Topic 9: The Electron
T9VA: Duality of Nature
Particle v.s Wave
Light as a wave
Matter as something that has mass and position
Reminder
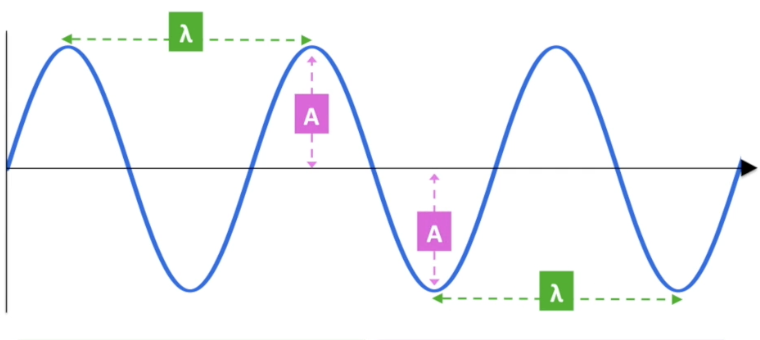
λ represents wavelength
distance between 2 consecutive troughs or peaks
Amplitude:
height of the wave as defined from the centre to the top of a trough of peak
Characteristics of a Wave
Diffraction: when a wave moves through a small slit, it will spread out in a process called diffraction
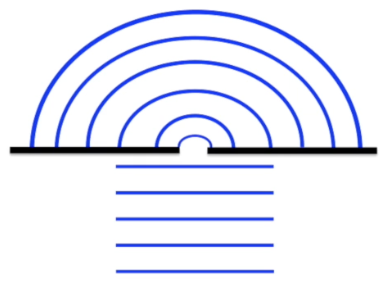
HOWEVER, a particle hitting a slit will either bounce back or go straight through, NOT diffract
Interference: When waves move through a double slit they both diffract and thus interfere with one another
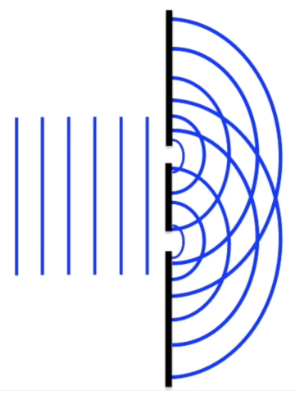
However, particles will go through the slit or bounce off of it without necessarily interacting
NOTE: wave interference can either be constructive or destructive
we can understand interference by looking at what happens when we add two waves together
Constructive Interference
