Ch5: Light and Matter: Reading Messages from the Cosmos
Light in Everyday Life
Our goals for learning:
How do we experience light?
→ Wave theory
Electromagnetism (E&M)
How do light and matter interact?
→ Quantum Mechanics
How do we experience light?
The warmth of sunlight tells us that light is a form of energy
We can measure the flow of energy in light in units of watts: 1 watt = 1 joule/s.
Note: [J]=[N*m]
Colors of Light
White light is made up of all the colors of the rainbow
How do light and matter interact?
Emission
Absorption
Transmission
Transparent objects transmit light.
Opaque objects block (absorb) light.
Reflection/scattering
Reflection and Scattering
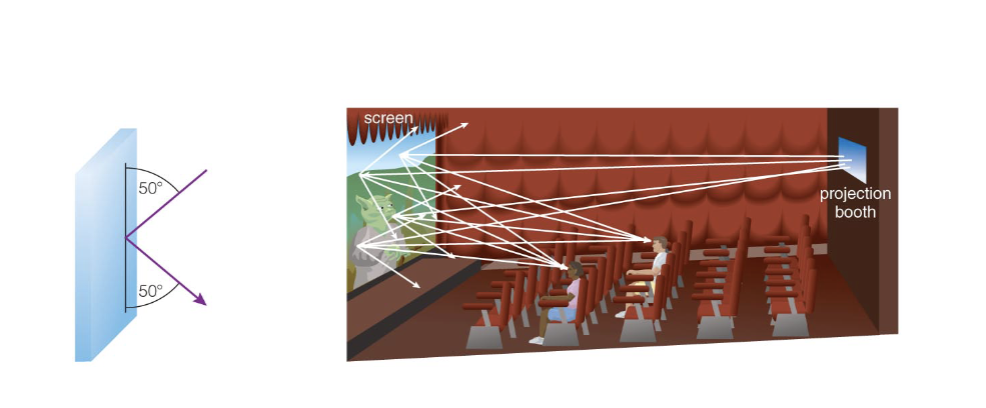
A mirror reflects light in a particular direction.
A projector screen scatters light in all directions.
Interactions of Light with Matter
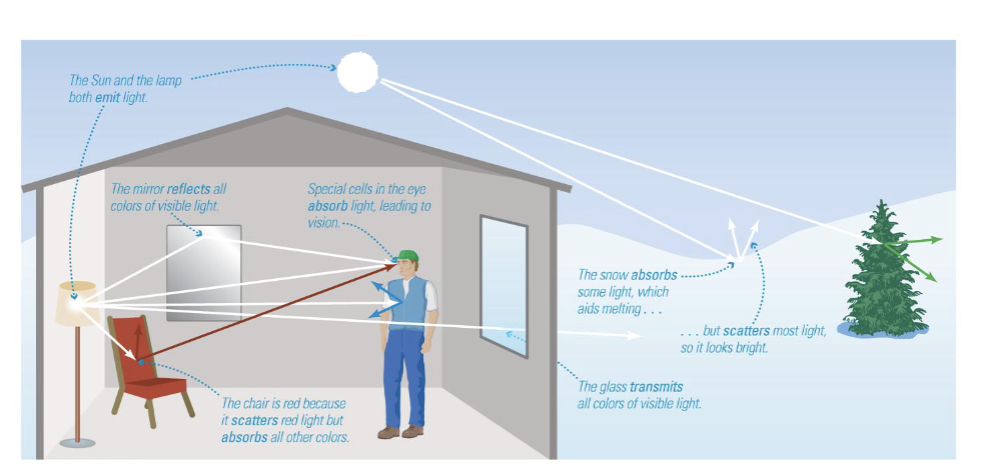
Interactions between light and matter determine the appearance of everything around us.
Thought Question
Why is rose red?
→ The rose reflects red light.
What have we learned?
How do we experience light?
Light is a form of energy
Light comes in many colors that combine to form white light
How do light and matter interact?
Matter can emit light, absorb light, transmit light, and reflect (or scatter) light,
Interactions between light and matter determine the appearance of everything we see.
Properties of Light
What is light?
Light is a small portion of Electromagnetic wave spectrum.
Can behave either like a wave or like a particle.
Particles of light are called photons.
Waves
A wave is a pattern of motion that can carry energy without carrying matter along with it.
