amino acids 1
Glycine is the only non-chiral amino acid
Alpha carbon must’ve COOH and NH2 bonded to it
Proteins contain only L-isoforms. They’re amphoteric
Major 4 elemtents in amino acids, C H O N (S)
Structures of proteins
primary; the straight linear chain
seoncdary: i. alpha helix; bacbone lies inside the helix and side chains project outwards. The Oxygen from Amino Acid #1 reaches out and "grabs" the Hydrogen from Amino Acid #4 (specifically, the 4th one ahead of it).
ii. beta pleated sheets;chains lying side by side, their hydrogen bonds are oriented towards the long axis of the chaintertiary: van der waals, hydrogen bond and ionic bond, contain secondary structures
quartenary: subunits can be made of disulfide covalent bonds.
Hemoglobin molecule,consists of four globin chains joined by noncovalent bonds
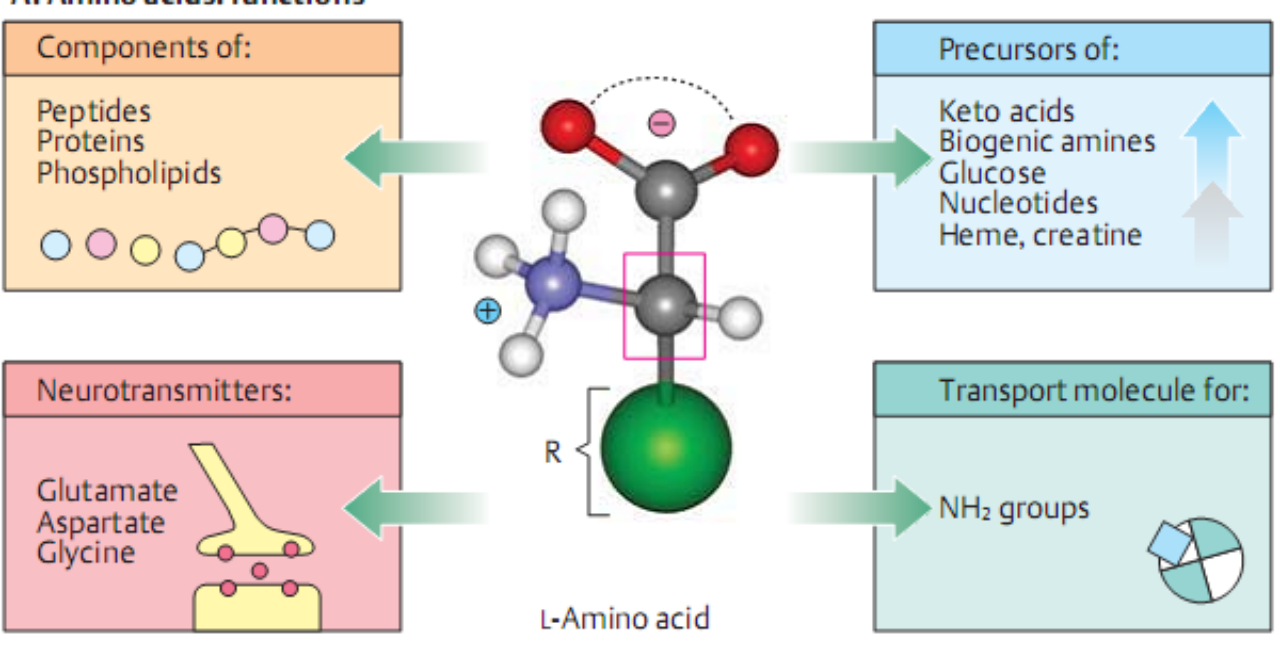
Functions
precursors for other metabolites; egs purines and pyrimiedine bases
Some non-proteinogenic amino acids function as intermediates in the synthesis and breakdown of proteinogenic amino acids and the urea cycle
Components of lipids; serine in phopholipids,glycine in bile salts
As neurotransmitters, hormones and chemical mediators
Properties
colorless, non-volatile, crystalline solids and melt at temp above 200 degrees celsius, varied structure,chirality
Ionizable properties
COOH =>COO- + H+ (proton donor)
-NH2 + H+ =>NH3+ (proton acceptor)
Ubiquitin-Proteasome System (UPS) destroys damged proteins
