20.1 Bronsted - Lowry acids and bases
Development of models to explain acid-base behaviour

Bronsted - Lowry acids and bases
Arrhenius model of acids and bases
acids dissociate and release H+ ions in aqueous solution
alkalis dissociate and release OH- ions in aqueous solution
H+ are neutralised by OH- ions to form water.
An alkali is a soluble base.
Bronsted-Lowry model extends to this model to emphasise the role or proton transfer between species
A Bronsted-Lowry acid is a proton donor
A Bronsted-Lowry base is a proton acceptor.
Conjugate acid-base pairs
single arrow - can be used to indicate that the forward reaction effectively goes to completion.
HCL and CL- are called a conjugate acid-base pair, meaning it contains two species that can be interconverted by transfer or a proton.
In the forward direction , HCL releases a proton to form its conjugate base, CL-
In the reverse direction, CL- accepts a proton to forms its conjugate acid H+.
Examples of conjugate acid-base pairs.
Neutralisation
H+ (aq) + OH-(aq) → H20(l)
In hydrochloric acid, H+ ions have been supplied by dissociation
HCL(aq) →← H+(aq) + CL-(aq)
When these two equations are combined, there are not two conjugate acid-base pairs which are labelled acid 1, base 1, acid 2, base 2.

Forward reaction
HCl is an acid and donates H+
OH- is a base and accepts H+
Reverse reaction
H20 is an acid and donates H+
CL- is a base and accepts H+
The hydronium ion (H3O+)
In aqueous solution, dissociation requires a proton to be transferred from an acid to a base.
The dissociation does not take place unless water is present.
 H2O has accepted a proton to form its conjugate acid, the hydronium ion H3O+. This hydronium ion is very important as it acts as an active acid ingredient in any aqueous acid.
H2O has accepted a proton to form its conjugate acid, the hydronium ion H3O+. This hydronium ion is very important as it acts as an active acid ingredient in any aqueous acid.

Monobasic, dibasic and tribasic acids
This refers to the total number of hydrogen ions in the acid that can be replaced per molecule in an acid-base reaction. This would typically be the replacement of protons by metal ions or ammonium ions to form a salt.
The number of hydrogen atoms in the formula gives a clue for the type of acid.
The role of H+ in the reactions of acids
Acids react with metals, carbonates, metal oxides and alkalis to form salts. The active species from the acid is H+ (aq) and ionic equations can be written for each type of reaction to emphasise the role of H+ ions.
When writing ionic equations with H+(aq), the acid does not matter.
The ionic equation for the reaction of magnesium with dilute hydrochloric acid and dilute sulfuric acid is the same.
Spectator ions - are ions that do not change during the reaction, they can be cancelled out.
Redox reactions between acids and metals
Dilute acids undergo redox reactions with some metals to produce salts and hydrogen gas.
acid + metal → salt + hydrogen
For zinc, the ionic equation for a reaction with any dilute acid is
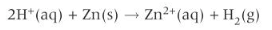
study tip - a few acids, like nitric acid are also strong oxidising agents, except when it is very dilute, nitric acid reacts differently with most metals.
Reactions between acids and bases
Neutralisation of acids with carbonates
carbonates are bases that neutralise acids to form a salt, water and carbon dioxide.
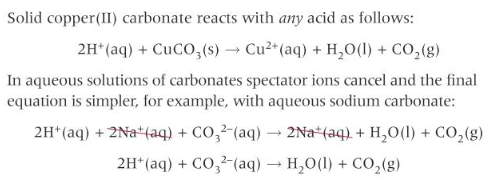
For a solid, you still write out the ionic equation with the full formula of the carbonate because the carbonate changes state during the reaction.
Neutralisation of acids with metal oxides
An acid is neutralised by a solid metal oxide or hydroxide to form a salt and water only.
Acid + base → salt + water
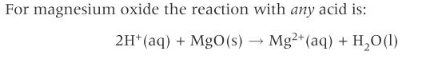
Neutralisation of acids with alkalis
With alkalis the acid and base are in solution, the overall reaction is the same for metal oxides.
acids + alkali → salt + water
The ionic equation is
H+(aq) + OH-(aq) → H2O (l)