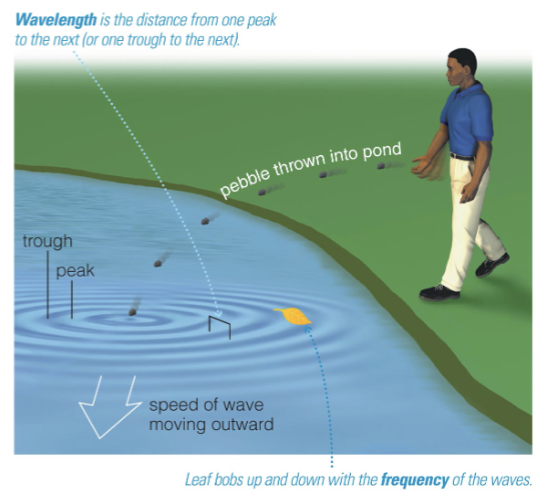
Properties of Waves
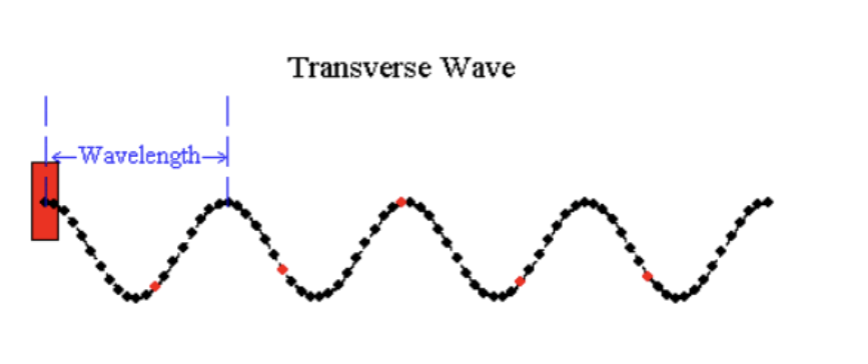
Wavelength is the distance between two wave peaks.
Frequency is the number of times per second that a wave vibrates up and down.
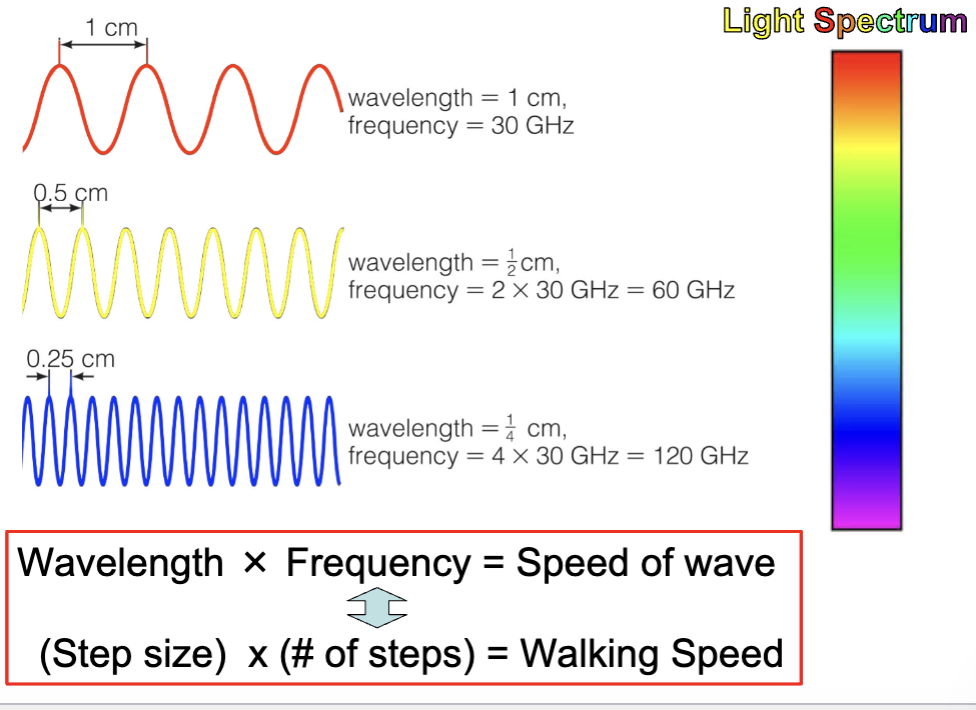
Wave speed = wavelength x frequency
Transverse & Longitudinal Waves

Light: Electromagnetic Waves
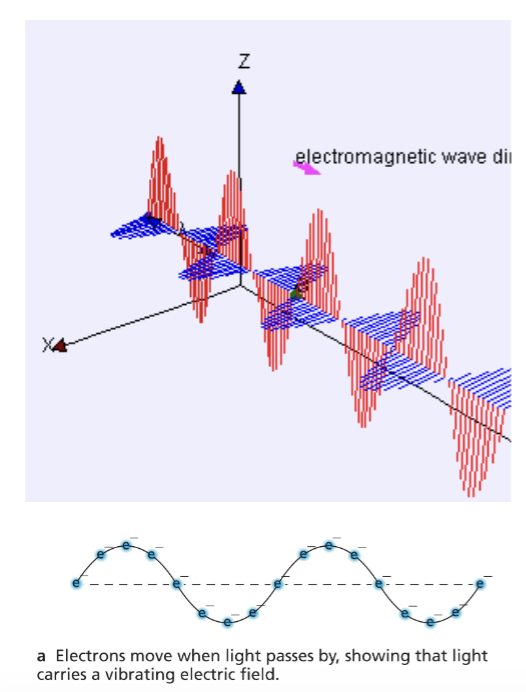
A light wave is a vibration of electric and magnetic fields.
EM wave is a transverse wave.
Interacts with charged particles.
Can travel in vacuum.
