Citric Acid Cycle and Glycolysis
Citric Acid Cycle Overview
Chapter Reference: Lehninger, Chapter 16
Key Components:
Oxaloacetate
Acetyl-CoA
Citric acid (Citrate)
Isocitrate
α-Ketoglutarate
Succinate
Fumarate
Malate
Succinate
Succinyl-CoA
Fates of Glucose
Glycolysis and its Limitations:
Captures only a small amount of energy from glucose.
Need for additional energy extraction methods via cellular respiration.
Comparison of Energy Yield:
Conditions:
Oxygen Present: More energy (12 ATP)
No Oxygen (Anaerobic): Less energy extracted from glucose yielding only 2 ATP (lactic acid formation).
Cellular Respiration Process (Chapter 19)
Definition:
Cellular respiration is the process by which cells consume oxygen and produce carbon dioxide.
Energy Yield:
Cellular respiration provides significantly more ATP from glucose than glycolysis alone.
Substrate Utilization:
In addition to glucose, cellular respiration captures energy stored in lipids and amino acids.
Three Major Steps:
Acetyl-CoA production from pyruvate.
Acetyl-CoA oxidation via the Citric Acid Cycle.
Electron transfer and oxidative phosphorylation.
Citric Acid Cycle Specifics
Location:
Occurs in the mitochondria of eukaryotic cells.
Key Enzyme:
Succinate dehydrogenase is located in the inner mitochondrial membrane.
Genetic Considerations:
Proper functioning depends on genetic integrity.
For CAC to work we should have oxygen too
Step 1: Pyruvate to Acetyl-CoA
Net Reaction: Completely irreversible
Oxidative decarboxylation of pyruvate, resulting in CO2 loss and Acetyl-CoA formation.
In this step removal of CO2 is done to and Acetyl-CoA is formed by oxidation reduction reaction.
Cofactors Required:
5 essential cofactors required for the three-enzyme complex involved in the reaction.
1. TPP (thiamine pyrophosphate) - a coenzyme derived from vitamin B1 that Holds the Acetyl Group after CO2 is taken off
2. FAD (riboflavin) Vitamin B2, does the oxidation reduction reaction
3. Coenzyme A (CoA), THIS IS VITAMIN B5
4. NAD (from niacin) B3, redox rxn
5. Lipoate - this holds the acetyl group while it gets oxidized
Energy Aspect:
The coupling mechanism involves thioester formation providing high-energy storage.
Regulators:
Two critical regulators that influence Acetyl-CoA’s activity (CoA and B vitamins).
Step 1: Pyruvate Dehydrogenase Complex
Advantages:
Close proximity of catalytic sites ensures efficient substrate transfer and minimizes side reactions.
Regulation of one subunit can influence the overall complex activity due to spatial arrangement.
Regulatory Proteins : Protein kinase and Phosphoprotein phosphatase
Detailed Mechanism of Step 1
Decarboxylation:
CO2 removed, leaving a 2-carbon molecule bound to TPP (Thiamine). Takig off CO2 leaving acetyl group
Oxidation and Transfer:
The 2C molecule is oxidized and transferred to lipoic acid forming an acetyl group. Lipoic acid holds the acetyl group while it gets oxidized.
Acetyl Group Attachment:
Acetyl group is then attached to CoA, leaving lipoic acid reduced.
Reoxidation of Lipoic Acid:
FAD reduces lipoic acid back to its oxidized form (FAD to FADH2). so everything can run again
NAD+ Reduction:
NAD+ oxidizes FADH2 back to FAD, creating NADH in the process.
If there is NO Glycolysis there will be NO CAC
Acetyl-CoA the S-CoA is removed (releasing energy) added to oxaloacetate to form Citrate
**Citrate can INHIBIT CAC AND CAN INHIBIT GLYCOLYSIS.
**OXALOACETATE REGULATES CAC, IF NONE THERE WILL BE NO CAC
Step 2: Oxidation of Acetyl-CoA in the Citric Acid Cycle
Dehydrogenation:
Oxidation of -OH, initiating the conversion of substrates through Claisen condensation.
Generates an intermediate carbonyl that facilitates further reaction steps.
Citrate Formation:
Reaction of acetyl-CoA with oxaloacetate forming citrate.
Enzymatic action ensures stereospecificity in product formation.
Dehydrate citrate and rehydrate it forming Isocitrate( only enxyme that binds to alpha ketoglutarate)
Step 3: Formation of α-Ketoglutarate and CO2
Exergonic Reaction:
Isocitrate is oxidized and subsequently decarboxylated to form α-ketoglutarate.
