen-starved areas (like muscles), promoting oxygen unloading
Oxygen dissociation curves
Show the relationship between oxygen levels (as partial pressure) and haemoglobin saturation
Because binding potential changes with each additional O2 molecule, the saturation of haemoglobin is not linear
Adult Haemoglobin
The oxygen dissociation curve for adult haemoglobin is sigmoidal (i.e. S-shaped) due to cooperative binding
There is a low saturation of haemoglobin when oxygen levels are low (haemoglobin releases O2 in hypoxic tissues)
There is a high saturation of haemoglobin when oxygen levels are high (haemoglobin binds O2 in oxygen-rich tissues)
Oxygen Dissociation Curve – Adult Haemoglobin

Foetal Haemoglobin
Foetal haemoglobin has a higher affinity for oxygen (dissociation curve is shifted to the left)
This is important as it means fetal haemoglobin will load oxygen when adult haemoglobin is unloading it (i.e. in the placenta)
Following birth, fetal haemoglobin is almost completely replaced by adult haemoglobin (~ 6 months post-natally)
Fetal haemoglobin production can be pharmacologically induced in adults to treat diseases such as sickle cell anaemia
Oxygen Dissociation Curve – Fetal Haemoglobin

Myoglobin
Myoglobin is an oxygen-binding molecule that is found in skeletal muscle tissue
It is made of a single polypeptide with only one heme group and hence is not capable of cooperative binding
Consequently, the oxygen dissociation curve for myoglobin is not sigmoidal (it is logarithmic)
Myoglobin has a higher affinity for oxygen than adult haemoglobin and becomes saturated at lower oxygen levels
Myoglobin will hold onto its oxygen supply until levels in the muscles are very low (e.g. during intense physical activity)
The delayed release of oxygen helps to slow the onset of anaerobic respiration and lactic acid formation during exercise
Oxygen Dissociation Curve – Myoglobin

Carbon dioxide transport
Carbon dioxide is transported between the lungs and the tissues by one of three mechanisms:
Some is bound to haemoglobin to form HbCO2 (carbon dioxide binds to the globin and so doesn’t compete with O2 binding)
A very small fraction gets dissolved in water and is carried in solution (~5% – carbon dioxide dissolves poorly in water)
The majority (~75%) diffuses into the erythrocyte and gets converted into carbonic acid
Transport as Carbonic Acid
When CO2 enters the erythrocyte, it combines with water to form carbonic acid
The carbonic acid (H2CO3) then dissociates to form H+ and bicarbonate (HCO3–)
Bicarbonate is pumped out of the cell in exchange with chloride ions (exchange ensures the erythrocyte remains uncharged)
The bicarbonate in the blood plasma combines with sodium to form sodium bicarbonate (NaHCO3), which travels to the lungs
The hydrogen ions within the erythrocyte make the environment less alkaline, causing haemoglobin to release its oxygen
The haemoglobin absorbs the H+ ions and acts as a buffer to maintain the intracellular pH
When the red blood cell reaches the lungs, bicarbonate is pumped back into the cell and the entire process is reversed
Carbon Dioxide Transport in the Bloodstream
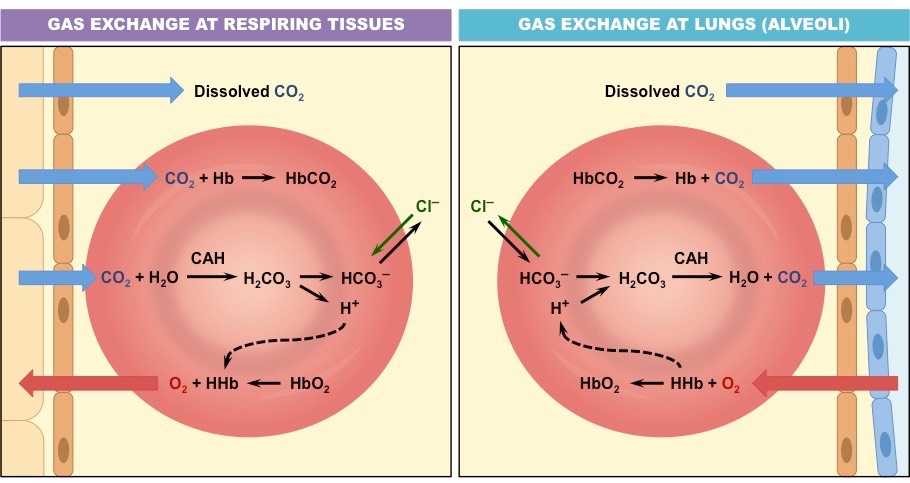
Chemoreceptors
Chemoreceptors are sensitive to changes in blood pH and can trigger body responses in order to maintain a balance
The lungs can regulate the amount of carbon dioxide in the bloodstream by changing the rate of ventilation
The kidneys can control the reabsorption of bicarbonate ions from the filtrate and clear any excess in the urine
Blood pH
The pH of blood is required to stay within a very narrow tolerance range (7.35 – 7.45) in order to avoid the onset of disease
This pH range is, in part, maintained by plasma proteins which act as buffers
Blood as a buffer solution
A buffering solution resists changes to pH by removing excess H+ ions (↑ acidity) or OH– ions (↑ alkalinity)
Amino acids are zwitterions – they may have both a positive and negative charge and hence can buffer changes in pH
The amine group may take on H+ ions while the carboxyl group may release H+ ions (which form water with OH– ions)
Oxyhaemoglobin dissociation curve
Demonstrates the saturation of haemoglobin by oxygen under normal conditions
pH changes alter the affinity of haemoglobin for oxygen and hence alters the uptake and release of O2 by haemoglobin
Bohr effect
Carbon dioxide lowers the pH of the blood (by forming carbonic acid), which causes haemoglobin to release its oxygen
This is known as the Bohr effect – a decrease in pH shifts the oxygen dissociation curve to the right
Cells with increased metabolism (i.e. respiring tissues) release greater amounts of carbon dioxide (product of cell respiration)
Hence haemoglobin is promoted to release its oxygen at the regions of greatest need (oxygen is an input of cell respiration)
The Bohr Shift