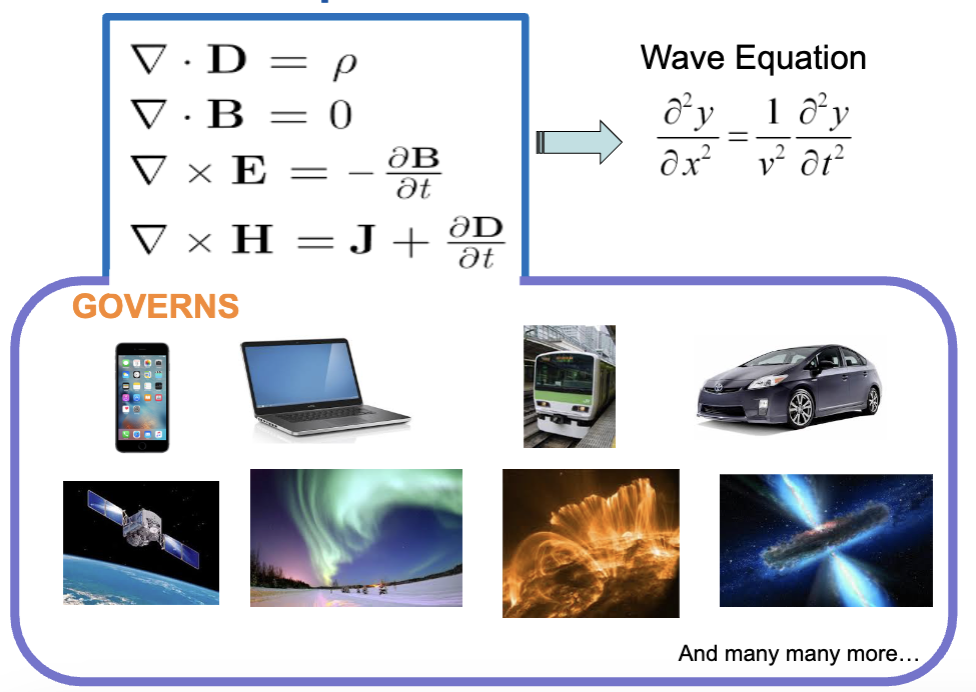
→ Maxwell’s Equation
Maxwell’s Equation
Wavelength and Frequency
Particles of Light
Massless (at rest) Particles of light are called photons. ← Quantum Mechanics
Each photon has a wavelength and a frequency
The energy of a photon depends on its frequency
Wavelength, Frequency, and Energy

What have we leaned?
What is light?
Light can behave like either a wave or a particle.
A light wave is a vibration of electric and magnetic fields.
Light waves have a wavelength and a frequency
Photons are particles of light.
