Chapter 5 Microbiology
Cells can generate high-energy molecules (ATP) using two pathways: aerobic or anaerobic. During fermentation, which is the anaerobic pathway, little ATP is produced. This is because the reactants and the products have oxidation states that are relatively close to each other. As such, a relatively small amount of energy is released and then transformed into high-energy compounds like ATP. For example, yeast can perform glycolysis followed by fermentation (both anaerobic processes). Overall, these processes produce a net gain of 2 ATP molecules as glucose is transformed into alcohol and CO2. The specific products of fermentation vary between species, as do the sugars that a species can ferment.
Some cells can metabolize sugar using a pathway that uses oxygen or another terminal electron acceptor whose oxidation state is much further from that of the reactants (sugars). Due to this difference in oxidation levels, more energy can be released and harvested as the sugar is processed. Unfortunately, this process is a bit complicated, involving a few different pathways, each of which involves multiple steps.
The first pathway that we are going to address is the Electron Transport Chain (ETC). This process uses the coenzyme NADH (a diffusible high-energy electron carrier) to deliver high-energy electrons to a series of proteins that are embedded in the cell membrane. As the electrons are shuffled through the ETC, redox reactions convert the electrons' energy into a concentration gradient of H+ ions. This gradient is called the Proton Motive Force and is used to power the production of ATP. The enzyme that makes this conversion is called ATP synthase (ATPase). The flow of H+ ions into the cell provides energy to ATPase to add a phosphate group to ADP (Adenosine diphosphate) through oxidative phosphorylation.
The second pathway that we must consider is called the Krebs Cycle. The reactions of the Krebs Cycle follow the process of glycolysis, and the pyruvate molecule produced in glycolysis is completely oxidized, releasing significantly more energy than when it serves as a terminal electron acceptor in fermentation. As the cycle processes the pyruvate molecule in a stepwise fashion, energy is harvested as high-energy electrons and carried by NADH. The NADH can then be used in the ETC to power the Electron Motive Force, and ultimately the production of ATP. The waste product of the Krebs Cycle is CO2.
Enzymes and Cellular Regeneration
Digestive enzymes are protein-based biological catalysts that play important roles in our lives. They help remove stains from our shirts, turn milk into cheese, and are responsible for turning our dinner into usable fuel for our bodies. However, enzymes do not work universally. Some are meant to work at high temperatures, while others work best at low temperatures. They may function optimally in acidic or neutral conditions.
In this activity, we will explore the optimal conditions for two different enzymes. The digestive enzyme lipase is produced in the pancreas and breaks down lipids in the small intestine, while pepsin breaks down proteins in the stomach.
The rate of an enzyme-catalyzed reaction can also be influenced by the presence of other molecules that can bind to the enzyme, altering its shape. In some reactions, a coenzyme is essential. This molecule binds to the protein strands of the enzyme, modifying its shape to be receptive to the substrate molecule. Without the coenzyme, the enzyme would not be able to attach to the substrate. Other molecules can decrease the reaction rate of enzymes by binding to the protein and either obstructing the binding site for the substrate or rendering the enzyme's shape incompatible with the substrate. These molecules are known as inhibitors.
Cellular Respiration
All cells require energy constantly, and their primary source of energy is ATP. The methods cells use to produce ATP vary depending on the availability of oxygen and their biological makeup. In many cases, cells are in an oxygen-rich environment. For example, as you sit and read this sentence, you are breathing in oxygen, which is then carried throughout your body by red blood cells. However, some cells grow in environments without oxygen (such as yeast in winemaking or the bacteria that cause botulism in canned food), and occasionally animal cells must function without sufficient oxygen (as in running sprints). In this activity, you will begin to explore the aerobic and anaerobic processes used by all organisms to produce ATP.
