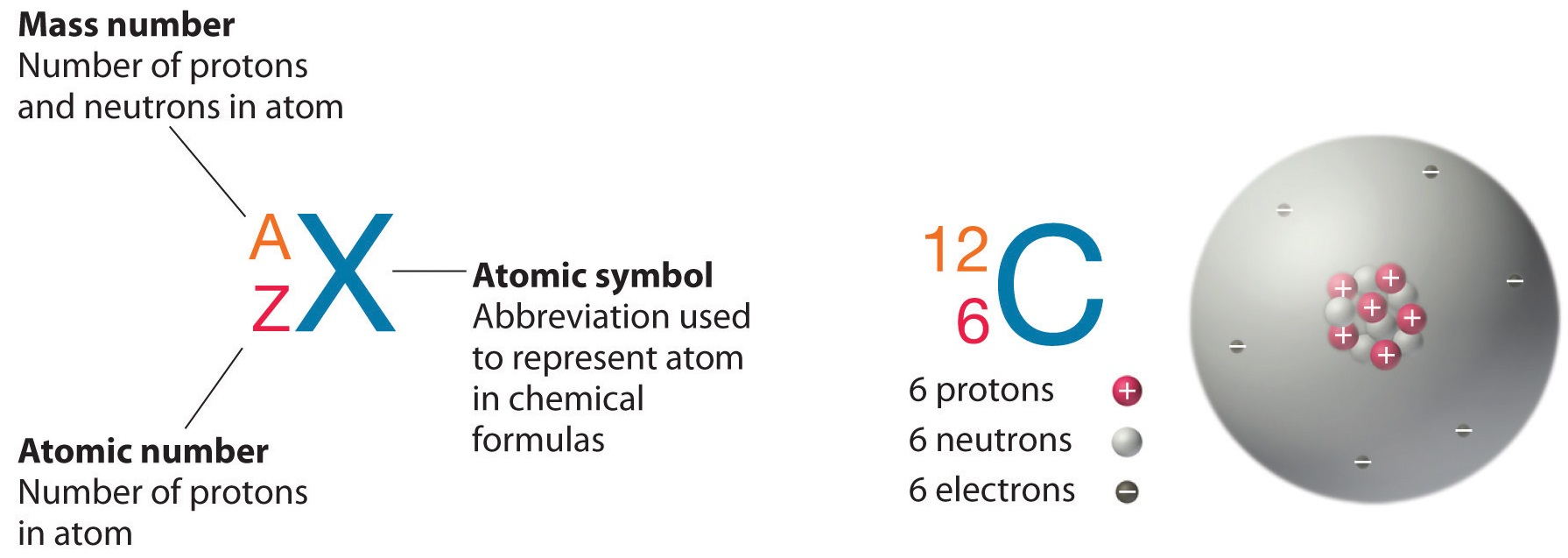
Atoms and Isotopes
The structure of an atoms
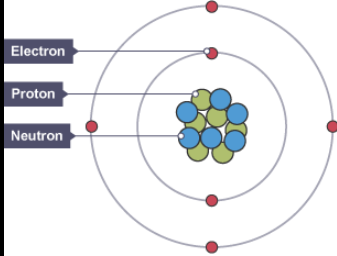
an atom is the smallest unit of matter, consisting of three particles:
protons (positive charge particles, in the nucleus),
neutrons (neutral, in the nucleus),
and electrons (negative charge, orbiting the nucleus).
The nucleus holds protons and neutrons, while electrons exist in energy levels, influencing the atom's chemical properties.
Ions:
An atom becomes a positive ion if it loses one or more electrons
An atom becomes a negative ion if it gains one or more electrons
Internal energy
The internal energy of a system is stored in the atoms and molecules that make up the system.
This energy comes from two things:
The kinetic energy of the particles (how fast they're moving)
The potential energy from the forces between them(solid particles would have less potential energy due to the strong force holding them in place so melting or boiling would increase potential energy)
So, internal energy is the total energy of all the particles inside the system.
Isotopes:
Similarities:
Charges – Usually No Difference
Isotopes are usually neutral — the number of electrons = protons, so the overall charge is zero.
Unless the atom is ionized, the charge stays the same across isotopes.
Ionization can happen to any isotope, but that's a separate process.
Same Element, Same Identity
Isotopes are atoms of the same element (same number of protons)
So they have the same atomic number and the same chemical symbol
Example: Carbon-12 and Carbon-14 are both carbon (C), with 6 protons
Differences:
Different Mass Numbers
Isotopes differ in the number of neutrons
This changes their mass number (protons + neutrons)
Shown in the isotope name or symbol