4.5 - Energy changes - notes
4.5.1 Exothermic and endothermic reactions
4.5.1.1 Energy transfer during exothermic and endothermic reactions
Chemicals store a certain amount of energy - and it varies with each chemical
If the product’s molecules of a reaction store more energy than the original reactants:
They must’ve take in the difference in energy from the surroundings
But if the products molecules store less:
then the excess energy was transferred to the surrounding during the reaction
Overall amount of energy doesn’t change (energy is conserved)
An exothermic reaction is one which transfers energy to the surroundings, usually by heating. This is shown by a rise in temperature.
Example of Exothermic Reactions:
Burning fuels (combustion)
Neutralisation reactions (acid + alkali)
Oxidation reactions
Exothermic reactions have lots of everyday uses:
Some hand warmers use the exothermic oxidation of iron in the air with a salt solution catalyst) to release energy
Self heating cans of hot chocolate and coffee also rely on exothermic reactions
An endothermic reaction is one which takes in energy from the surroundings. This is shown by a fall in temperature.
Examples of Endothermic reactions:
Reaction between citric acid and sodium hydrogencarbonate
Some sports injury packs
Thermal decomposition
Electrolysis
Exothermic and Endothermic reaction r.p. flashcards on pmt
4.5.1.2 Reaction profiles
For atoms or particles to react with each other in a chemical system they must first come into contact with each other in a collision
Several factors come into play when analysing collisions such as:
Energy
Orientation
Number of collisions per second
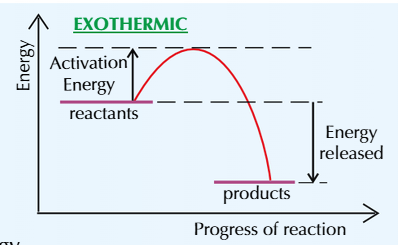
This exothermic reaction profile shows that the products are at a lower energy than the reactants
The difference in height represents the overall energy change in the reaction (the energy given out) per mole
The initial rise in the line represents the energy needed to break the old bonds and start the reaction
This is called the activation energy which = the minimum amount of energy the reactants need to collide with each other and react
The greater the activation energy, the more energy needed to start the reaction
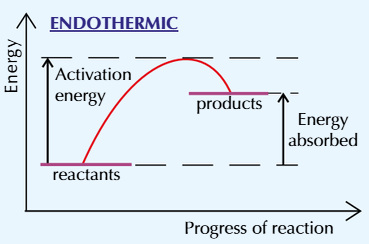
This endothermic reaction shows that the products are at a higher energy than the reactants
The difference in height represents the overall energy change in the reaction (the energy taken in) per mole
4.5.1.3 The energy change of reactions (HT only)
During a chemical reaction old bonds are broken and new bonds are formed:
Energy must be supplied to break existing bonds - so this must be an Endothermic process
More energy is required to break the bonds than is released from making the new bonds
The change in energy is positive since the products have more energy than the reactants
Therefore an endothermic reaction has a positive change in energy
Energy must be released when new bonds are formed - so this must be an Exothermic process
More energy is released when new bonds are formed than energy required to break the bonds in the reactants
The change in energy is negative since the products have less energy than the reactants
Therefore an exothermic reaction has a negative change in energy
Exothermic reactions is when the energy released forming the bonds is greater than the energy used to break them
Endothermic reactions is when the energy used to break bonds is greater that the energy needed to form them
Practice Bond energy calculations
4.5.2 Chemical cells and fuel cells
4.5.2.1 Cells and batteries
An electrochemical cell = a basic system made up of 2 different electrodes in contact with an electrolyte
Electrolyte = a liquid that contains ions which react with the electrodes
The two electrodes of different reactivity are metals because they must conduct electricity
A common example is zinc and copper:
Zinc is the more reactive metal and forms ions more easily, readily releasing electrons
The electrons give the more reactive electrode a negative charge and sets up a charge difference between the electrodes
The electrons then flow around the circuit to the copper electrode which is now the more positive electrode
A wire connected to the electrodes enables charge to flow and electricity is produced
The voltage of a cell depends on many factors:
Different metals will react differently with the same electrolyte
The bigger the difference in the reactivity of the electrodes the bigger the voltage of the cell
The electrolyte used in a cell will also affect the size of the voltage with different ions in solution will react differently with the electrodes used
A battery is formed by connecting two or more cells together in series
The voltages of the cells in the battery are combined so there is a bigger voltage overall
In Non-rechargeable batteries the reactants get used up
Over time the ions in the electrolyte and the metal ions on the electrode get used up and turn into products of the reaction
Once the limiting reactant is used up the reaction can’t happen so no electricity is produced
The products can’t be turned back into the reactants so the cell can’t be recharged
A good example -
Non-rechargeable batteries (alkaline batteries) are made up of cells which use irreversible reactions
In a rechargeable cell, the reaction can be reversed by connecting it to an external electric current
4.5.2.2 Fuel cells
A fuel cell is an electrical cell that’s supplied with a fuel and oxygen or air and uses energy from the reaction to produce electrical energy efficiently
When the fuel enters the cell it becomes oxidised and sets up a p.d within the cell
One type of fuel cell is the hydrogen-oxygen fuel cell
This fuel cell combines hydrogen and oxygen to produce nice clean water and energy
Fuel cell vehicles don’t produce as many pollutants as other fuels
They’re only by-products are water and heat
Electric vehicles don’t produce as much pollutants either - but their batteries are much more polluting
Batteries in ev cars are rechargeable but there’s a limit to how many times they can be recharged before they need replacing
Batteries are also more expensive to make than fuel cells
Batteries store less energy than fuel cells so would need to recharge more often - which can take more time
Advantages of hydrogen fuel cells
They do not produce any pollution
They produce more energy per kilogram than either petrol or diesel
No power is lost in transmission as there are no moving parts, unlike an internal combustion engine
No batteries to dispose of which is better for the environment
Continuous process and will keep producing energy as long as fuel is supplied
Disadvantages of hydrogen fuel cells
Materials used in producing fuel cells are expensive
High pressure tanks are needed to store the oxygen and hydrogen in sufficient amounts which are dangerous and difficult to handle
Fuel cells are affected by low temperatures, becoming less efficient
Hydrogen is expensive to produce and store
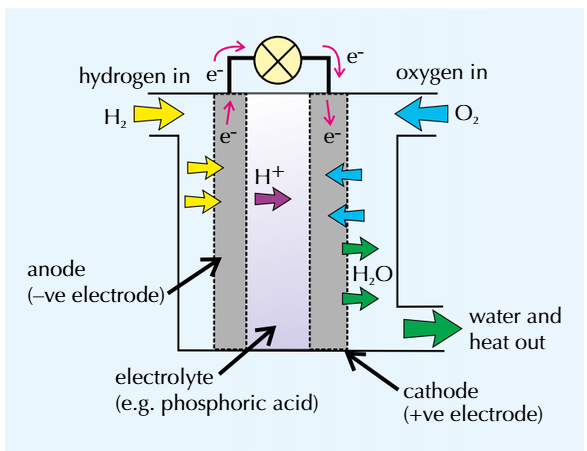
Electrolyte is often an acid
Electrodes are often porous carbon with a catalyst
At the negative electrode (the anode):
Hydrogen loses electrons to produce H2+ - this is oxidation as shown by the following equation:
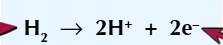
At the positive electrode (cathode):
These H+ ions in the electrolyte move to the cathode
Oxygen gains electrons from the cathode and reacts with h+ ions to form water. This is reduction as shown by the following equation:
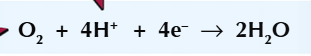
The overall reaction is hydrogen plus oxygen which gives water:
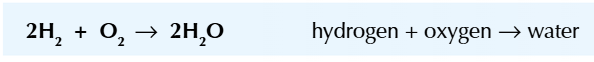
There’s reduction at the cathode and oxidation at the anode which = a REDOX reaction