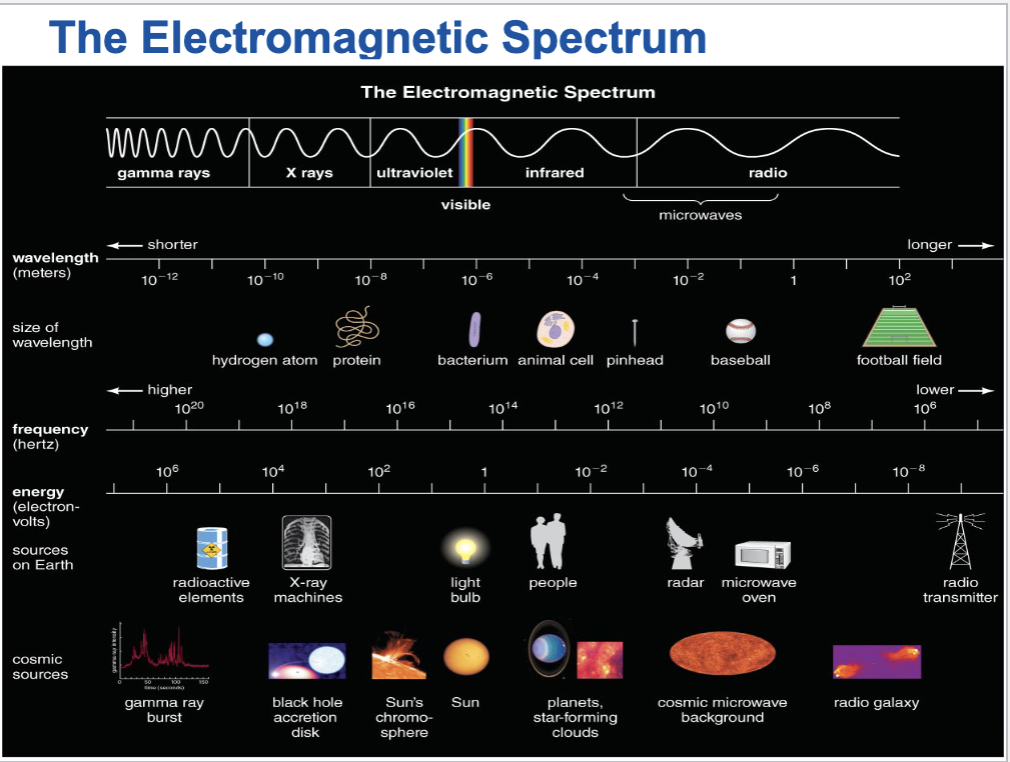
What is the electromagnetic spectrum?
Human eyes cannot see most forms of light
The entire range of wavelengths of light is known as the electromagnetic spectrum.
Properties of Matter
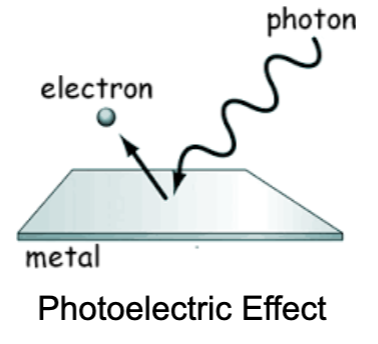
What is the structure of matter?
