BONDING, STRUCTURE AND STATES OF MATTER
Ionic bonding
An ion is a charged particle, which is formed when elements gain or lose electrons to gain a full outer shell
Elements are more likely to form ions if there are not many electrons lost or gained
Takes less energy
Groups 1,2,6 and 7 are most likely
Groups 3,4 and 5 are rarely seen as ions
Happens between a metal and non-metal
For example, NaNa+ + e- and Cl + e- Cl-
Electrons are transferred
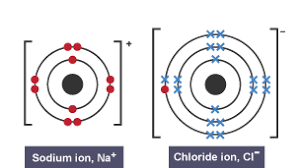
NaCl becomes an ionic compound and are attracted by strong electrostatic forces (have opposite charges)
Ionic compounds
Metals and non-metals form ionic compounds with strong electrostatic forces and ionic bonds
Usually group in large numbers
Regular lattice structures (3D)
Each ion is attracted to all those around it
Properties - very high melting points as lots of energy is required to overcome strong ionic bonds
can conduct electricity when molten or aqueous as charged particles are free to move
NEED TO KNOW - Hydroxide = OH-
Sulphate = SO4-
Nitrate = NO3-
Carbonate = CO3-
Ammonium = NH4+
Covalent bonding
The sharing of electrons in the outer shell between non-metals to gain a full outer shell
Cl - Cl
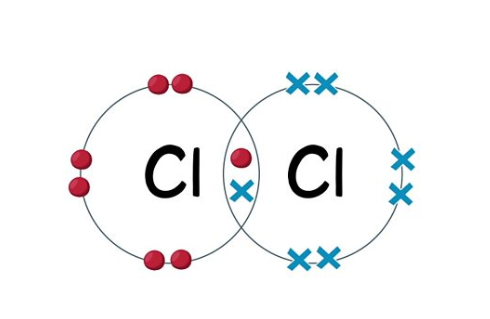
They can become:
Simple molecular substances - Small molecules with strong covalent bonds between atoms and weak intermolecular forces between molecules
Like water, ammonia, chlorine or methane
Polymers - Long chains of repeating units (monomers)
Giant covalent structures - diamond, graphite + silica dioxide
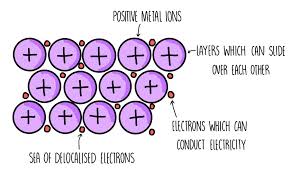
Metallic bonding
Happens between metal atoms
Solid metals are in a giant structure arranged in a regular pattern with delocalised electrons
They give up their outer shell electrons and share them with the other metals
The atoms all become + ions
The lost electrons can freely move so are delocalised
There are strong forces of attraction between the ions and electrons, which hold everything together in a regular structure
Metals are strong, so have high melting and boiling points
They are good conductors of heat and electricity ⚡
Their delocalised electrons can feely move and carry electrical current through the structure
Metals are malleable - their regular structure allows layers to slide
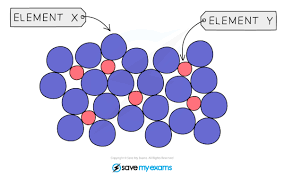
Alloys are when 2 or more different metals or a metal and non-metal form metallic bonds, with different sized atoms
This disrupts the regular structure, so layers can no longer slide
Alloys are stronger than pure metals
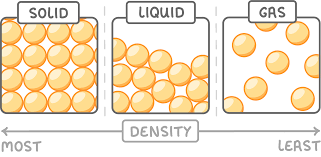
States of matter
Solids - Strong forces of attraction (holds them close together)
Fixed, regular position
Definite shape and volume - can vibrate
Liquids - Weak forces of attraction (particles are free to move and flow)
Can flow
Compact and definite volume, but not shape
Move to fit a container
Gas - Very very weak forces of attraction
No definite shape of volume
Fill a container
Particles are free to move
Constantly moving with random motion - move in a straight line and are deflected when hit
State symbols
Solid - s
Liquid - l
Gas - g
Aqueous - aq
Properties of ionic compounds
High melting and boiling point
Lots of energy is required to overcome strong electrostatic forces of attraction, and there are lots of forces
Can conduct when aqueous or molten
Charged particles are free to flow through the structure
Properties of small molecules
Low melting and boiling points
Weak intermolecular forces between molecules, that need little energy to break
COVALENT BONDS ARE NOT BROKEN
Generally liquids or gases at room temperature
