Topic 5 Solids, Liquids and gases
Density and pressure
Density is mass per unit volume
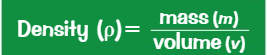
Density: Refers to how much mass is in a given volume of a substance.
Factors: Density depends on the substance's material, not its size or shape.
Finding Density: Measure an object's mass and volume to calculate its density.
Methods: For irregular solids, use water displacement in a measuring cylinder to find volume.


Pressure: The force applied over a surface area; expressed in pascals (Pa).
Effect of Area: Greater area results in lower pressure; pressure acts equally in all directions.
Depth in Liquids: Pressure increases with depth due to weight of the liquid above.
Pressure Difference: In liquids and gases, it depends on height difference and density.
Change of state
States of Matter: Matter exists as solids, liquids, or gases, differing in particle arrangement and energy.
Solids: Particles are closely packed and vibrate around fixed positions.
Liquids: Particles are less tightly packed, allowing movement in random directions at moderate speeds.
Gases: Particles have high energy, moving freely and rapidly.
Energy Transfer: Energy in matter is held by particles; heating increases energy, while cooling decreases it.
Temperature Changes: Changes in state (like melting or freezing) occur at consistent temperatures.
Evaporation
Evaporation Process: Evaporation occurs when particles transition from a liquid to a gas at temperatures lower than the boiling point. Not all particles escape; only those with sufficient energy can break free from the liquid's surface.
Kinetic Energy: The particles that escape are typically the fastest, possessing enough kinetic energy to overcome the attractive forces holding them in the liquid. This energy allows them to overcome the intermolecular forces and enter the gas phase.
Impact on Remaining Liquid: When these high-energy particles leave, the average kinetic energy of the remaining particles decreases. This leads to a reduction in temperature of the liquid, as temperature is a measure of the average kinetic energy of particles.
Cooling Effect: This cooling effect is significant; for example, when you sweat during exercise, the evaporation of sweat from your skin helps lower your body temperature, providing a natural cooling mechanism.
Specific heat capacity
Heating a substance increases the thermal energy store.
Temperature is a way of measuring average internal heat
Energy Storage: Materials that absorb a lot of energy to heat up also release significant energy when cooling down.
Key Relationships: Understanding the connections between mass, specific heat capacity, and temperature change is essential for energy transfer calculations.