Reaction of transition metal ions with sodium hydroxide
If you add sodium hydroxide solution to most transition metal ions, you will get a coloured precipitate.
If the precipitate dissolves on addition of excess sodium hydroxide solution, then the transition metal ion is amphoteric.
An amphoteric species can react as both an acid and a base.
Here is a table of the reactions you need to know:
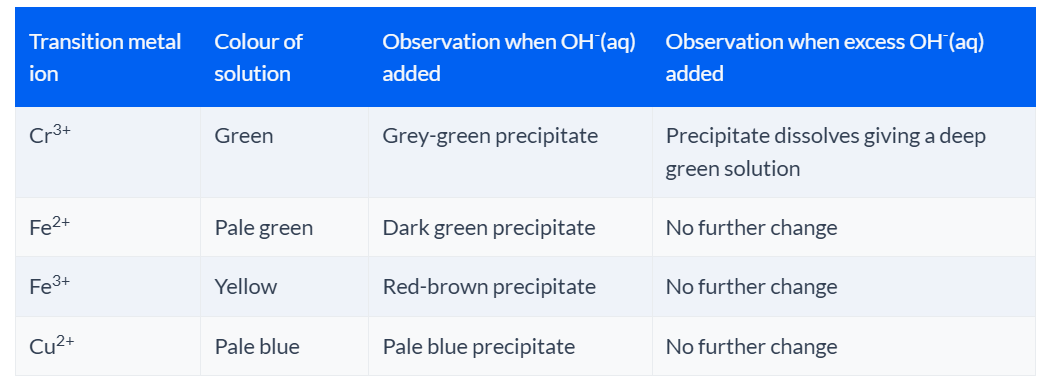
The ionic equation for the reaction of Cr3+ are:
Cr3+(aq) +3OH−(aq) → Cr(OH)3 (s)
Cr(OH)3(s) + 3OH−(aq)→ [Cr(OH)6]3−(aq)