Acid-Base Property of Amino Acids
strongly acidic conditions → positive ion forms an amino acid as a base and COO- gains a proton , resulting in a zwitterionic form that has a net charge of zero. This protonation can significantly affect the amino acid's solubility and reactivity.
strongly alkaline conditions→ negative ion forms amino acid that behaves as an acid, NH3 loses a proton.
acidic amino acids → Aspartate & Glutamate
basic amino acids → Histidine, Lysine, Arginine.
Zwitterions → can have both positive and negative ions in a molecule in physiological pH ~7
Isoelectric point (pl) → pH at which a particular molecule can have no net electrical charge dominated by zwitterions.
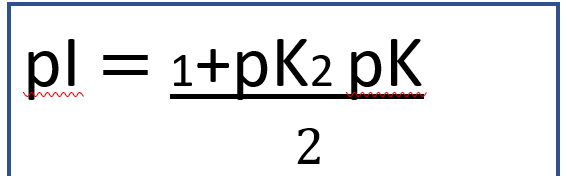

Conceptual Explanation
1. Acidic form (left side)
At low pH (acidic conditions), there are lots of free H⁺ ions in solution.
So, everything that can accept a proton does.
The amino group is –NH₃⁺ (it has gained an extra H⁺).
The carboxyl group is –COOH (it keeps its proton).
So the overall form is:
H₃N⁺–CH(R)–COOH
When the solution pH equals the pKa of a functional group, 50% of the molecules are protonated and 50% of the molecules are deprotonated.
As pH increases, more protons leave. As pH decreases, more protons are gained → gaining and losing protons (H+)