History of the atom
Start of 19th century, John Dalton described atoms as solid spheres, different spheres made up different elements
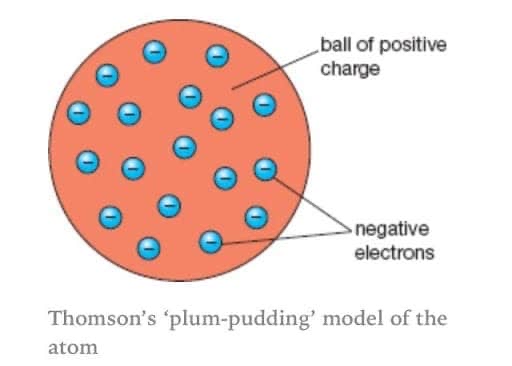
Plum Pudding
In 1897 JJ Thomson concluded that they weren’t solid spheres
His measurements of charge and mass showed they had to contain even smaller negatively charged particles - electrons.
This was called the plum pudding model.
Nuclear model
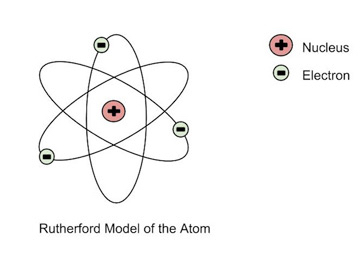
In 1909 Rutherford and his student Marsden conducted an alpha particle scattering experiment.
Fired positively charged alpha particles at an extremely thin sheet of gold.
More particles deflected than expected and lots in the middle, suggested there was a positively charged nucleus and electrons orbiting nucleus
Called nuclear model
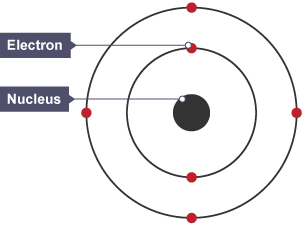
Bohrs Nuclear model
Electrons in a cloud around nucleus would be attracted to positive charge and atom would collapse, proved Rutherford wrong.
1914 Bohr suggested that electrons orbiting nucleus in fixed shells at specific distances from nucleus
James Chadwick
Discovered neutron