Ch. 4 Organizaciones de Electrones dentro de un Átomo
Section 1 - The Development of a New Atomic Model
Properties of Light
The Wave Description of Light:
Electromagnetic radiation - A form of energy that exhibits wavelike behavior as it travels through space.
Electromagnetic spectrum - Formed by all the forms of electromagnetic radiation together.
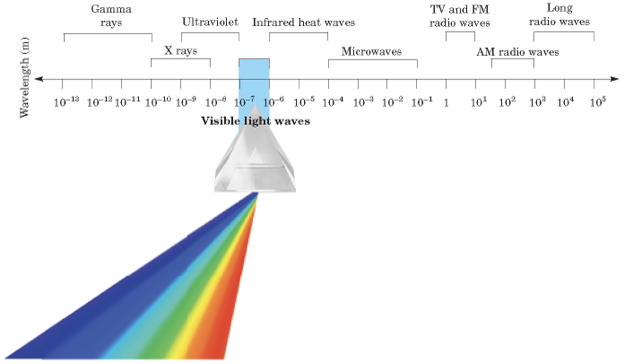 Low frequency: Gamma rays, x-rays, ultraviolet, infrared, and microwaves.
Low frequency: Gamma rays, x-rays, ultraviolet, infrared, and microwaves.
High frequency: TV and FM, AM radio waves, and long radio waves.
The more purple it is, the more low frequency it is, the more red it is, the higher it is.
White: The combination of all colors.
Wavelength - The distance between corresponding points on adjacent waves. (m)

Frequency - Defined as the number of waves that pass a given point at a specific time, usually one second. (s^-1)
 Frequency and wavelength are related:
Frequency and wavelength are related:
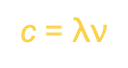
The higher it is, the more waves crash.
C is the speed of light (m/s)
C = 3×10^8 m/s
The Photoelectric Effect
Photoelectric effect - Refers to the emission of electrons from a metal when light shines on the metal.
The Particle Description of Light:
A quantum of energy is the minimum quantity of energy that can be lost or gained by an atom.
German physicist Max Planck proposed the relationship between a quantum of energy and the frequency of radiation:
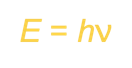
E is the energy in joules of a quantum of radiation
h = 6.626 × 10^-34 j/s
A photon is a particle of electromagnetic radiation having zero mass and carrying a quantum of energy.
The Hydrogen-Atom Line-Emission Spectrum
Ground State - The lowest energy state of an atom.
Excited State - Higher potential energy of atoms rather than it had in its ground state.
Investigators passed electric current through a vacuum tube containing hydrogen gas at low pressure, they observed the emission of a characteristic pinkish glow. When a narrow beam of the emitted light was shined through a prism, it was separated into four specific colors of the visible spectrum: Red, green, blue, indigo.
Line-emission spectrum: The four bands of light. (Hydrogen’s)
Every element in the periodic table has a specific emission of light.
Niels Bohr proposed a hydrogen-atom model that linked the atom’s electron to photon emission.
Orbits - Where the electron can circle the nucleus.
The further away the electrons are, the greater the energy needed.
Emission - The process when an electron falls to a lower energy level, a photon is emitted.
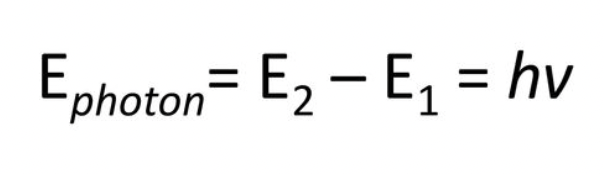
^ Final - initial
Absorption - Process where energy must be added to an atom in order to move an electron from a lower energy level to a higher energy level.
Section 2 The Quantum Model of the Atom
Electrons as Waves
Louis de Broglie - suggested that electrons should be considered waves confined to the space around an atomic nucleus.
It followed that the electron waves could exist only at specific frequencies.
According to E = hv, these frequencies corresponded to specific energies- the quantized energies of Bohr’s orbits.
Electrons, like light waves, can be bent, or diffracted.
