Gen Chem Reviewer FINALS
GAS LAWS
The Concept of Ideal Gas
For a gaseous system to become ideal, there are four conditions that must be satisfied first, and these are the following:
The gas particles have negligible volume.
The gas particles are equally sized and do not interact with neighboring gas particles.
The gas moves in a random motion.
Collisions between gas particles are perfectly elastic.
EMPIRICAL GAS LAWS
BOYLE’S LAW relates the pressure and volume of an ideal gas under constant temperature. It states that at a constant temperature, the pressure of a fixed amount of gas is inversely proportional to the volume of the gas.
Robert Boyle, a British chemist and natural philosopher, demonstrated this phenomenon in the 17th century using a bent glass tube and mercury.
FORMULA:
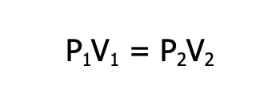
P is the pressure, and V is the volume of the gas under the two different sets of conditions.
NOTE: Keep in mind that this formula is valid only under constant temperature and as long as the amount of the gas does not change.
Practice Problem: A sample of oxygen gas exerts a pressure of 2.50 atm in a 20.0 L container. What will be the new pressure it will exert if all of it will be transferred to a 16.0 L container at constant temperature? Assume that the gas behaves ideally.

CHARLES’S LAW states that the volume of a fixed amount of gas maintained at constant pressure is directly proportional to the absolute temperature of the gas.
FORMULA:
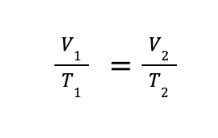
V1 and T1 are the initial volume and temperature, while V2 and T2 are the final volume and temperature, respectively.
NOTE: When working with temperatures in gas laws, make sure that the temperature is always on the Kelvin scale.
GAY-LUSSAC’S LAW relates the pressure and temperature of a fixed amount of gas under constant volume conditions.
FORMULA:
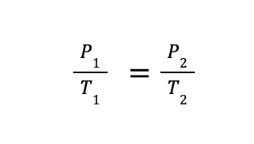
P1 and T1 are the initial volume and pressure, while P2 and T2 are the final volume and pressure, respectively.
AVOGADRO’S LAW states that at constant temperature and pressure, the volume of a gas is directly proportional to the number of moles of gas present.
FORMULA:
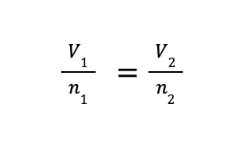
V1 and n1 are the initial volume and number of moles, while V2 and n2 are the final volume and number of moles, respectively.
COMBINED GAS LAW is obtained by combining Boyle’s law, Charles’ law, and Gay-Lussac’s law. This law expresses the relationship between the volume, pressure, and absolute temperature of a fixed amount of gas.
FORMULA:
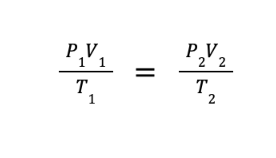
Practice Problem: A fixed amount of an ideal gas is compressed to half of its original volume. If initially, the gas exerts 1.5 atm at a temperature of 300 K, what would be the ratio of final temperature over the final volume after compression if the pressure becomes 2.0 atm?
Solution: It was stated that only the amount of gas is constant. Hence, we should use the combined gas law.
Since the gas is being compressed to half of its initial volume, and there is no initial volume given, then we can assign arbitrary values. For simplicity, we let V1 = 2.0 L and V2 = 1.0 L. Furthermore, P1 = 1.5 atm, T1 = 300 K, and P2 = 2.0 atm.
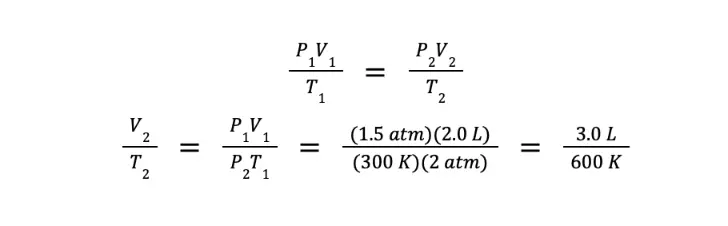
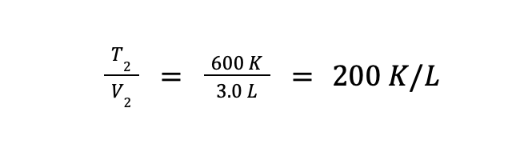
We are asked to determine the ratio of the final temperature over the final volume. We can getthis by getting the reciprocal of the equation above, which gives us
IDEAL GAS EQUATION accounts for the behavior of all ideal gas. This equation relates the pressure, absolute temperature, volume, and amount of the gas. This means that if we know three out of four properties of the gas, then we can compute the missing property using the ideal gas equation.