NADH FORMED
CO2 RELEASED
Regulation Mechanisms:
Regulated by ATP concentration and NADH as product inhibitors.
Energy Carrier Role:
NAD(P)H functions as an electron carrier aiding ATP synthesis later.
Step 4: Formation of Succinyl-CoA and CO2
Similarities to Step 1:
Utilizes mechanisms akin to pyruvate dehydrogenase for oxidative decarboxylation.
Energy Storage:
CoA-S is added to alpha-ketogluterate to produce Succinyl-CoA as high-energy storage molecule, which upon conversion leads to ATP synthesis.
NADH formed
CO2 released
Energetic Output:
This cycle releases NADH which aids in ATP production ultimately yielding about 2.5 ATP.
Step 5: Succinate Formation
Substrate-Level Phosphorylation:
ATP IS PRODUCED
ATP formation occurs directly from the reaction for the first time in the CAC.
Thioester is taken off, energy is release, ADP+Pi making ATP.
Energy Conversion:
The thioester bond from Succinyl-CoA is cleaved to drive ATP production.
Succinyl-CoA forms Succinate
Step 6: Oxidation to Fumarate
This is where CAC and ETC are physically connected!
Dehydrogenation:
Conversion of alkane (Succinate) to alkene (Fumarate) occurs by succinate dehydrogenase, producing FADH2.(fixed electron carrier)
ETC Connection:
This step links the CAC to the electron transport chain.
Step 7: Formation of Malate
Stereospecific Addition:
Only L-malate is formed through the addition of water across a double bond.
Step 8: Regeneration of Oxaloacetate
End Reaction:
The last step regenerates oxaloacetate, which is essential to keep the cycle iterating.
L-Malate is oxidized to Oxaloacetate
Energetics:
This process is endergonic overall and relies on the catalytic action of citrate synthase.
General Implications and Questions
CO2 Release Points:
Key points of decarboxylation events where CO2 is released. Steps 3 And 4
Energy Release Points:
Areas where energy is transformed through exergonic reactions.
NADH released at Step 3,4,8
ATP released at Step 5
FADH2 released at Step 6
Coupling Mechanisms:
How exergonic processes can drive endergonic reactions in biochemistry. Through coupling reactions, energy from exergonic processes can be harnessed to power the formation of high-energy molecules, such as ATP
Glycolysis Outcomes
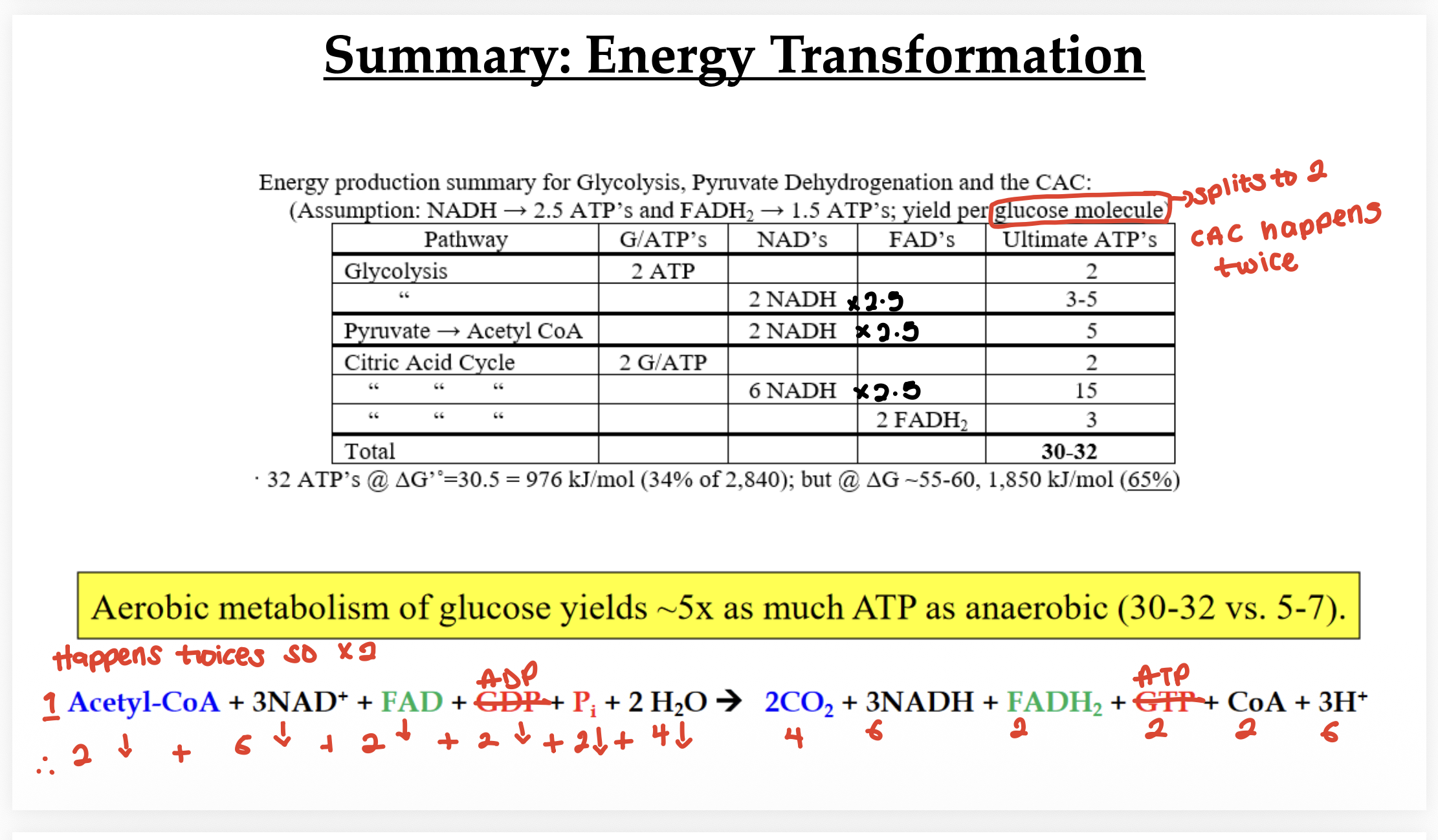
Summary of energy outputs from glycolysis and the CAC:
2 NADH and 2 ATP processed from glycolysis.
Total ATP yield combining glycolysis, CAC, and oxidative phosphorylation is approximately 30-32 ATP depending on shuttle mechanisms.
8 NADH, 2 ATP, 2 FADH2.
