Unit 5 Review Notes on Kinetics
Unit 5 Review: Kinetics
Overview of Kinetics
Kinetics is the study of rates of reaction.
Topics covered in this unit include:
Reaction rates
Rate laws
Concentration changes over time
Elementary reactions
Collision model
Reaction energy profiles
Reaction mechanisms and their relationship to rate laws
Steady-state approximation
Multistep reaction energy profiles
Catalysis
1. Rates of Reaction
Definition of a Rate:
A rate measures how some property changes over time, akin to how speed measures distance traveled per unit time.
Reaction rates measure the change in concentration per unit time, specifically how much reactant is consumed or how much product is produced, usually measured in seconds.
Measuring Reaction Rates:
Strategies for measuring reaction rates include:
Measuring changes in gas pressure when a product is a gas.
Observing changes in light absorption if a reactant forms a colored product from a colorless solution.
Rates of reaction are expressed as:
where [C] is concentration and ( \Delta ) denotes change.
Positive and Negative Values:
For a reactant, as concentration decreases, the rate is negative; for a product forming, the rate is positive.
Stoichiometric Coefficients:
Example: Combustion of ammonia.
Coefficients relate rates: for reactants (negative) and products (positive).
Types of Rates:
Instantaneous Rate: The rate at a specific moment.
Initial Rate: The rate at the very beginning of the reaction.
Calculating Instantaneous Rate: Approximate by averaging over a very short time or using the slope of the tangent line on the graph of concentration vs. time.
2. Factors Influencing Reaction Rates
Factors that influence reaction rates include:
Reactant concentration: Higher concentration leads to more collisions.
Temperature: Higher temperatures increase kinetic energy, thus increasing reaction rates.
Surface area: Increased surface area facilitates more collisions.
Presence of a catalyst: Lowers activation energy, increasing reaction rate.
3. Rate Laws
Definition: A rate law expresses the relationship between the rate of a chemical reaction and the concentration of its reactants.
General form:
where k is the rate constant, [A] and [B] are molar concentrations of reactants, and m and n are reaction orders determined experimentally.Importance of Reaction Order:
Reaction orders are generally not related to stoichiometric coefficients in a balanced equation and must be found experimentally.
Overall reaction order: Sum of individual orders: .
Example of Rate Laws:
Nitrogen dioxide and carbon monoxide reacting:
If reaction is second order in NO2 and zero order in CO, then:
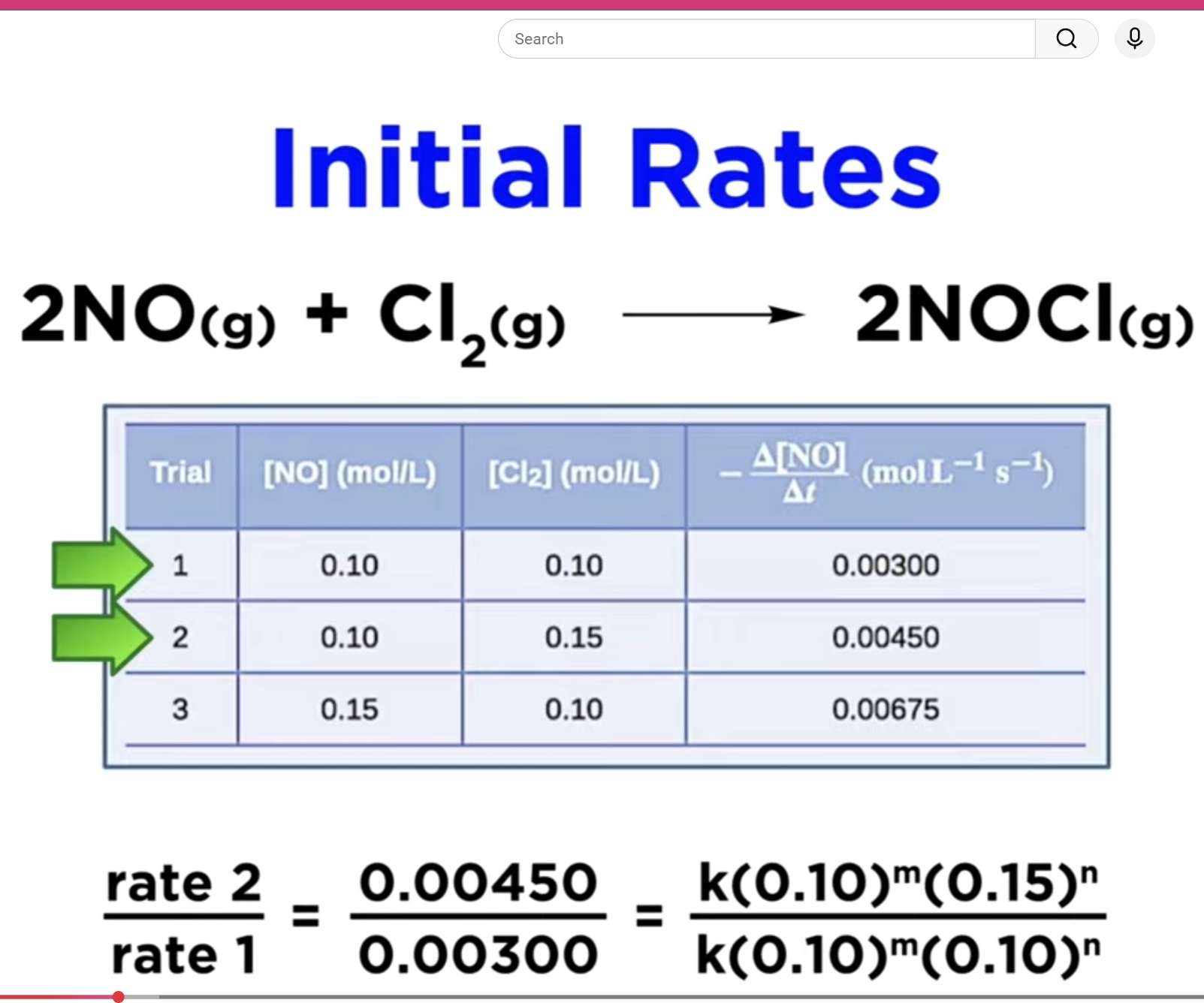
4. Determining Reaction Order via Initial Rates
Initial Rates Method:
Compare rate data with varying concentrations from trials to determine reaction orders.
Example with nitrogen monoxide and ozone:
Changing ozone concentration led to a doubled rate, indicating the reaction is first order with respect to ozone.