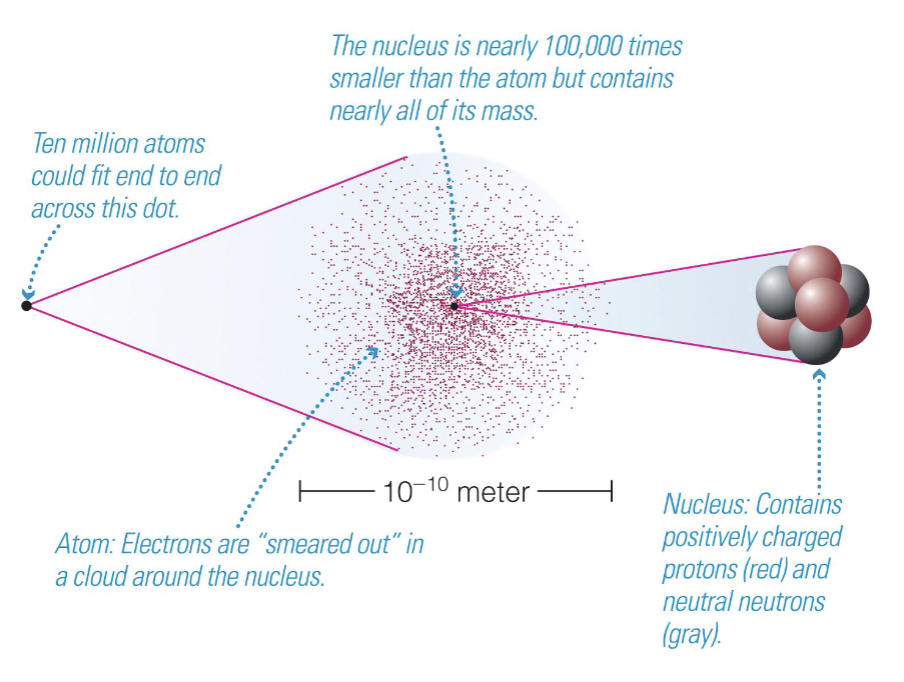
Atomic Terminology
Atomic number = # of protons in nucleus
Atomic mass number = # of protons + neutrons
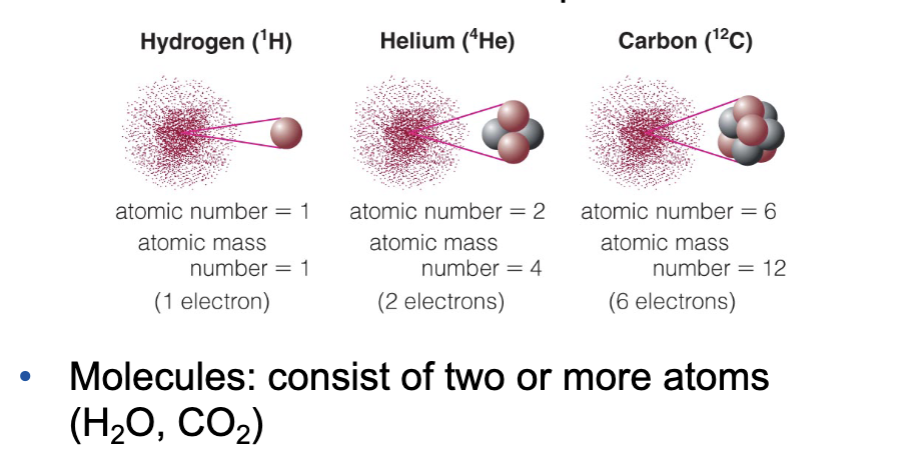
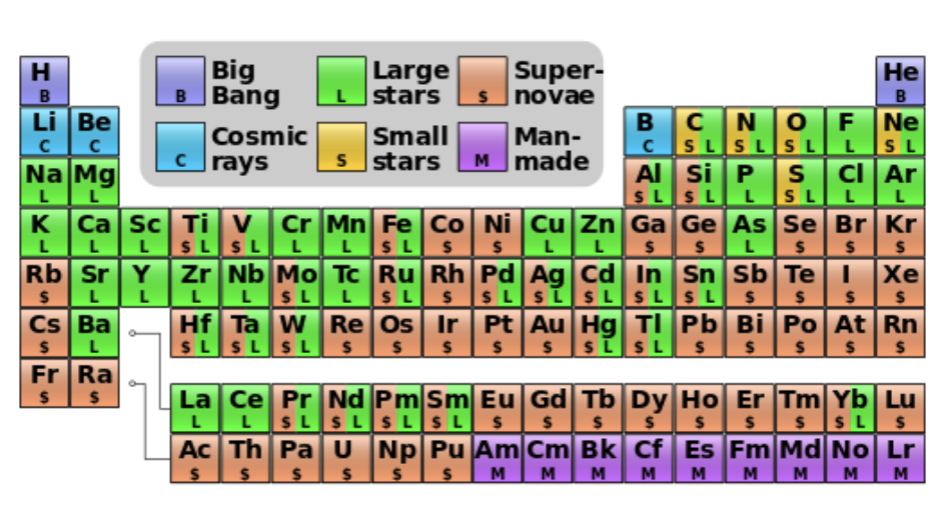
What made us?