Glucose molecules split into two this means CAC happens twice so every cycle through the citric acid cycle yields an additional 2 NADH, 1 ATP, and 1 FADH2 per molecule of glucose.
Role of CAC Anabolism
Lipid synthase
hemoglobin
gluconeogenesis
amino acid synthase
Central Role of CAC
This is an AMPHIBOLIC pathway , meaning it plays a role in both catabolism and anabolism, facilitating the conversion of substrates into energy while also providing intermediates for biosynthetic processes.
ANAPLEROTIC: the immediate replenishing reaction.
Replenishes MALATE AND OR OXALOACETATE 4 ways to replenish OA
OAA from Pyruvate, PEP
Malate from Pyruvate
Summary of Energy Transformation
Regulation of the Citric Acid Cycle
Regulatory Points:
Highly exergonic steps serve as regulatory points.
Regulation Mechanisms:
Categories include substrate availability(Feed), product feedback inhibition(feed) allosteric factors (ALL), and covalent modification.(feed)
Key Molecular Players:
Involvement of AMP, NADH, ATP, Citrate, and calcium ions in regulation.
Higher ADP/ATP makes CAC go, Higher ATP/ADP stops CAC
Lower energy states lead to an increase in the activity of the citric acid cycle, as the cell seeks to generate more ATP to meet energy demands. Conversely, an abundance of ATP signals that energy needs are met, thereby inhibiting further activity of the cycle.
Regulation of Feed-in Steps by Covalent Modification (Pyruvate Dehydrogenase)
This enzyme plays a crucial role in linking glycolysis to the citric acid cycle, and its activity is modulated by phosphorylation. When pyruvate dehydrogenase is phosphorylated, it becomes inactive, which reduces the conversion of pyruvate into acetyl-CoA. Conversely, dephosphorylation activates the enzyme, promoting the entry of pyruvate into the cycle, thereby enhancing the production of ATP under low energy conditions. Additionally, the regulation of pyruvate dehydrogenase is influenced by various metabolites, such as NADH and acetyl-CoA, which act as feedback inhibitors when present in high concentrations, further controlling the flow of carbon into the citric acid cycle.
3 THREE ENZYMES
5 COFACTORS
2 REGULATORY ENZYMES (KINASE AND PHOSPHATASE)
Citric Acid Cycle (CAC) Regulation Summary
Overview of allosteric regulators, substrate inhibition, and signal activityin the CAC.
Key Factors:
Acetyl-CoA, ATP, ADP and their interactions influence the cycle’s efficiency and operation.
Final note mentions other metabolic pathways including gluconeogenesis, amino acid synthesis, and lipid synthesis, linking the importance of the CAC to broader metabolic networks.