Digestion of proteins
trypsinogen=>trypsin via enzyme enteropeptidase
Trypsin cleaves:
Chymotrypsinogen to chymotrypsin
Proelastase to elastase
Procarboxypeptidase to carboxypeptidase
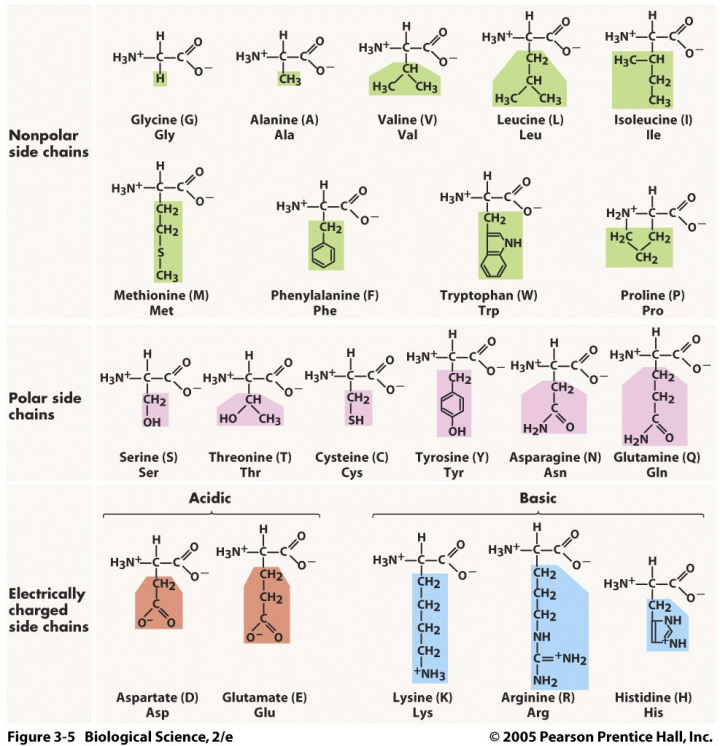
CLASSIFICATION OF AMINO ACIDS
They may be classified
1into five groups based on the reactivities of their R groups
2metabolically on their catabolic products
3On weather they are essential or non essential
4On the extra-functional groups available
4 major rxns of aa
transamination,transdeamination, oxidative deamination, decarboxylation
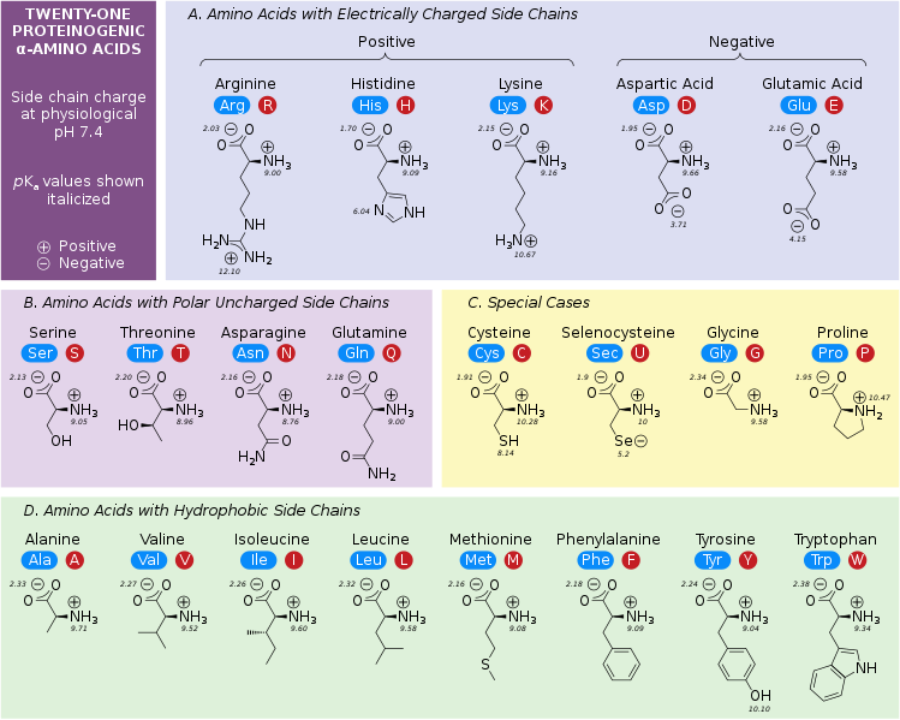
Table of the 20 Standard Amino Acids
Amino Acid | 3-Letter | 1-Letter | Side Chain Property |
|---|---|---|---|
Glycine | Gly | G | Nonpolar, aliphatic |
Alanine | Ala | A | Nonpolar, aliphatic |
Valine | Val | V | Nonpolar, aliphatic |
Leucine | Leu | L | Nonpolar, aliphatic |
Isoleucine | Ile | I | Nonpolar, aliphatic |
Methionine | Met | M | Nonpolar, aliphatic |
Proline | Pro | P | Nonpolar, aliphatic |
Phenylalanine | Phe | F | Aromatic |
Tyrosine | Tyr | Y | Aromatic |
Tryptophan | Trp | W | Aromatic |
Serine | Ser | S | Polar, uncharged |
Threonine | Thr | T | Polar, uncharged |
Cysteine | Cys | C | Polar, uncharged |
Asparagine | Asn | N | Polar, uncharged |
Glutamine | Gln | Q | Polar, uncharged |
Lysine | Lys | K | Positively charged (Basic) |
Arginine | Arg | R | Positively charged (Basic) |
Histidine | His | H | Positively charged (Basic) |
