Carbonyl Compounds
Carbonyl groups
Aspects of the Carbonyl C=O Bond
The C=O group the most important functional group in organic chemistry
An extremely large Class of Compounds (Carbonyl Compounds) contains the C=O group
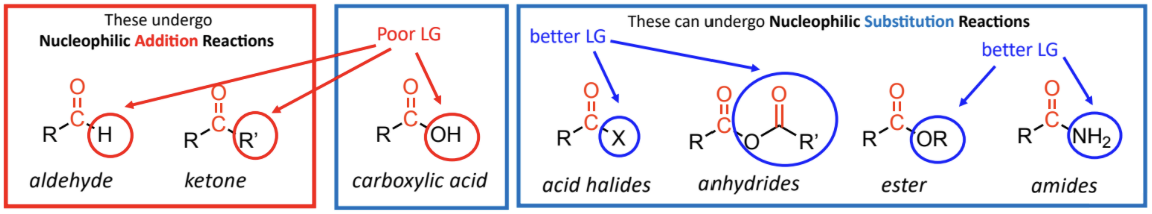
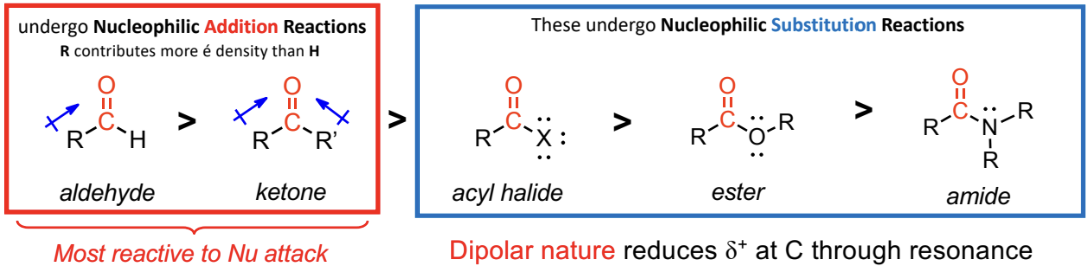
aldehyde and ketones do not have good leaving groups
only undergo nucleophilic addition reactions
acid halides, anhydrides, esters and amides undergo nucleophilic substitution reactions - have good leaving groups.
Structure and Reactivity
Carbonyl compounds show sp2 hybridization leading to planar geometry (120° bond angles).
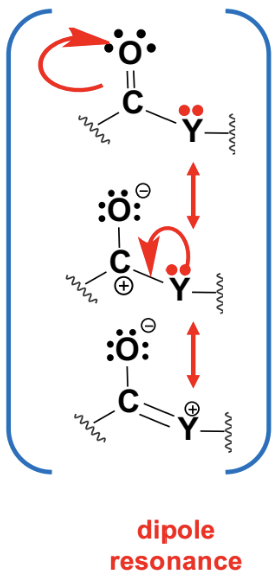
The C=O bond acts as an electrophile due to electronic characteristics.
C=O is the central functional group in many biological molecules.
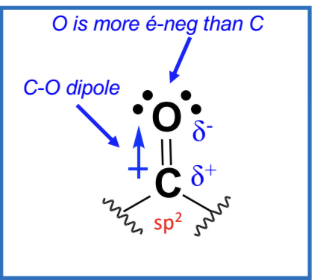
The carbon in the carbonyl group is electrophilic due to the polarization of the C=O bond.
Oxygen is more electronegative, creating a dipole moment.
This makes the carbon susceptible to nucleophilic attack.