Respiratory control centre
The respiratory control centre in the medulla oblongata responds to stimuli from chemoreceptors in order to control ventilation
Central chemoreceptors in the medulla oblongata detect changes in CO2 levels (as changes in pH of cerebrospinal fluid)
Peripheral chemoreceptors in the carotid and aortic bodies also detect CO2 levels, as well as O2 levels and blood pH
Respiration during exercise
During exercise metabolism is increased, which results in a build up of carbon dioxide and a reduction in the supply of oxygen
These changes are detected by chemoreceptors and impulses are sent to the respiratory control centre in the brainstem
Signals are sent to the diaphragm and intercostal muscles to increase the rate of ventilation (this process is involuntary)
As the ventilation rate increases, CO2 levels in the blood will drop, restoring blood pH (also O2 levels will rise)
Long term effects of continual exercise may include an improved vital capacity
Partial pressure
The pressure exerted by a single type of gas when it is found within a mixture of gases
Determining partial pressure
The concentration of the gas within the mixture (e.g. oxygen forms roughly 21% of the atmosphere)
The total pressure of the mixture (e.g. atmospheric pressure)
Gas exchange at high altitudes
Air pressure is lower and hence there is a lower partial pressure of oxygen (less O2 because less air overall)
This makes it more difficult for haemoglobin to take up and transport oxygen (lower Hb % saturation)
Consequently, respiring tissue will receive less oxygen – leading to symptoms such as fatigue, headaches and rapid pulse
Adaptations of high altitudes
Red blood cell production will increase in order to maximise oxygen uptake and transport
Red blood cells will have a higher haemoglobin count with a higher affinity for oxygen
Vital capacity will increase to improve rate of gas exchange
Muscles will produce more myoglobin and have increased vascularisation to improve overall oxygen supply
Kidneys will begin to secrete alkaline urine (removal of excess bicarbonates improves buffering of blood pH)
People living permanently at high altitudes will have a greater lung surface area and larger chest sizes
High altitude training
Professional athletes will often incorporate high altitude training in order to adopt these benefits prior to competition
Athletes may commonly either train at high altitudes (live low – train high) or recover at high altitudes (live high – train low)
Emphysema
Emphysema is a lung condition whereby the walls of the alveoli lose their elasticity due to damage to the alveolar walls
The loss of elasticity results in the abnormal enlargement of the alveoli, leading to a lower total surface area for gas exchange
The degradation of the alveolar walls can cause holes to develop and alveoli to merge into huge air spaces (pulmonary bullae)
Causes of emphysema
The major cause of emphysema is smoking, as the chemical irritants in cigarette smoke damage the alveolar walls
The damage to lung tissue leads to the recruitment of phagocytes to the region, which produce an enzyme called elastase
This elastase, released as part of an inflammatory response, breaks down the elastic fibres in the alveolar wall
A small proportion of emphysema cases are due to a hereditary deficiency in this enzyme inhibitor due to a gene mutation
Treatments of emphysema
There is no current cure for emphysema, but treaments are available to relieve symptoms and delay disease progression
Bronchodilators are commonly used to relax the bronchiolar muscles and improve airflow
Corticosteroids can reduce the inflammatory response that breaks down the elastic fibres in the alveolar wall
Elastase activity can be blocked by an enzyme inhibitor (α-1-antitrypsin), provided elastase concentrations are not too high
Oxygen supplementation will be required in the later stages of the disease to ensure adequate oxygen intake
In certain cases, surgery and alternative medicines have helped to decrease the severity of symptoms