Combining two waves that have their peaks and troughs aligned will result in a wave with a greater amplitude than that of the combining waves
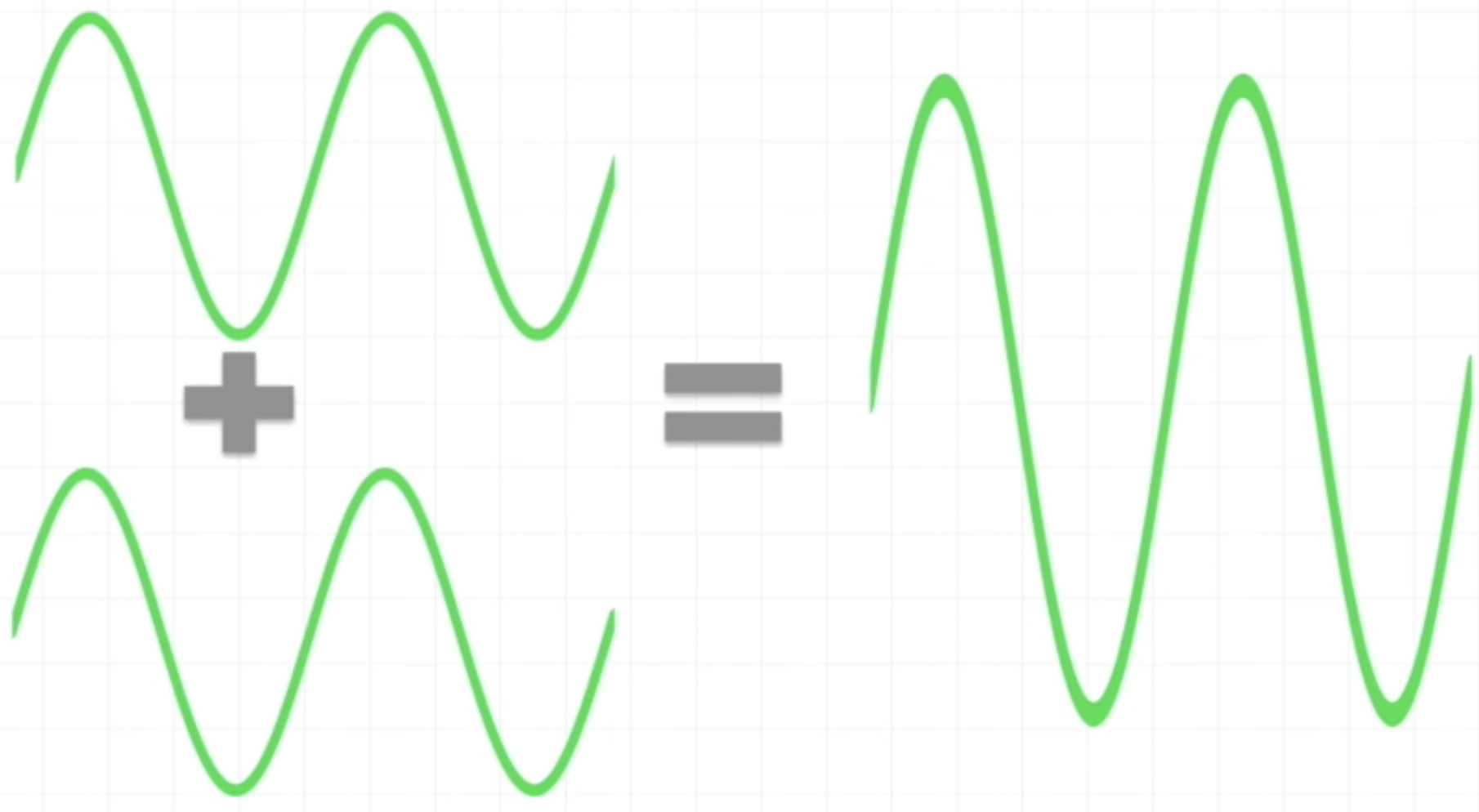
These two similar waves are said to be in phase
These waves interact by the ADDITION of their amplitudes
Destructive Wave Interference
Combining two waves that DO NOT have their peaks and troughs aligned will result in a wave with a lower amplitude than the original waves
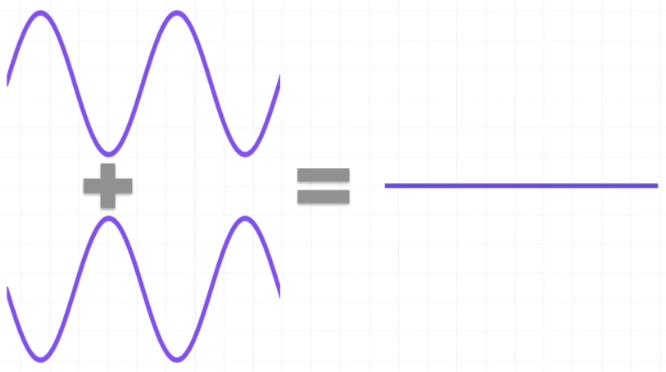
these two waves are said to be out of phase
sometimes, the resulting wave will have no amplitude
These waves interact by the SUBTRACTION of their amplitudes
Characteristics of particles
recoil (bouncing off of stuff)
mass
By the end of the 19th century, scientists knew that
light is a wave because it shows interference and diffraction patterns
HOWEVER, photons act like particles
The energy of the photon is proportional to the frequency of the light, and not to its amplitude

Based on this, scientists posed the question:
If we can model light, in photons, as a particle, shouldn’t it also have a measurable mass ?