Do not conduct
Properties of metals and alloys
Metals are soft and malleable, shiny, good conductors of heat and electricity and have high melting and boiling points
Alloys are hard (no layers), have high melting and boiling points and are good conductors
Giant covalent structures
Simple molecular substances have low melting points, strong bonds between atoms and don’t conduct
Giant covalent structures have huge numbers of non-metal atoms
Arranged in a regular repeating lattices
Have high melting and boiling points as there are a lot of covalent bonds
Very strong - lots of bonds
Generally don’t conduct (apart from graphite and graphene)
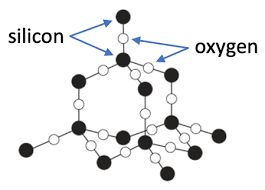
Silica dioxide is made of silicon and oxygen in a ration of 1:2
Makes up sand 🏖
Diamond and graphite
Allotropes of carbon
Diamond is a giant covalent structure💎
Is very strong
Each carbon is bonded to 4 other carbons (max amount)
It doesn’t conduct as there are no delocalised electrons
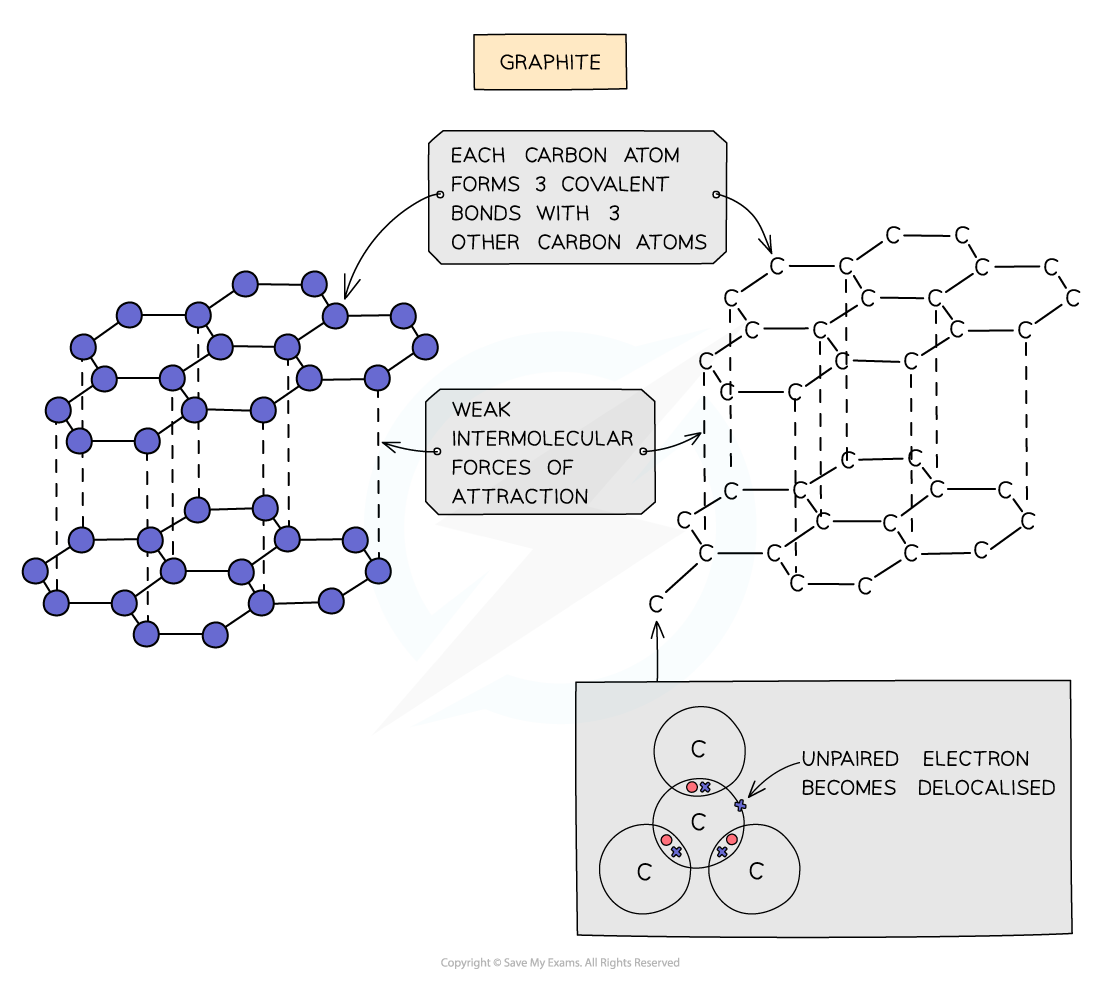
Graphite is a giant covalent structure
Is very strong
Each carbon makes 3 out of 4 covalent bonds possible
Is arranged in layers with weak intermolecular forces between them
This allows the layers to slide over one another and makes it soft
Has a high melting and boiling point
Can conduct electricity - only ¾ bonds are made, so there are left over electrons
Become delocalised (one per carbon atom) and are free to move through the structure and carry charge
Graphene is a single layer of graphite
Graphene and fullerenes
Are allotropes of carbon
Graphene is a single layer of graphite and can conduct electricity as there are delocalise electrons
Useful in electronics (conducts and is small)
Fullerenes are tubes and spheres made out of a single sheet of graphite
Spheres can be used to surround molecules (like drugs) and used to deliver to specific areas of the body
They have a large surface area : volume ratio, so make good industrial catalysts
Tubes can be used in nanotechnology as conductors, to strengthen tennis rackets (adds strength without weight as high length : diameter)
Buckminster fullerene is a hollow sphere that is made of 60 carbon atoms and is used for drug delivery
Using tiny structures is called nanotechnology
Medicine, fashion, batteries and food
Nanoparticles
Nanoparticles are really really really tiny particles - 1nm - 100nm (0.00000001m)
Nanoscience is produces new nanoparticle materials
They have a large surface area : volume ratio
Good for catalysts as surface area increases its efficiency
Nanomedicine uses fullerenes to deliver drugs around the body
Electrical circuits use them to make tiny computer chips as some can conduct
Silver nanoparticles have antibacterial properties so can be infused into wound dressings and masks
Issues with nanoparticles
They are relatively new so we are not aware of all risks (long term)
For example, sun cream with nanoparticles allows for better skin coverage but we are unaware if it can enter our body through the skin and potential damage cells
They are also possibly damaging to the environment
Sizes of particles and their properties
Atoms and small molecules - 0.1 nm
Nanoparticles - 1 to 100nm
Fine particles - 100 to 2500 nm
Coarse particles - 2500 to 10000 nm
The smaller the particle, the higher the surface area : volume ratio, so increased reactivity
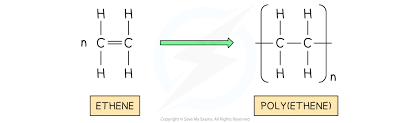
Polymers
Polymers have very large molecules and their atoms are joined by strong covalent bonds in long chains
Solid at room temperature
Higher boiling points than strong intermolecular forces and lots of bonds to overcome
DONE!!!