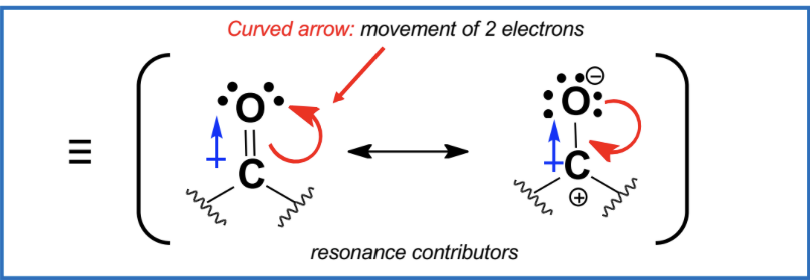
nucleophile can substitute leaving group - circled blue
Reactions with Carbonyl Groups
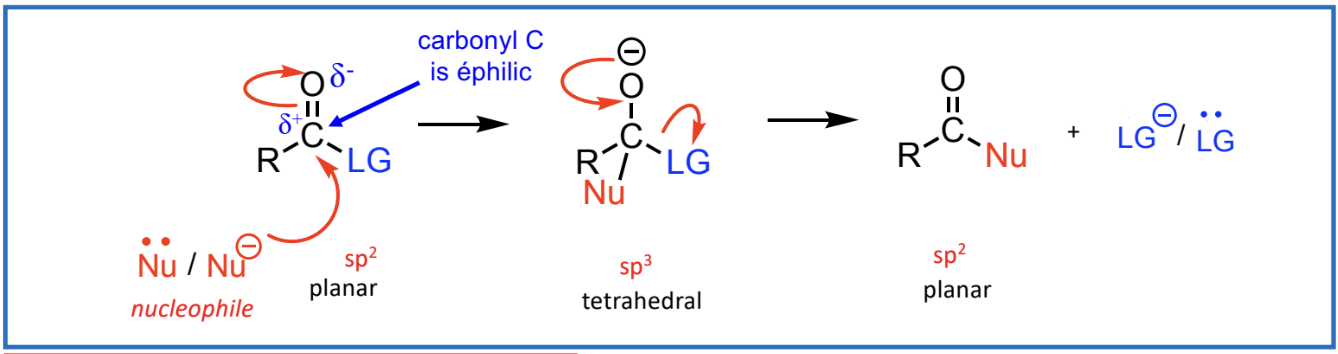
Nucleophilic Substitution Reactions (Nucleophile attacks replace a leaving group):
Requires a good leaving group (LG).
Example of good LCs: -OR (alkoxy) > -OH (hydroxy).
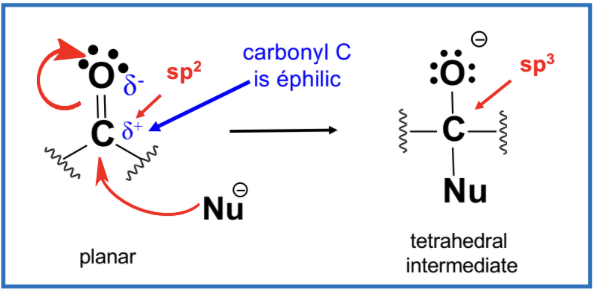
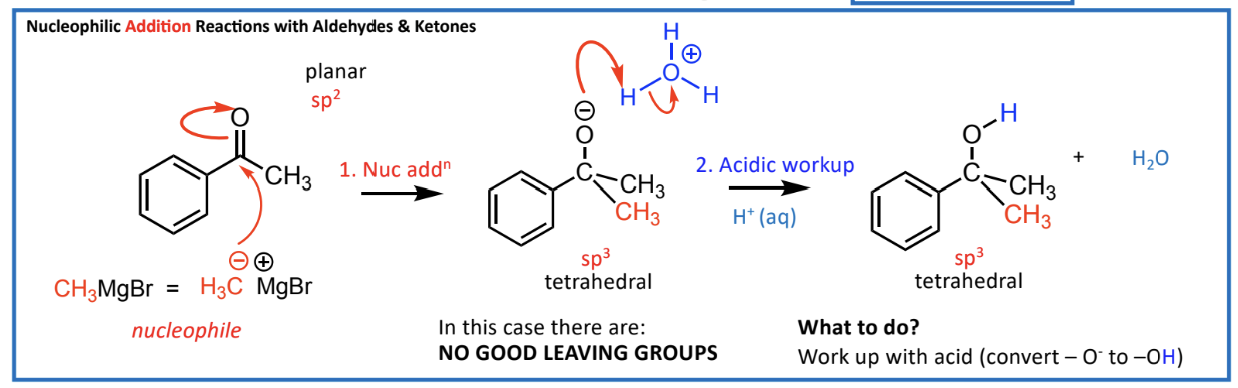
Nucleophilic Addition Reactions (Addition across the C=O):
Example: If there is no good leaving group available, an acid workup is necessary post-reaction.