Glucose, or any carbon-based molecule, can be oxidized in oxygen to produce carbon dioxide and water. Combustion reactions release large amounts of energy. However, the energy release is uncontrolled. An organism would not be able to handle all that energy at once to perform the work of the cell. Cellular respiration is essentially the same reaction as combustion, but the oxidation of glucose occurs in several controlled steps. The same amount of energy is ultimately released, but it is gradually released in small, controlled amounts. High potential energy molecules of ATP are produced while the carbon atoms are used to form various other molecules of lower potential energy. Each of these steps is catalyzed by an enzyme specific to that step. Model 1 illustrates the ideal circumstances for cellular respiration. In some situations, however, one glucose molecule may not result in 38 ATP molecules being produced.
Oxidation is a loss of electrons, while reduction is a gain of electrons. These two processes must go hand in hand. In other words, electrons cannot be added to something out of thin air; they must have been taken off of something first.
Glycosis & Krebs Cycle
Glucose is a high-potential energy molecule, while carbon dioxide is a stable, low-potential energy molecule. During cellular respiration, when a glucose molecule is converted to carbon dioxide and water, energy is released and stored in high-potential energy ATP molecules. The three phases of cellular respiration that oxidize glucose to carbon dioxide are glycolysis, the Link reaction, and the Krebs cycle.
Glycolysis can occur in a cell with or without oxygen. In the presence of oxygen, the Link reaction, Krebs cycle, and oxidative phosphorylation complete the process of oxidizing glucose and maximizing energy output. Without oxygen, glycolysis is coupled with fermentation processes to provide a continuous energy supply to the cell. It's important to note that although the Link reaction and Krebs cycle do not require oxygen as a reactant, they will not occur in the cell without oxygen.
Oxidative Phosphorylation
The final phase of cellular respiration is oxidative phosphorylation. Both the electron transport chain and chemiosmosis make up oxidative phosphorylation. During this phase, all of the NADH and FADH2 produced in other phases of cellular respiration (glycolysis, the link reaction, and the Krebs cycle) are used to make ATP. The process occurs in the protein complexes embedded in the inner mitochondrial membrane.
The embedded protein complex, ATP synthase, is more of a machine than a chemical enzyme. Research has shown that a protein “rotor” down the middle of the ATP synthase complex turns as hydrogen ions flow through. This rotation causes other proteins to “squeeze” the ADP and inorganic phosphate groups together to form ATP.
Summary
Cells can make energy in two ways: aerobic (with oxygen) or anaerobic (without oxygen). In fermentation, a type of anaerobic process, cells make a small amount of energy because the reactants and products have similar oxidation states. Yeast, for example, can make energy through fermentation. This process produces 2 ATP molecules from glucose, turning it into alcohol and CO2. Different species produce different products during fermentation.
Some cells use oxygen to make more energy. This involves complex pathways like the Electron Transport Chain (ETC) and the Krebs Cycle. In the ETC, high-energy electrons are passed through proteins in the cell membrane, creating a gradient of H+ ions. This gradient powers ATP production by an enzyme called ATP synthase. The Krebs Cycle oxidizes pyruvate, producing high-energy electrons carried by NADH for ATP production.
Enzymes are important for cellular functions. They work best under specific conditions like temperature and pH. Enzyme-catalyzed reactions can be influenced by coenzymes or inhibitors. Lipase breaks down lipids in the small intestine, while pepsin breaks down proteins in the stomach.
Cells need ATP for energy. They can produce ATP through aerobic or anaerobic processes. Glucose can be oxidized to produce ATP in controlled steps during cellular respiration. Oxidation is a loss of electrons, while reduction is a gain of electrons. Glycolysis, the Link reaction, and the Krebs Cycle are phases of cellular respiration that convert glucose to carbon dioxide.
Oxidative phosphorylation is the final phase of cellular respiration, where NADH and FADH2 are used to make ATP through the electron transport chain and chemiosmosis. ATP synthase is a protein complex that helps make ATP by rotating as hydrogen ions flow through it.
Questions
1. Define the following terms: catabolism, anabolism, metabolism, catalyst.
Catabolism: Breaking down complex molecules into simpler ones
Anabolism: Building complex molecules from simpler ones
Metabolism: all Chemical reactions that occur within an organism, both catabolic and anabolic reactions
Catalyst: Substance that speeds up chemical reactions
2. Name three sources of energy for cells.
Carbs, lipids, proteins
3. Compare and contrast exergonic and endergonic reactions.
Exergonic: Release energy to the environment. Free energy of reactants is higher than product.