Demonstrating for NO, if initial concentration is doubled, rate doubles, indicating first order in NO.
Conclusion: Overall rate law would be second order overall.

5. Integrated Rate Laws
Integrated Rate Laws: Useful for analyzing concentration vs. time.
Zero-order reaction:
, slope is the opposite of the rate constantFirst-order reaction:
Second-order reaction:
slope is the rate constant
Graphical representation helps identify reaction order. Suitable plots yield straight lines, indicating the order:
Zero-order: Concentration vs. time
First-order: Natural log of concentration vs. time
Second-order: Inverse concentration vs. time
6. Collision Theory
Postulates of Collision Theory:
The rate of reaction is proportional to the number of collisions per unit time.
Molecules must collide in the correct orientation.
Collisions must have sufficient energy to overcome activation energy.
Activation Energy: The energy required to form the transition state in a reaction.
7. Reaction Mechanisms
Definition: The pathway via which a reaction occurs, often comprising multiple elementary steps.
Example of ozone decomposition:
Ozone loses one oxygen atom to become dioxygen.
The oxygen atom reacts with ozone to generate more dioxygen.
Molecularity:
The number of reacting species in an elementary step (Unimolecular, Bimolecular, Termolecular).
Rate-Determining Step: The slowest step in a reaction mechanism, which controls the overall rate of the reaction.
Steady-State Approximation: Assumes the concentration of intermediates remains approximately constant throughout a reaction.
8. Reaction Energy Profiles
Energy Diagrams: Display potential energy changes over the course of a reaction.
Horizontal axis: Reaction coordinate (time)
Vertical axis: Potential energy
Key Terms:
Activation energy: Energy from reactants to the transition state.
Endothermic and Exothermic: Determined by the energy state of products relative to reactants.
9. Catalysis
Definition: A catalyst is a substance that increases reaction rate by lowering activation energy but is not consumed in the process.
Types of Catalysis:
Acid-base catalysis: Proton transfer enhances reactivity.
Surface catalysis: Interactions on solid catalysts promote reactions.
Biological catalysis: Enzymes catalyze biochemical reactions.
10. Arrhenius Equation
Relates activation energy to rate constant:
where A is the frequency factor, E_a is activation energy, R is the gas constant, and T is temperature in Kelvin.
11. Nuclear Reactions and Half-life
Nuclear Reactions: Involve changes in the nucleus, altering identity of atoms.
Nuclide symbols: Represent nuclear particles (mass number, atomic number).
Half-life: Time required for half of a radioactive sample to decay.
For first-order processes, it is:
Half-lives are logarithmic; the amount remaining is not reduced by a constant proportion but halves each period.
12. Conclusion
Understanding these concepts allows for the analysis and prediction of reaction behavior in various scenarios, laying groundwork for future topics in thermodynamics.