for nucleophilic attacks, aldehyde more reactive than ketone
Carbonyl Group as Electrophile
Nucleophilic Substitution Reactions
need a good leaving group (LG)
an electronegative subsbtuent that can act as a LG (eg. – OR and – Cl)
aldehydes & ketones don’t have a good LG (– R and – H are poor LG) so NO nucleophilic subsbtubon reaction
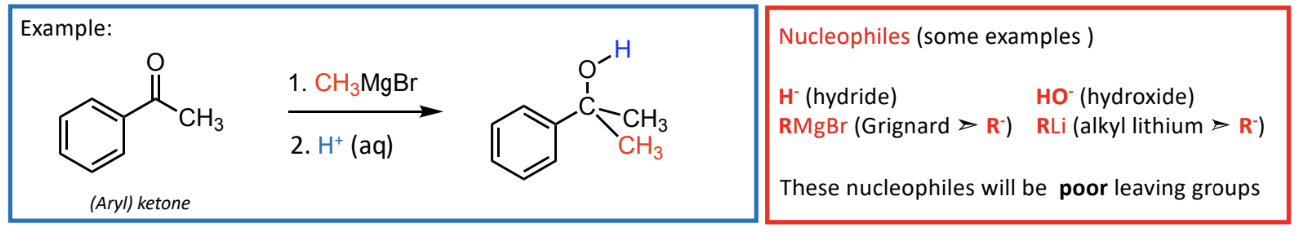
Nucleophiles examples
H- (hydride)
HO- (hydroxide)
RMgBr (Grignard ➣ R-)
)RLi (alkyl lithium ➣ R-
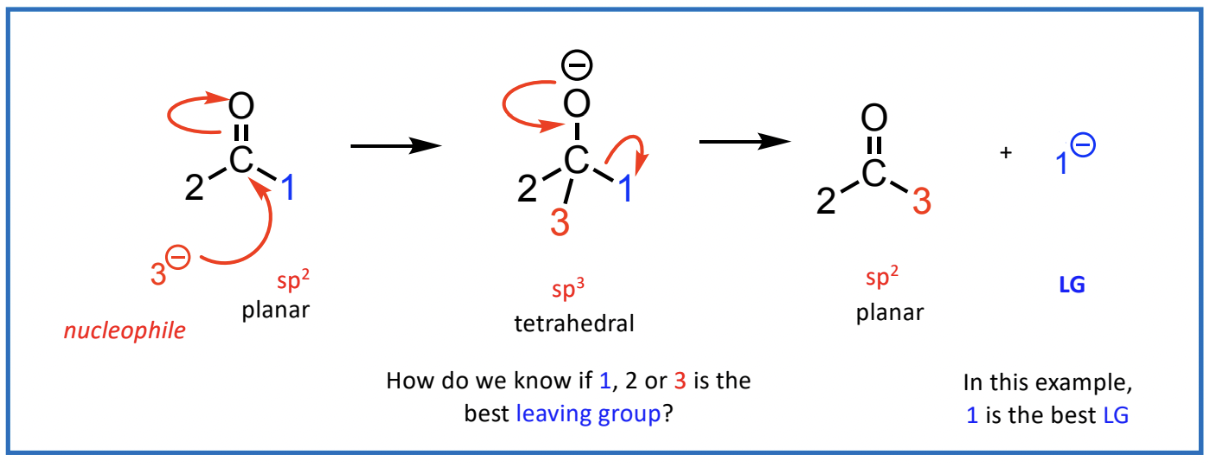
Identifying the Best Leaving Group
The better the leaving group, the easier it can exit the molecule during a reaction.
for this reaction to proceed to completion, a group has to leave from the sp3 (tetrahedral) structure
A Leaving Group needs to be a stable sink for electron density
Leaving group form anions such as Cl-, RO-, and RCO2- that can be expelled from molecules taking their negative charge with them.