Einstein proposed that E = mc2, determining that energy does have mass.
The mass of a photon can be theoretically computed with this equation:
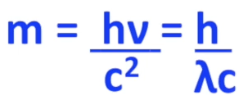
The Compton effect also describes the fact that light (photons) have mass. Remember, that when light hits something, it diffracts. When a particle hits something, it recoils. Compton shot light at a metal.
If light were a wave, it would have diffracted; however
if ended up kicking an electron out of the metal and recoiling, just like a billiard ball would
Compton used this experiment and Einsteins equation to demonstrate that the mass of the displaced electron and the recoil of photons was consistent with the predicted mass of a photon
So light is both a wave and a particle at the same time, depending on how it is measured
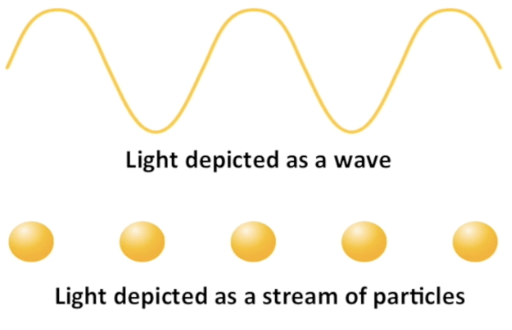
If light can be both a wave and a particle, what about matter ?
Matter also has a wavelength

h: Planck’s Constant = 6.626×10-34 J-s
m = mass (kg)
v = speed (m/s)
this is similar to Einstein’s equation except, particles don’t move at the speed of light, so instead of the constant c, it is a variable speed (v)
This λ is called the deBroglie wavelength, and it describes the wavelength of matter
NOTE: for macroscopic matter, the wavelength would be too small, and we can’t make experiments to measure them. So on a macroscopic level, we cannot experimentally determine that matter is a wave. However, the wavelengths of electrons are on a large enough order of magnitude that their wavelengths can physically be measured using solid crystals.
In summary, both matter and light have both particle and wave behavious
LIGHT:
particle:
Compton mass
recoiling
Ephoton is quantized and related to its frequency and mass:

wave
interference
diffraction (double slit experiment)
MATTER:
particle
observable mass
wave
electrons and neutrons show diffraction patterns (double slit experiment)
the wavelength of matter can be determined from its mass:

NOTE: v in this equation is NOT the speed of light
T9VB: Birth of Quantum Mechanics
After the establishing of matter and light being both particles and waves, the planetary model (Niels Bohr) was deemed unproductive bc
electrons cant orbit a central nucleus without losing energy and therefore spiralling inwards
atoms would have decayed if this were accurate
So scientists needed to come up with a new model for the structure of the atom
Remember that the fundamental equation of classical mechanics is the equation f = ma
atoms cannot be explained this way
Quantum Mechanics
from quantum mechanics we get the principle that not all energies are allowed (only those in specific states); and that
we cannot actually predict with certaintly where small particles are found (they are not localised)
Fundamental equation of quantum mechanics, the Schrodinger Equation:
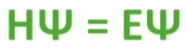
E: energy → gives us the energy levels of the atom
H: hamiltonian operator → contains the mathematic operations of partial derivatives
Ψ = the critical wave function → contains all the information we need about the electron
This equation relates the particle nature of an electron to its wave nature. A stable atom can be predicted using Schrodinger’s Equation
there are always two types of solutions from this equation:
E: the energy levels; or
Ψ: the wave functions
small particles are described by these wave functions (expressed with Ψ)
The principle of quantization is that matter and electrons are restricted to certain energy states, and that they cannot “land” between levels. Why does this happen ?