Endergonic: Requires energy to proceed, free energy of products is higher than reactants.
4. Where do the building blocks for biosynthetic pathways (catabolism) come from?
Carbon sources, like amino acids, purines, pyramids, lipids, sugars, enzyme cofactors
5. What is the role of ATP in the cell?
The main carrier of energy within a cell, when energy is needed ATP is hydrolyzed to release energy.
6. What is the role of NAD+ in the cell?
Important in metabolism, Transfers molecules from one molecule to another
7. What is an enzyme? What is the active site?
Enzymes are biological catalysts that speed up chemical reactions without being consumed
Active sites are where enzymes bind to substrate molecules
8. What is the optimum temperature for an enzyme?
37C or body temp, extreme temps cause denaturation
9. Considering an enzyme catalyzed reaction, what must happen for the reactants to be
converted to products?
Binding: The substrates must bind to the active site of the enzyme. The active site is a specific region on the enzyme where the substrates bind. This forms an enzyme-substrate complex.
Catalysis: Once the substrates are bound, the enzyme facilitates the chemical reaction between the substrates. The enzyme does this by lowering the activation energy required for the reaction to occur.
Release: After the reaction, the products (the transformed substrates) are released from the enzyme. The enzyme is then free to bind with new substrates.
10. What does an enzyme do to the activation energy of a reaction?
An enzyme lowers the activation energy of a reaction. The activation energy is the minimum amount of energy required for a chemical reaction to occur. By lowering this energy barrier, enzymes speed up the rate of the reaction. This is crucial in biology because it allows necessary reactions to occur more quickly and efficiently than they would without the enzyme.
11. How many times can an enzyme be used?
An enzyme can be used repeatedly. Once an enzyme has catalyzed a reaction and the products have been released, the enzyme is free to bind with new substrates and catalyze the same reaction again.
12. An enzyme can act on how many different substrates?
Typically, an enzyme acts on a specific substrate or a group of closely related substrates. This is due to the unique shape and structure of the enzyme’s active site, which is complementary to the shape and structure of its substrate(s).
13. What is the reason why an enzyme reacts on a specific substrate?
The reason an enzyme reacts with a specific substrate is due to the lock and key model. The enzyme’s active site (the ‘lock’) has a unique shape and structure that fits perfectly with the substrate (the ‘key’). This specificity allows the enzyme to bind only with its specific substrate(s) and catalyze the intended reaction.
14. What is an Oxidation/Reduction reaction?
An Oxidation/Reduction (or Redox) reaction involves the transfer of electrons from one molecule to another. In this process, one molecule is oxidized (loses electrons) and another molecule is reduced (gains electrons).
15. What happens to a substance that is oxidized? What happens when something is
Reduced?
When a substance is oxidized, it loses electrons. This often involves the substance gaining an oxygen atom or losing a hydrogen atom. When something is reduced, it gains electrons. This often involves the substance losing an oxygen atom or gaining a hydrogen atom.
16. When a biological molecule undergoes a Redox reaction, what atom is nearly always
also involved in the reaction?
When a biological molecule undergoes a Redox reaction, the atom that is nearly always involved is hydrogen. Hydrogen atoms are often transferred in biological Redox reactions, carrying with them a positive charge (a proton) and an electron. This allows for the transfer of electrons during the reaction.