Diffraction - The bending of a wave as it passes by the edge of an object or through a small opening.
Electro beams, like waves, can interfere with each other.
Interference - occurs when waves overlap.
The Heisenberg Uncertainty Principle
Werner Heisenberg - Proposed that any attempt to locate a specific electron with a photon knocks the electron off its course.
Heisenberg uncertainty principle - States that it is impossible to determine simultaneously both the position and velocity of an electron or any other particle.
The Scröndinger Wave Equation
Erwin Schröndinger - (1926) Developed an equation that treated electrons in atoms as waves.
Quantum theory - Describes mathematically the wave properties of electrons and other very small particles. Formed with the Schröndinger Wave Equation and the Heisenberg Uncertainty Principle.
Electrons do not travel around the nucleus in neat orbits;
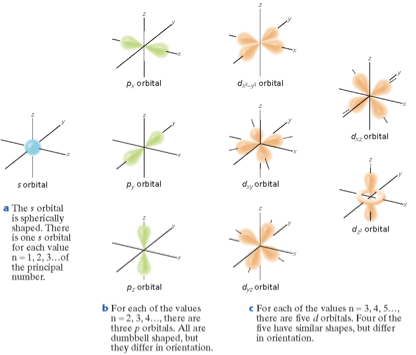
Orbitals - Where electrons exist. It is a three-dimensional region around the nucleus that indicates the probable location of an electron.
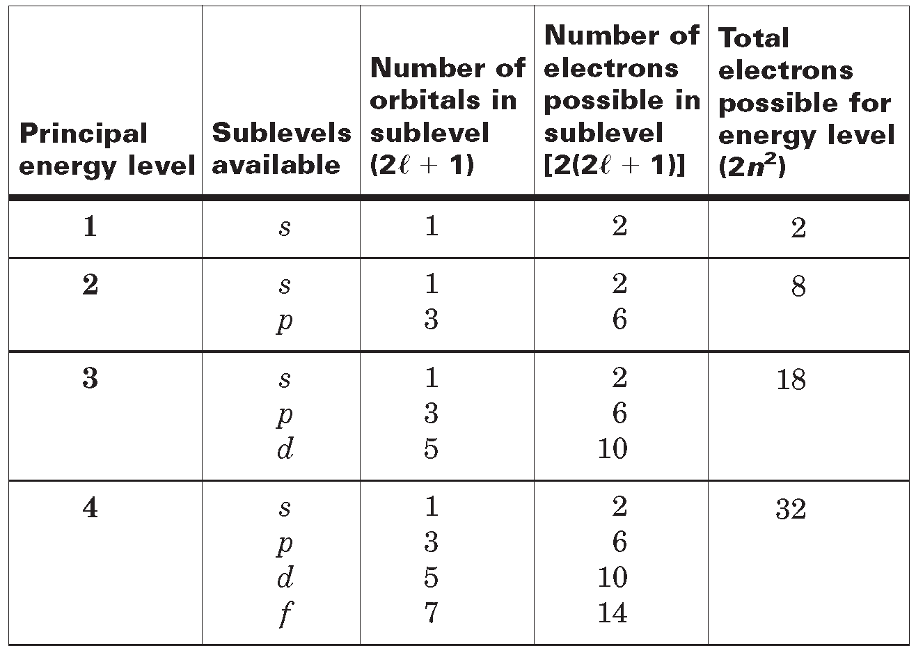
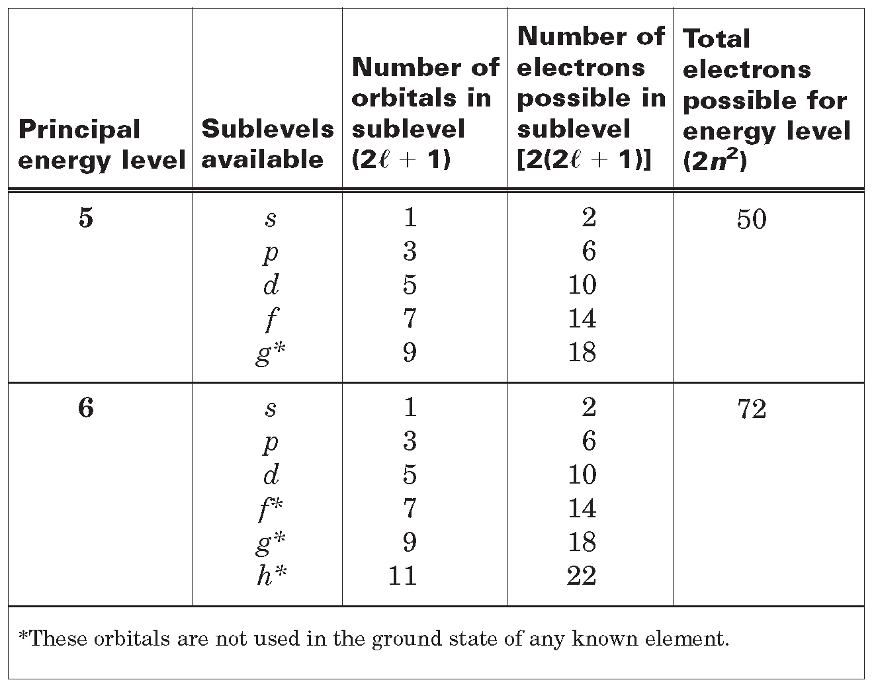
Atomic Orbitals and Quantum Numbers
Quantum numbers - Specify the properties of atomic orbitals and the properties of electrons in orbitals.
Principal quantum number - (n), indicates the main energy level occupied by the electron.
Angular momentum quantum number - (l), indicates the shape of the orbital.
Magnetic quantum number - (m), indicates the orientation of an orbital.
Spin quantum number - Has only two possible values-(+1/2, -1/2)- which indicate the two fundamental spin states of an electron in an orbital.
Section 3 Electron Configurations
