L7: Spinodal decomposition and ordering
Learning Objectives:
Part 1: spontaneous separation
• Be able to explain using diagrams under which conditions a mixture can undergo spontaneous (spinodal) decomposition
ΔHmix > 0
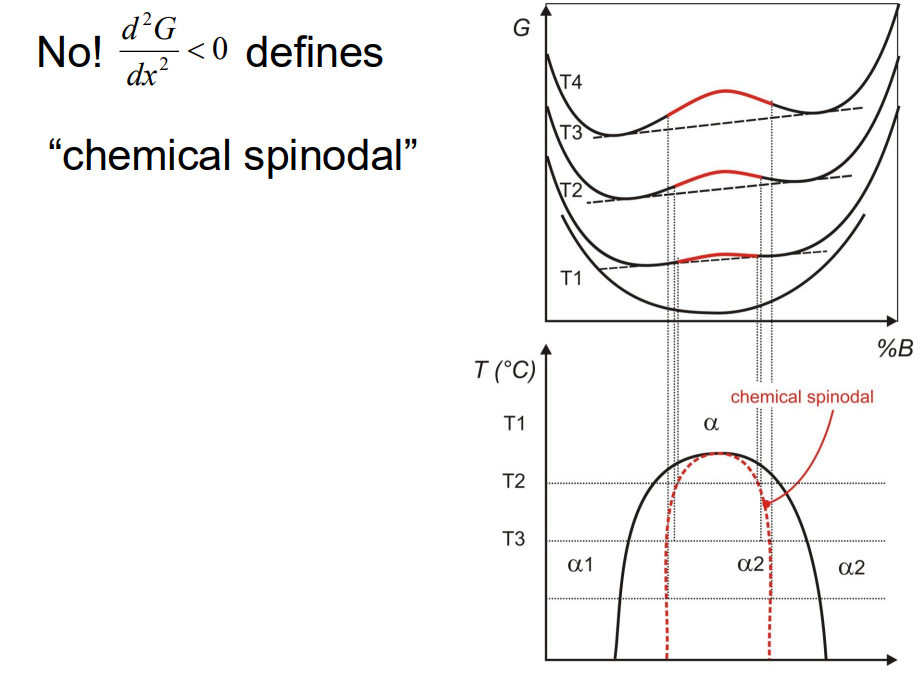
d²G/dX² < 0
Remaining questions:
- What is driving force?
- When does it end?
- What determines wavelength?
• Discuss the 2 energy contributions that oppose this
(1) Chemical
(2) gradient
(3) size mis-match
Coherent spinodal defines the where (1) overcomes (3) – Wavelength depends on undercooling
Part 2: ordering
• Argue why ordering occurs, why at low temperatures, and how it affects properties.
A-B bond enthalpy exceeds entropy gain of random mixture.
ΔHmix << 0
• Be aware of how quickly ordering reactions can occur.
Either nucleation and growth (most typical) or spinodal-like
Part 1: Spontaneous Separation (Spinodal Decomposition)
1. Conditions for Spontaneous Decomposition:
- Spinodal decomposition occurs when a single phase spontaneously separates into two phases without the need for nucleation. This happens when the free energy curve is convex, i.e.
d²G/dX² < 0
- The process is driven by uphill diffusion, where atoms move against the concentration gradient to regions of higher concentration of their own type (e.g., B atoms move to regions with more B atoms, and A atoms move to regions with more A atoms).
- This behaviour is only possible at low temperatures where the ΔHmix > 0, meaning atoms prefer to bond with like atoms rather than unlike atoms.