Aspartic Acid | Asp | D | Negatively charged (Acidic) |
Glutamic Acid | Glu | E | Negatively charged (Acidic) |
Non-Polar | Polar (Uncharged) | Electrically Charged |
|---|---|---|
Glycine | Serine | Aspartic Acid (Acidic) |
Alanine | Threonine | Glutamic Acid (Acidic) |
Valine | Cysteine | Lysine (Basic) |
Leucine | Tyrosine | Arginine (Basic) |
Isoleucine | Asparagine | Histidine (Basic) |
Methionine | Glutamine | |
Phenylalanine | ||
Tryptophan | ||
Proline |
mnemonic for non polar GAVI Liked My Fat Pro Twin
G - Glycine
A - Alanine
V - Valine
I - Isoleucine
L - Leucine
M - Methionine
F - Phenylalanine (the "Ph" sounds like F)
P - Proline
T-Tryptophan
the first five thats G,A,V,I and L are aliphatic
mnemonic for polar Santa's Team Crafts Yearly New Quilts
S – Serine
T – Threonine
C – Cysteine
Y – Tyrosine (The letter Y)
N – Asparagine (The letter N)
Q – Glutamine (The letter Q — "Q-tamine")
mnemonic for acidic and neg charged is Dont Eat
D-Aspartate
E-Glutamate
mnemonic for basic and pos charged is King Roy’s Home
K-Lysine R-Arginine H-Histidine
They’ve an extra NH in their structure
NB:1.Phenylalanine and tryptophan are aromatic and non polar
2.Tyrosine is aromatic
Aromatic
All very hydrophobic
All contain aromatic group
Absorb UV at 280 nm
Phenylalanine (Phe, F)
Tyrosine (Tyr, Y) – -OH ionizable (pKa = 10.5), H-Bonding
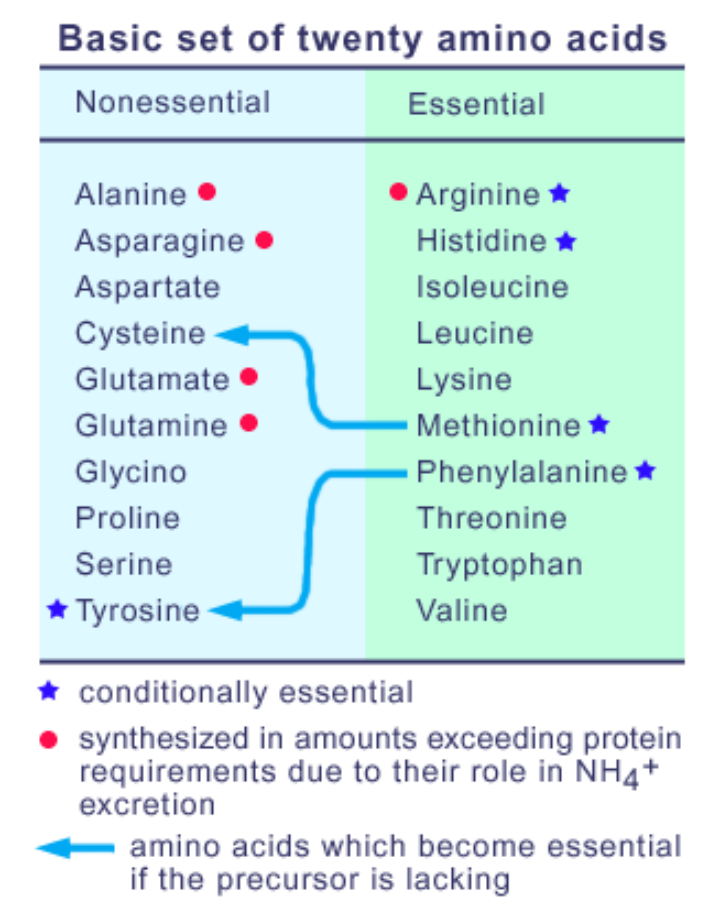
Conditionally essential
(i) ARG: can be made, but not enough
(ii) HIS: controversial (essential for growth in children)