FORMULA:
PV = nRT
R = 0.0821 (L • atm/ mol • K)
NOTE: A mole of ideal gases, regardless of identity, occupies a molar volume of 22.414 L at 273.15 K (or 0 ℃) with a pressure of 1 atm. This temperature and pressure condition is known as the standard temperature and pressure (STP).
STP: 273.15 K (0 °C) and 1 atm (101.325 kPa or 760 Torr)
Practice Problems: Determine the volume of occupied by 2.34 grams of carbon dioxide gas at STP.
SOLUTION: Rearrange PV = nRT to this: V = nRT / P
Substitute:
V = [(2.34 g / 44.0 g mol) (0.0821 L atm mol K) (273.0 K)] / 1.00 atm
V = 1.19 L (to three significant figures)
Problem #2: A sample of argon gas at STP occupies 56.2 liters. Determine the number of moles of argon and the mass of argon in the sample.
SOLUTION: Rearrange PV = nRT to this: n = PV / RT
Substitute:
n = [(1.00 atm) (56.2 L) / (0.08206 L atm mol K) (273.0 K)]
n = 2.50866 mol
Multiply the moles by the atomic weight of Ar to get the grams:
2.50866 mol x 39.948 g/mol = 100 g
Problem #3: At what temperature will 0.654 moles of neon gas occupy 12.30 liters at 1.95 atmospheres?
SOLUTION: Rearrange PV = nRT to this: T = PV / nR
Substitute:
T = [(1.95 atm) (12.30 L)] / [(0.654 mol) (0.08206 L atm mol K)]
T = 447 K
PERIODIC TRENDS
ATOMIC RADIUS is defined as one-half the distance between two adjacent atoms. Generally, atomic radius increases down the group and decreases across a period from left to right.
ELECTRONEGATIVITY is the tendency of an atom to draw shared electrons in a chemical bond towards itself.
IONIZATION ENERGY is the minimum energy required (in kJ/mol) to remove one electron from an isolated atom or molecule.
ELECTRON AFFINITY is the atom’s ability to accept one or more electrons. The more willing an atom is to accept an electron, the more positive its electron affinity is.
NOTE: In the periodic table, the halogens or the group 17 elements have the most positive electron affinity. Therefore, electron affinity increases from left to right of the periodic table and decreases down the group.
J.J Thompson - an English physicist who helped revolutionize the knowledge of atomic structure by his discovery of the electron (1897).
Pauli Exclusion Principle - only a maximum of two electrons can occupy an orbital and they must have opposite spins to minimize repulsion between them.
Schrodinger’s Model - suggests a 90% probability of finding an electron in a region around the nucleus.
Octet Rule - states that atoms are most stable when their valence shells are filled with eight electrons (noble gases has 8 valence electrons).
ELECTRON CONFIGURATION
ELECTRON CONFIGURATION of an element describes how electrons are distributed in its atomic orbitals.
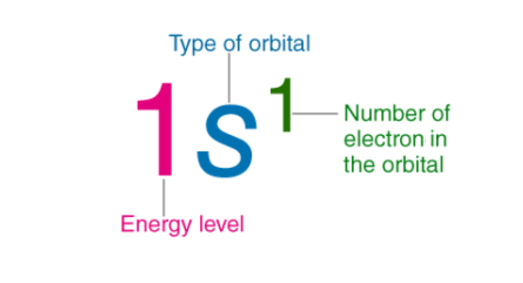
In such cases, an abbreviated or condensed notation may be used instead of the standard notation. In the abbreviated notation, the sequence of completely filled subshells that correspond to the electronic configuration of a noble gas is replaced with the symbol of that noble gas in square brackets.

LEWIS ELECTRON DOT STRUCTURES
LEWIS DOT STRUCTURES reflect the electronic structures of the elements, including how the electrons are paired.
Lewis Structure Examples
1. Lewis Structure of CO2
The central atom of this molecule is carbon. Oxygen contains 6 valence electrons which form 2 lone pairs. Since it is bonded to only one carbon atom, it must form a double bond.
Carbon contains four valence electrons, resulting in zero lone pairs. Therefore, it is doubly bonded to each oxygen atom.