17. What controls Oxidation/Reduction reactions in the body?
Oxidation/Reduction reactions in the body are controlled by metabolic processes. These processes are regulated by catabolic and anabolic hormones. Catabolic hormones stimulate the breakdown of molecules and the production of energy. These include cortisol, glucagon, adrenaline/epinephrine, and cytokines
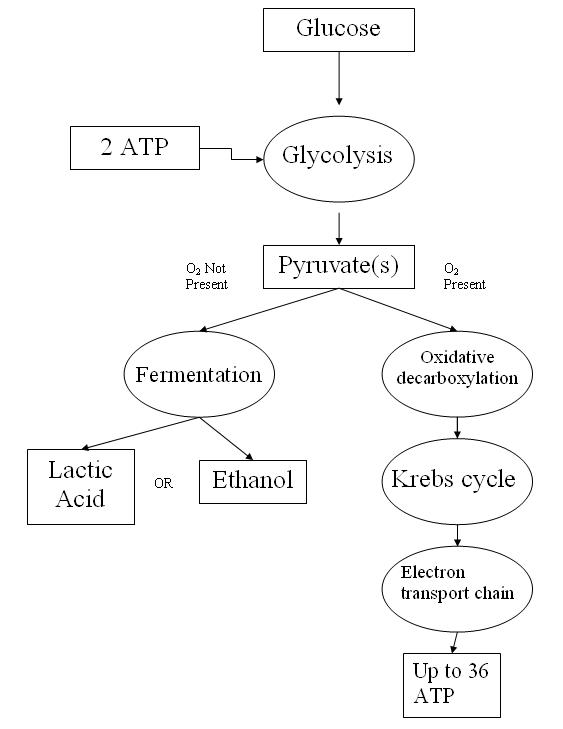
18. Draw a flow chart to show glycolysis, fermentation and the reactions of aerobic
respiration. Include any energy inputs our outputs.
Glycolysis:
Location: Cytoplasm
Inputs: One molecule of glucose, two molecules of NAD+, two molecules of ADP, and two molecules of inorganic phosphate (Pi).
Outputs: Two molecules of pyruvate, two molecules of NADH, and a net gain of two molecules of ATP (four ATP molecules are produced, but two are used during the process).
Process: The six-carbon sugar glucose is split into two three-carbon molecules called pyruvates. This process occurs in two main phases: the energy-requiring phase and the energy-releasing phase.
Fermentation:
Location: Cytoplasm
Inputs: Two molecules of pyruvate (from glycolysis) and two molecules of NADH.
Outputs: Depending on the type of fermentation, the end products can be ethanol and carbon dioxide. The process also regenerates NAD+ from NADH, which allows glycolysis to continue producing ATP in the absence of oxygen.
Process: Fermentation begins with the two pyruvate molecules produced by glycolysis. In the absence of oxygen, these molecules are converted into other forms, such as lactate or ethanol, depending on the type of fermentation.
Aerobic Respiration:
Location: Mitochondria
Inputs: Two molecules of pyruvate (from glycolysis), oxygen, and ADP.
Outputs: Carbon dioxide, water, and ATP.
Process: Aerobic respiration consists of three stages:
Pyruvate Decarboxylation: Each pyruvate molecule is converted into a two-carbon molecule bound to Coenzyme A, known as acetyl CoA. This process releases one molecule of carbon dioxide and generates one NADH molecule per pyruvate.
Krebs Cycle: The acetyl CoA combines with a four-carbon molecule and goes through a cycle of reactions, ultimately regenerating the four-carbon starting molecule. This cycle produces two molecules of carbon dioxide, one ATP, three NADH, and one FADH2 per acetyl CoA.
Electron Transport Chain and Oxidative Phosphorylation: The NADH and FADH2 deposit their electrons in the electron transport chain. As electrons move down the chain, energy is released and used to pump protons out of the matrix, forming a gradient. This gradient drives the synthesis of ATP in a process called oxidative phosphorylation. Oxygen is the final electron acceptor in this process and gets reduced to form water.
In summary, these processes convert glucose into usable energy in the form of ATP, with different processes taking over depending on the availability of oxygen. Glycolysis and fermentation can occur without oxygen, while aerobic respiration requires oxygen. The total ATP yield can be up to 38 ATP molecules per glucose molecule during aerobic respiration, while it’s only 2 ATP per glucose molecule during glycolysis and fermentation.
19. In which pathway of respiration is the most NADH generated?
The most NADH is generated in the Krebs Cycle. This cycle is part of cellular respiration, where each pyruvate from glycolysis is turned into acetyl CoA, which then goes through a series of reactions, producing ATP, NADH, and FADH2.