(iii) PHE essential, TYR can be made from PHE but when enzyme is missing, PKU. then PHE not equal to TYR; Therefore TYR is essential
(iv) MET& CYS; Similarly, if MET not essential CYS then CYS essential
1amino acids with aliphatic side chains : These are monoamino monocarboxylic acids. This group consists of the most simple amino acids—glycine, alanine, valine, leucine and isoleucine. The last three amino acids (Leu, Ile, Val) contain branched aliphatic side chains, hence they are referred to as branched chain amino acids.
2Hydroxyl group containing amino acids :Serine, threonine and tyrosine are hydroxyl group containing amino acids. Tyrosine—being aromatic in nature—is usually considered under aromatic amino acids.
3Sulfur containing amino acids : Cysteine with sulfhydryl group and methionine with thioether group are the two amino acids incorporated during the course of protein synthesis. Cystine, another important nt sulfur containing amino acid, is formed by condensation of two molecules of cysteine.
4Acidic amino acids and their amides : Aspartic acid and glutamic acids are dicarboxylic monoamino acids while asparagine and glutamine are their respective amide derivatives. All these four amino acids possess distinct codons for their incorporation into proteins.
5Basic amino acids : The three amino acids lysine, arginine (with guanidino group) and histidine (with imidazole ring) are dibasic monocarboxylic acids. They are highly basic in character.