Think of standing waves, waves that are physically anchored at the end, like the strings of a guitar. If we pluck a guitar string, we hear fixed tones from fixed frequencies. This is because the ends of the strings are anchored, and are effectively quantizing the energy of the vibration, or restricting the energy to certain levels. See below:

The string’s anchors on the guitar are equivalent to boundary conditions for the wave function (Ψ). Like the guitar string, the anchoring of boundary conditions is what produces fixed and stable energy states for an atom.
Anchoring (boundary conditions) leads to quantization of energy levels.
energy level and wave functions are labelled by quantum numbers
T9VC: Quantum Numbers
Reminder: we solve Schrodinger’s Equation for two things: energy and wave functions (Ψ)
An important outcome to the Schrodinger equation is a set of numbers used to label and identify these energies and wavefunctions: these are called quantum numbers
n
ℓ
mℓ
ms (this will be discussed in T9VD)
n
the principle quantum number
represents the orbital energy level
it is an integer value
NOTE: in the Rydberg equation, E is restricted by the value of n. If n could be any real number, all the E would be possible, so E would not be quantized.
n = 1, 2, 3, …
ℓ
angular momentum quantum number
represents the orbital shape such as s, p, d, etc
ℓ = 0: s orbital
ℓ = 1: p orbital
ℓ = 2: d orbital
ℓ = 3: f orbital
ℓ can continue with larger integer values***
possible ℓ values are restricted by the n value, we can determine the value of ℓ from n
ℓ = n-1, … 0
the maximum ℓ can be is n-1
the minimum ℓ can be is 0
ℓ also includes all the integers between n-1 and 0
***NOTE: elements in ground state can have electron configurations through the f-block. However, when electrons become excited, the energy levels can go higher, the value of n can be extended, so the value of ℓ can also be extended. ℓ values above 3 (those that represent orbitals above s, p, d, and f, are labelled with the alphabet, starting from f, g, h, and going on)
mℓ
magnetic quantum number
represents the number of orbitals, which represents the orbital orientation
the number of mℓ values dictates the number of orbitals
EXAMPLE: ℓ = 1: p orbital; mℓ = -1, 0, 1: 3p orbitals
possible mℓ are restricted by the ℓ value
mℓ= +ℓ, …, 0, …, -ℓ
if ℓ = 4, mℓ = -4, -3, -2, -1, 0, 1, 2, 3, 4
always integers
T9VD: Electron Spin
There is also a quantum number ms
When a stream of electrons goes through a magnetic field, the stream bends in one direction or another. This suggests that the electron itself has a specific spin (electronic spin) that can be redirected or manipulated by magnetic interference. An electron’s spin is represented with the quantum number ms
ms
spin quantum number
represents the spin on an electron
the value of ms = + ½, - ½ (spin up and spin down)
Putting all the quantum numbers together means that all electrons in an atom each have four quantum numbers: n, l, ml, and ms. No two electrons in an atom can have the same set of four quantum numbers
this means that electrons that sit in the same orbital must have opposite spins
Example of labelling an electron with its quantum numbers. Let’s say we’re given this configuration:
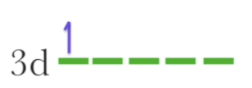
n: the electron is in the third shell, so n = 3
l: the electron is in the d suborbital, so l = 2
ml: based on l = 2, ml can be -2, -1, 0, 1, 2; we don’t know what the orbital looks like based on this image, so we can pick any of these numbers
let’s pick -2
ms: this electron is either down-spin or up-spin, so ms is either + ½, - ½. we can pick either one because we don’t have further information about this electron
let’s pick + ½
So, we are left with the set of these four quantum numbers: 3, 2, -2, + ½. All of the other electrons in the 3d suborbital have to have a different set, but since they are all 3d electrons, the n and l remain the same, and only the ml and ms change.
T9VE: Wave Functions
Let’s explore the wave functions (Ψ) for the hydrogen atom. Why hydrogen ? The hydrogen atom wave functions serve as models for all the other atoms.
NOTE: wave functions (Ψ) themselves are not observable. However, Ψ2 gives us the electronic density, which is the PROBABILITY of finding an electron in a given area of the atom
finish this vi