2. Energy Contributions Opposing Spinodal Decomposition:
- Gradient Energy: As the composition varies spatially, the gradient in concentration leads to an energy penalty due to the increased number of unlike bonds. This is termed gradient energy, which scales with the square of the concentration gradient
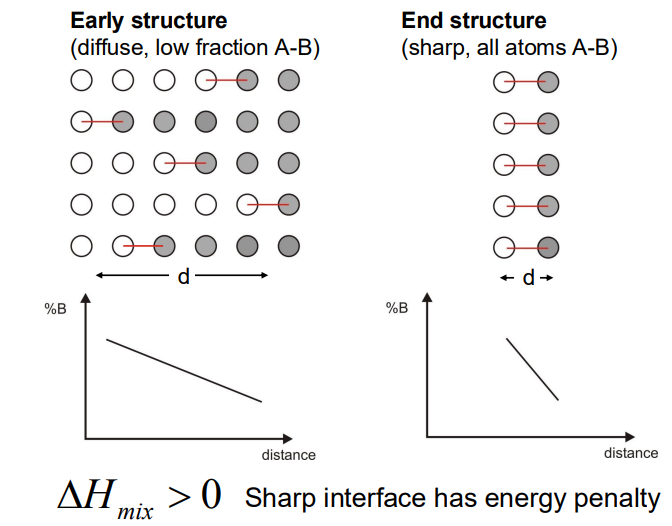
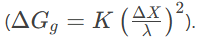
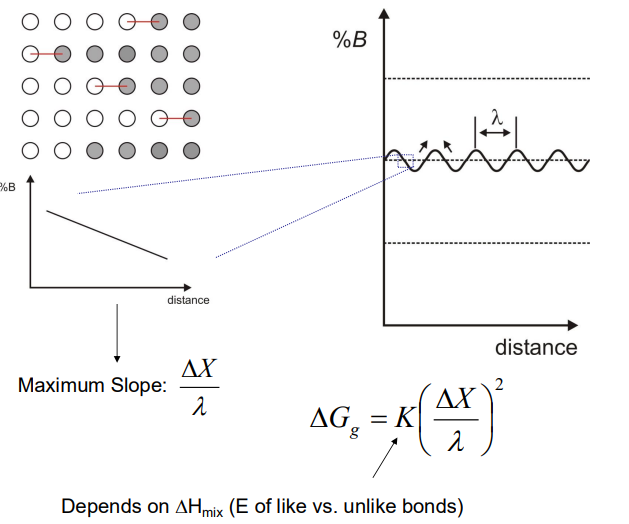
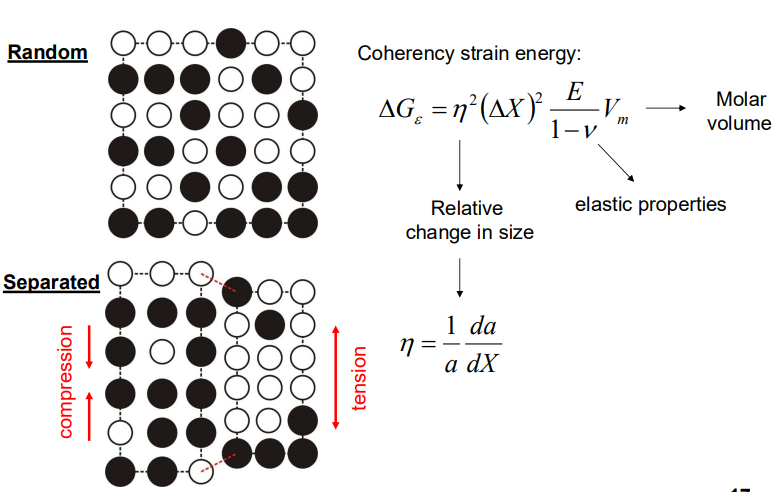
- Coherency Strain Energy: If the atoms A and B have different sizes, the composition variation leads to coherency strains, which also oppose the decomposition. The strain energy depends on the elastic properties of the material and the relative change in lattice parameter with composition
Chemical energy:
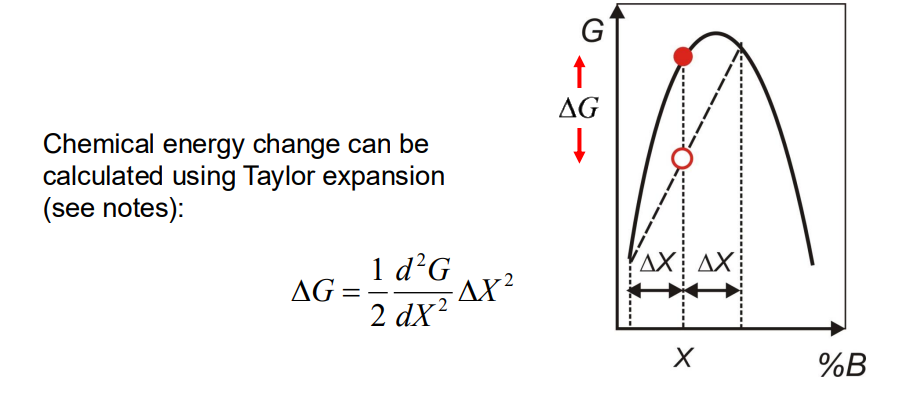
. Coherent Spinodal:
- The coherent spinodal is the practical limit for spinodal decomposition, taking into account both the chemical driving force and the coherency strain energy. It is defined by the condition:
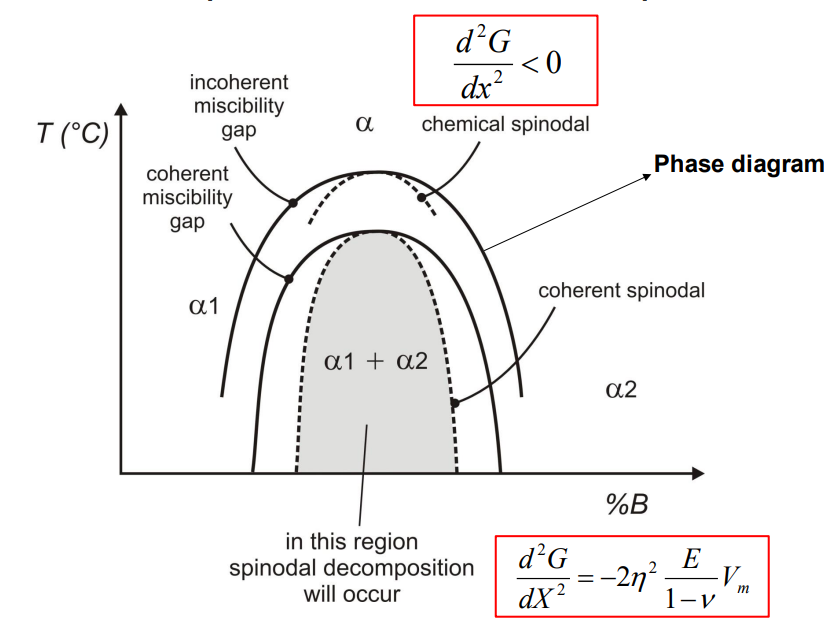
- Spinodal decomposition is only possible in the region where the chemical driving force overcomes the strain energy, which typically requires undercooling below the chemical spinodal.
Miscibility gap- mixture prefers to separate into 2 phases
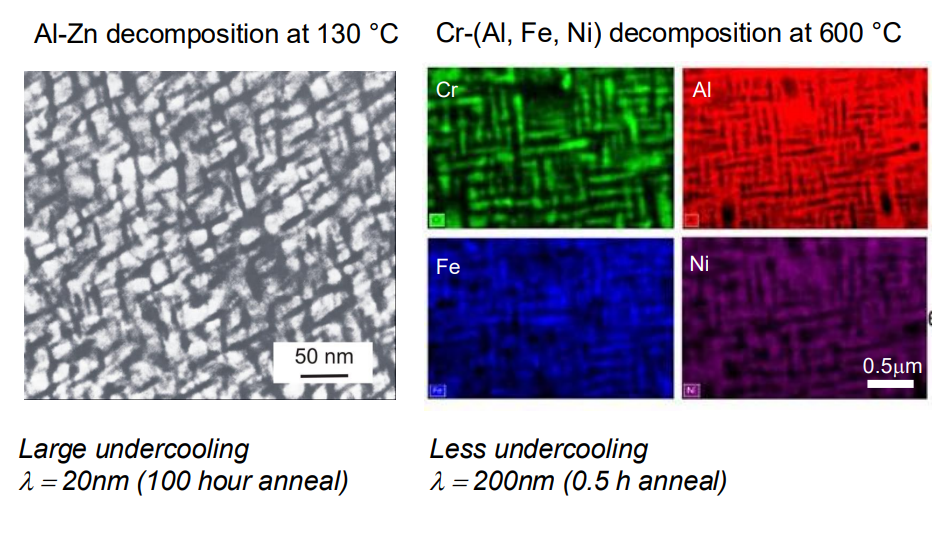
4. Wavelength of Decomposition:
- The wavelength λ of the composition modulation is determined by the balance between the chemical driving force, gradient energy, and strain energy. Larger undercooling leads to smaller wavelengths, allowing for faster transformation.
Part 2: Ordering
1. Why Ordering Occurs:
- Ordering occurs when the enthalpy of mixing (ΔHmix) is strongly negative, meaning atoms prefer to bond with unlike atoms rather than like atoms. This leads to the formation of ordered structures where atoms arrange themselves in a specific pattern to maximize the number of unlike bonds.