6Aromatic amino acids :
GROUPING BASED ON CATABOLIC INTERMEDIATES
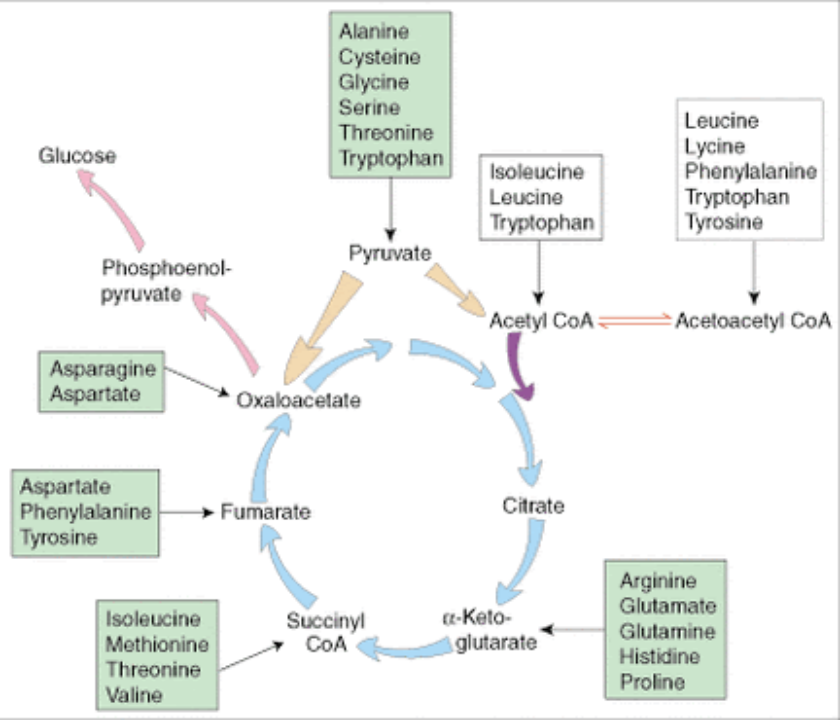
1.Amino acids may also be classified into glucogenic and ketogenic types depending on the metabolic intermediates they generate on catabolism
Glucogenic amino acids are catabolised to pyruvate, succinyl CoA, α-ketoglutarate, fumarate or oxaloacetate
Examples are Alanine, Asparagine, Aspartate, Arginine, Cysteine, Glutamine, Histidine, Methionine, Proline, Threonine and Valine
2.Ketogenic amino acids are catabolised to acetyl CoA or acetoacetyl CoA
Examples of purely ketogenic amino acids are Leucine and Lysine
Glucogenic and ketogenic : Isoleucine, Phenylalanine, Tryptophan and Tyrosine
Unusual amino acids
Result from modifications of the common amino acids (hydroxyproline, ornithine, norleucine)
Peptides
Oligopeptides − condensation of 2 – 10 AA units
Polypeptides − condensation of 11 – 100 AA units
Proteins − more than 100 AA units
GENERAL REACTIONS OF AMINO ACIDS
Decarboxylation
Amino acids are decarboxylated to amines. Eg. Histidine to Histamine
Histamine stimulates gastric secretion and involved in immune response (itching)
5 hydroxy tryptophan → hydroxy tryptamine + CO2
Serotonin - neurotransmitter mediating mood, memory..

DOPA → Dopamine + CO2
Dopamine is an intermediate in the formation of adrenaline, a vasoconstrictor
TRANSAMINATION
Transamination involves the transfer of an amino group from an amino acid to a keto-acid to form the corresponding amino acid
The reaction is catalyzed by amino transferases which require pyridoxal phosphate (PLP), a derivative of vitamin B6 as a prosthetic group. Its the carrier at the active site
PLP undergoes reversible transformation between the aldehyde form and the aminated form, pyridoxamine phosphate
Examples of aminotransferases are AST and ALT
What is their importance/role as biomarkers?

In mammals, all alpha amino acids except lysine and threonine can be transaminated
Proline, which has a secondary amino group, cannot participate in PLP dependent reactions including transamination and decarboxylation
OXIDATIVE DEAMINATION
The oxidative deamination of glutamate occurs in the mitochondria of the liver yielding ammonia ions
L-Glutamate + H2O ↔ α-ketoglutarate + NH4+
This reaction is catalyzed by glutamate dehydrogenase which is a complex allosteric enzyme
glutamate dehydrogenase allosteric activators are GDP and ADP. its a link between catabolic and anabolic pathways, and is, therefore, ubiquitous in eukaryotes
GLDH role
Its activity increases when fuel is needed(low energy, high GDP and ADP)
The combined action of the aminotransferases and glutamate dehydrogenase is referred to as transdeamination
The coupling of oxidative deamination with transamination of L-glutamate creates an avenue for deaminating all amino acids