Molecular Dipoles & MO Theory [Lecture 17]
Determining Molecular Polarity
Molecular (net) dipole:
Sum of all bond dipoles in a molecule via vector addition.
Bond Dipole (μ):
A measure representing the separation of charges due to electronegativity differences between atoms, indicated by δ+ (partial positive) and δ– (partial negative) charges.
Polar Molecule:
Molecule with non-canceling bond dipoles, resulting in an overall molecular dipole and uneven electron density.
Nonpolar Molecule:
All bond dipoles cancel out, leading to no net dipole.
Vector Addition of Bond Dipoles
Vector Definition:
A quantity with both magnitude and direction.
Adding Vectors:
Use methods like constructing a parallelogram to visualize how bond dipoles combine.
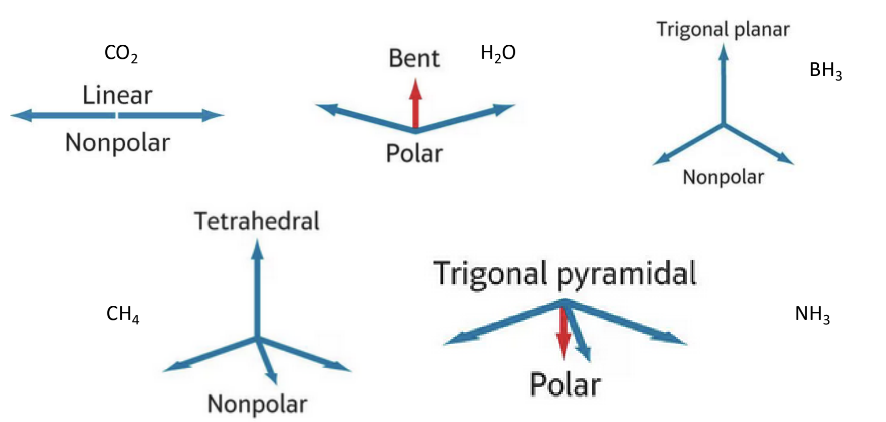
Common Molecular Shapes and Their Polarity
Linear (CO2):
Nonpolar
Trigonal Planar (BH3):
Nonpolar
Bent (H₂O):
Polar
Tetrahedral (CH4):
Nonpolar
Trigonal Pyramidal (NH3):
Polar
Steps to Determine Molecular Polarity
Draw Lewis Structure:
Identify molecular geometry.
Identify Polar Bonds:
Determine the presence of polar bonds and their direction.
Assess Bond Dipole Addition:
Check if polar bonds add to create a new dipole moment.
Symmetric Dipoles: Nonpolar
Asymmetric Dipoles: Polar
Introduction to Molecular Orbital Theory (MO Theory)
Description:
MO Theory provides a detailed view of how atoms bond by treating electrons as waves and utilizing molecular orbitals that span the entire molecule.
Key Principles:
Linear Combination of Atomic Orbitals (LCAO):
Wavefunctions of atomic orbitals combine to form molecular orbitals through interference.
Types of Interference:
Constructive: Adds up leads to bonding orbitals, (lower energy).
Destructive: Cancels out leads to antibonding orbitals, (higher energy).
Constructing Molecular Orbital Diagrams
Molecular Orbitals:
Number of MOs formed equals the number of atomic orbitals combined.
Distinction between bonding (lower energy) and antibonding (higher energy) orbitals.
Bond Order Calculation:
Bond Order = 1/2 (Number of electrons in bonding MOs - Number of electrons in antibonding MOs)
Electron Configuration Rules:
Place electrons in MOs starting from the lowest energy. No more than two electrons per MO, with opposite spins.