20. What is the final electron acceptor in aerobic respiration?
In aerobic respiration, the final electron acceptor is an oxygen molecule (O2). This oxygen molecule becomes reduced to form water (H2O) by the final electron transport system carrier.
21. What are the reactants and products of glycolysis? Is this process aerobic or
Anaerobic?
The reactants of glycolysis are one molecule of glucose, two molecules of NAD+, two molecules of ADP, and two molecules of inorganic phosphate (Pi). The products are two molecules of pyruvate, two molecules of NADH, and a net gain of two molecules of ATP (four ATP molecules are produced, but two are used during the process). Glycolysis is an anaerobic process, meaning it does not require oxygen.
22. When anaerobic respiration (fermentation) follows glycolysis, what is the total gain
of ATP?
When anaerobic respiration (fermentation) follows glycolysis, the total gain of ATP is 2 ATP per glucose molecule. These ATP molecules are generated during the glycolysis phase, as fermentation does not produce any additional ATP.
23. In the absence of oxygen, fermentation follows glycolysis. What molecule is
produced by fermentation that is needed for glycolysis to continue?
In the absence of oxygen, fermentation follows glycolysis. The molecule produced by fermentation that is needed for glycolysis to continue is NAD+. During fermentation, NADH is oxidized back to NAD+, which can then be reused in glycolysis to keep the process running and continue generating ATP.
24. What is the final electron acceptor in fermentation? What is the final electron
acceptor in aerobic respiration?
In fermentation, the final electron acceptor can vary depending on the type of fermentation. In lactic acid fermentation, the final electron acceptor is pyruvate, which gets reduced to lactic acid. In alcohol fermentation, the final electron acceptor is acetaldehyde, which gets reduced to ethanol. In aerobic respiration, the final electron acceptor is an oxygen molecule (O2).
25. If oxygen is present, what processes follow glycolysis?
If oxygen is present, the processes that follow glycolysis are the Krebs Cycle and the Electron Transport Chain. These processes are part of aerobic respiration and occur in the mitochondria of the cell. They allow for the complete breakdown of glucose and the generation of a large amount of ATP.
26. Describe the role of the Tricarboxylic Acid Cycle (Krebs cycle, Citric Acid Cycle) in
aerobic respiration?
The Krebs cycle plays a central role in the breakdown of organic fuel molecules in aerobic respiration. It is the second stage of cellular respiration, where acetyl coenzyme A (acetyl CoA), a two-carbon compound, enters the TCA cycle and is converted into carbon dioxide and energy1. This metabolic process occurs in most plants, animals, fungi, and many bacteria.
27. What are the products of the tricarboxylic acid cycle?
The products of the TCA cycle include two molecules of carbon dioxide, three NADH, one FADH2, and one ATP (or GTP) per turn of the cycle21.
28. Each NADH that enters the electron transport chain gives rise to how many ATP?
Each NADH that enters the electron transport chain gives rise to approximately 2.5 ATP.
29. During which phase of cellular respiration is the majority of ATP formed?
The majority of ATP is formed during the Oxidative Phosphorylation phase of cellular respiration
30. In bacterial cells, where is the electron transport chain located? Where does this
happen in eukaryotic cells?
In bacterial cells, the electron transport chain is located in the plasma membrane5. In eukaryotic cells, it is located in the inner mitochondrial membrane6.
31. As the electron transport chain shuttles electrons, the energy in the flow of
electrons is directly transferred into what form?
As the electron transport chain shuttles electrons, the energy in the flow of electrons is directly transferred into a proton gradient across a membrane67. This gradient drives the creation of ATP.
32. What is the total gain of ATP from the aerobic respiration of one glucose molecule in
bacterial cells?
The total gain of ATP from the aerobic respiration of one glucose molecule in bacterial cells is approximately 30-32 ATP.
33. Compared to fermentation, there is ______ times more net energy released through
aerobic respiration
Compared to fermentation, there is about 19 times more net energy released through aerobic respiration. This is because aerobic respiration extracts more energy from each glucose molecule.