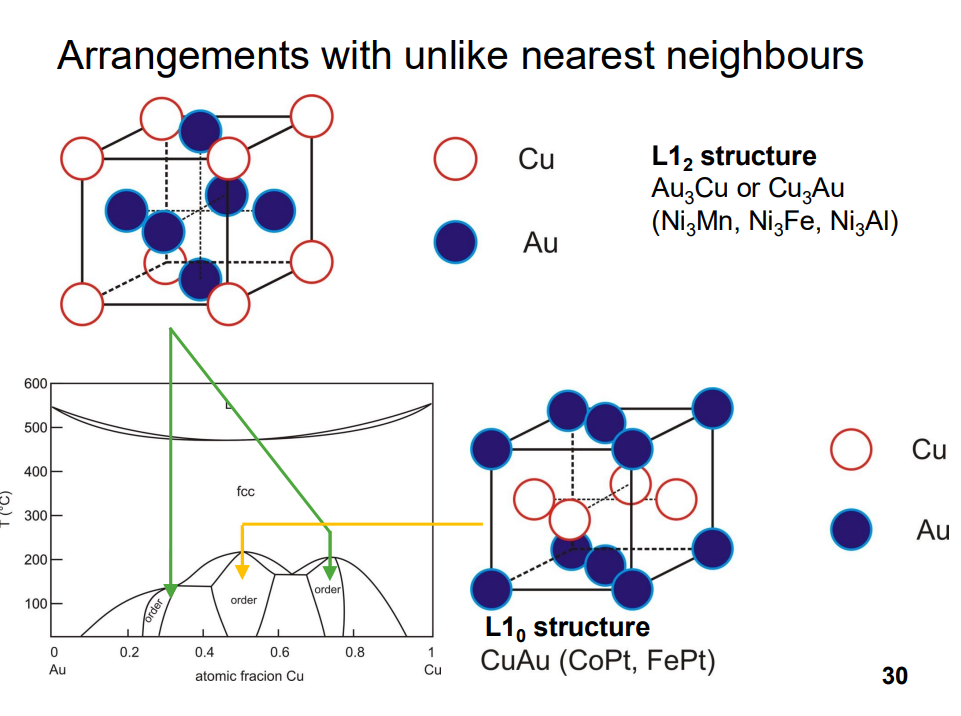
- Ordering is favoured at low temperatures because the entropic contribution to the free energy (which favours randomness) is less significant at lower temperatures.
2. Effect on Properties:
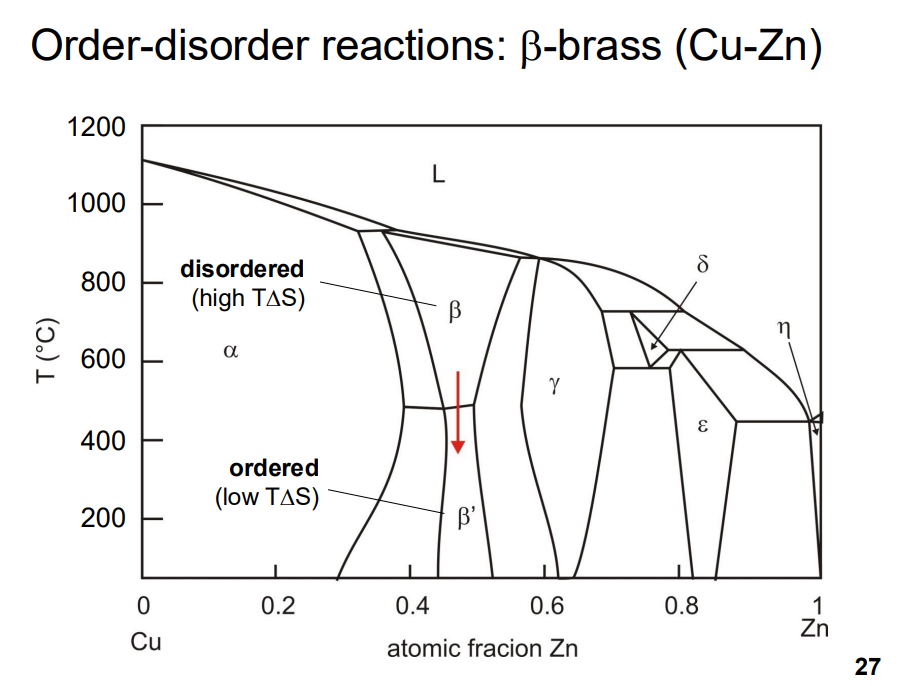
- Ordered structures can significantly affect material properties, such as hardness and strength. For example, in Cu-Zn alloys (β-brass), the ordered structure is harder because dislocations must disrupt the ordered arrangement, increasing the energy required for deformation.