rules to identify best leaving group
neutral molecules or stable anion
weaker basic strength of group, better leaving group
low pka value of its CA (~<13) - stable
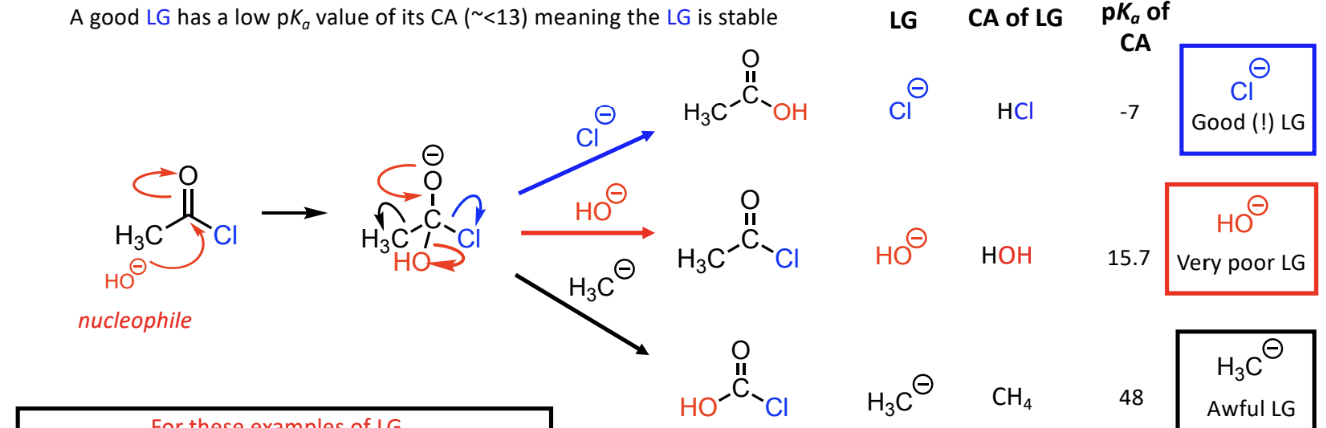
best = Cl-, poor =HO-, awful = H3C-
Comparison: Alkoxy (-OR) is generally a better leaving group than hydroxyl (-OH).
Hydroxyl groups can turn into bad leaving groups under strong base conditions due to deprotonation which leads to less stability.
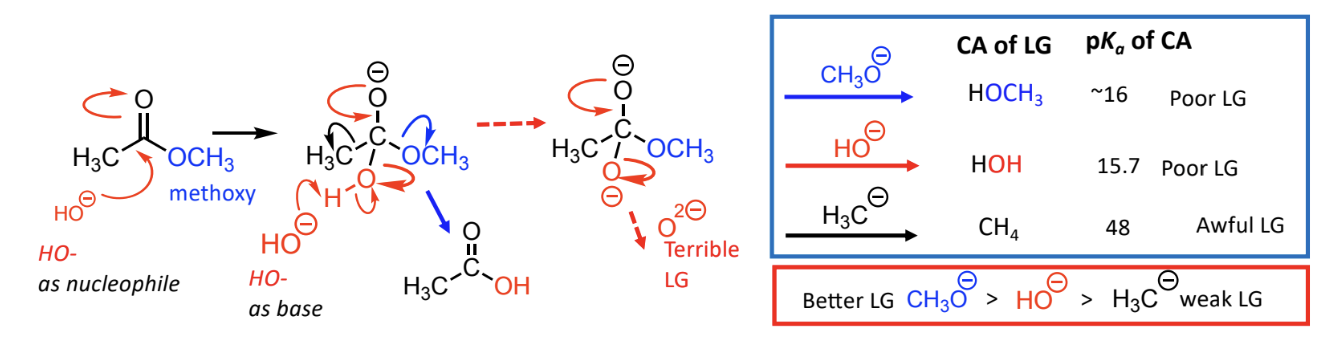
halide is the best leaving group
What If There Isn’t a Good Leaving Group?
If nucleophilic substitution isn't feasible:
Conduct nucleophilic addition.
Follow-up with an acidic workup to regain reactivity (convert O to -OH).
Functional Groups in Organic Chemistry
Types of Functional Groups
Hydrocarbons: Alkanes, alkenes, alkynes
Simple Oxygen Compounds: Alcohols, ethers, epoxides
Heteroatomics Include: Halogens, nitrogen, sulfur
Carbonyl Compounds: Aldehyde, ketone, carboxylic acid, esters, anhydrides, amides
**Name Relationships: **
Aldehydes (-al)
Ketones (-one)
Carboxylic acids (-oic acid)
Esters (-yl-oate)
Amides (-amide)
Characteristics of Carbonyl Groups
Nucleophilic Attack
Carbonyl carbons are susceptible to nucleophilic attacks leading to new reaction pathways forming tetrahedral intermediates.
Strong Leaving Groups
Good leaving groups tend to have low pKa values (weaker bases = better leaving group).
Examples of leaving groups ranked: Cl > HO > H3C (better to worse).
Nucleophilic Substitution Challenges
For reactions to progress, leaving groups must be identified and stability must be ensured, otherwise, nucleophilic addition will occur instead.
Hydroxyl groups can be poor leaving groups unless transformed in acidic workup conditions.