Electron Configurations
Electron configuration - The arrangement of electrons in an atom.
Ground-state electron configuration - The lowest-energy arrangement of the electrons for each element.
Rules Governing Electron Configurations
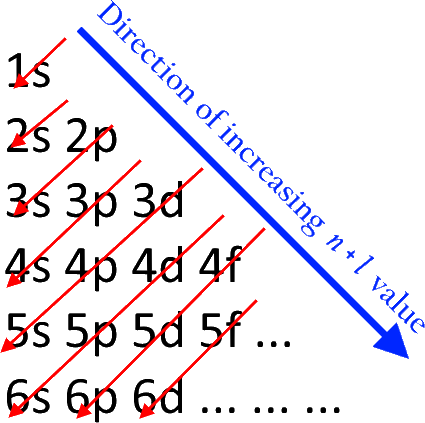
Aufbau principle - An electron occupies the lowest-energy orbital that can receive it.
Pauli exclusion principle - No two electrons in the same atom can have the same set of four quantum numbers.
(In each sublevel there is a maximum of 2 electrons. They are supposed to have an opposite turn: one positive and the other one negative)
Hund’s rule - Orbitals of equal energy are each occupied by one electron before any orbital is occupied by a second electron and all electrons in singly occupied orbitals must have the same spin state.
(Firstly positive turns are placed, then negative turns).
Moeller diagram:
Kernel’s rule: Condensed configuration.
(Simplifies the electric configuration).
Representing Electron Configurations
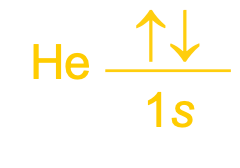
Orbital Notation:
An occupied orbital is represented by a line, with the orbital’s name written underneath the line.
One electron in an orbital:

Two electrons:
![]()
The lines are labeled with the principal quantum number and sublevel letter.
Electron-Configuration Notation
Highest-occupied energy level - The electron-containing main energy level with the highest principal quantum number.
Inner-shell electrons - These are the electrons that are not in the highest-occupied energy level.
Noble-Gas Notation:
Group 18 - Noble gases.
Noble-gas configuration - Refers to an outer main energy level occupied, in most cases, by eight electrons.