3. Speed of Ordering Reactions:
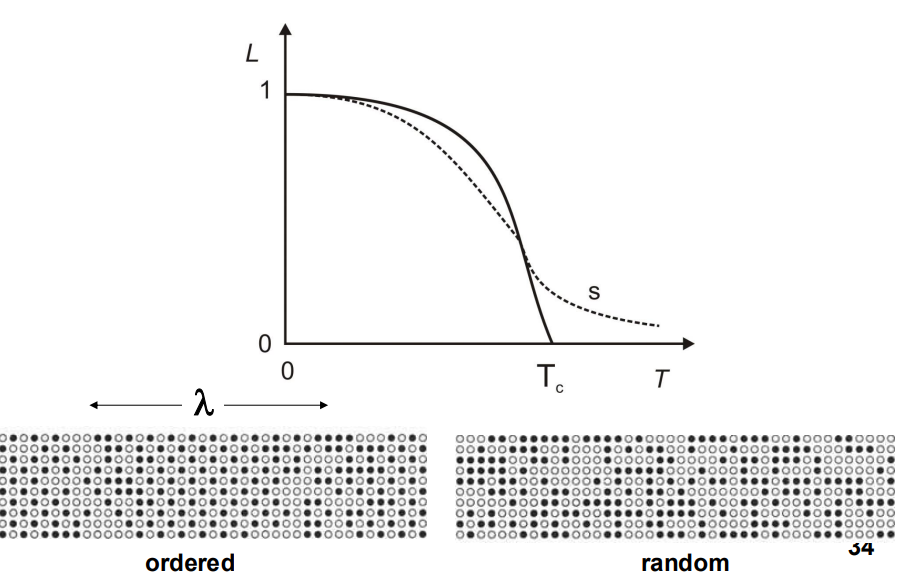
- Ordering reactions can occur either gradually (similar to spinodal decomposition)
or suddenly (similar to nucleation and growth).
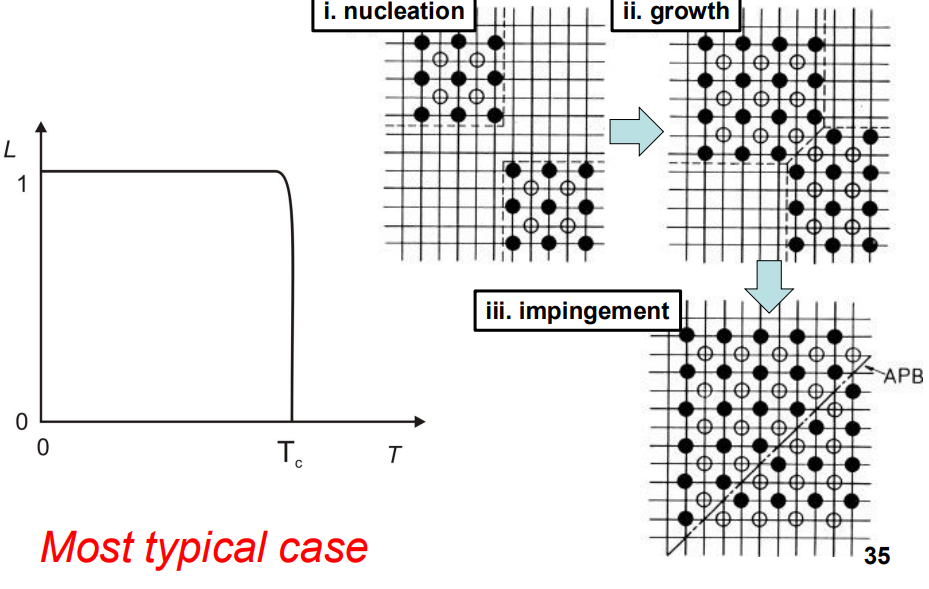
In systems like CuZn, ordering occurs gradually as the temperature decreases, while in systems like Cu3Au, ordering occurs suddenly at a critical temperature.
- The long-range order parameter L is used to quantify the degree of ordering,
where L=1 indicates fully ordered
and L=0 indicates completely random.


4. Nucleation and Growth in Ordering:
- In some systems, ordering occurs via nucleation and growth of ordered regions. The energy barrier for nucleation is relatively small because there is no change in composition or crystal structure, and the interfaces are coherent (without strain). This can lead to the formation of anti-phase boundaries (APBs) when ordered regions impinge on each other.
SUMMARY
- Spinodal decomposition is a spontaneous phase separation driven by a convex free energy curve and uphill diffusion, opposed by gradient energy and coherency strain energy.
- Ordering occurs when the enthalpy of mixing is strongly negative, leading to the formation of ordered structures at low temperatures, which can significantly affect material properties.
- Ordering reactions can occur gradually or suddenly, depending on the system, and are often quantified using the long-range order parameter L