Origin of elements
Atomic Terminology
Isotope : same # of protons but different # of neutrons (^4He, ³He)
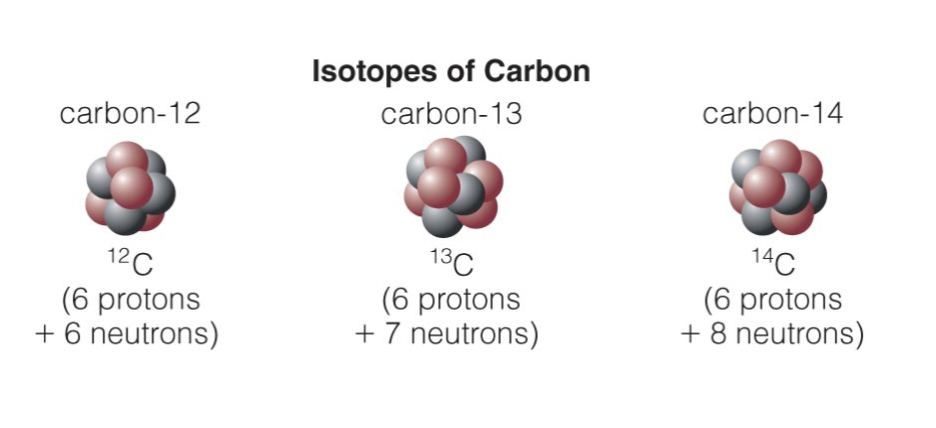
What are the phases of matter?
Familiar phases:
Solid (ice)
Liquid (water)
Gas (water vapor)
Phases of same material behave differently because of differences in chemical bonds.
Phase Change
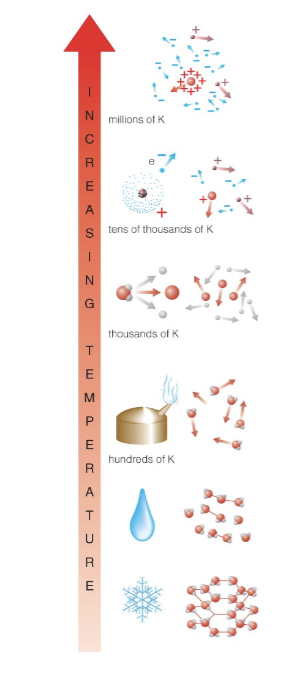
Ionization: stripping of electrons, changing atoms into plasma
Dissociation: breaking of molecules into atoms
Evaporation: breaking of flexible chemical bonds, changing liquid into gas
Melting: breaking of rigid chemical bonds, changing solid into liquid
Phases and Pressure
Phase of a substance depends on both temperature and pressure
Often more than one phase is present
How is energy stored in atoms?
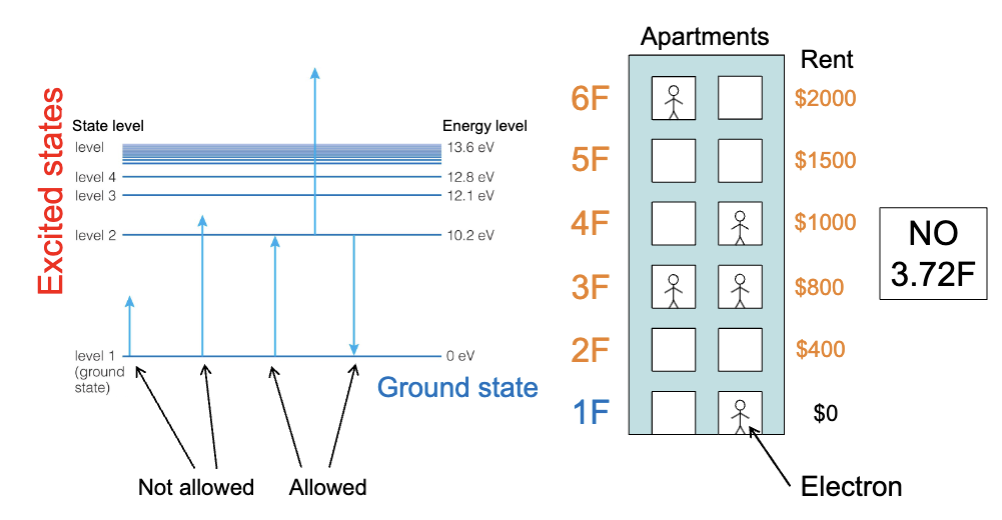
Electrons in atoms are restricted to particular energy levels. ← Quantum Mechanics
Energy Level Transitions
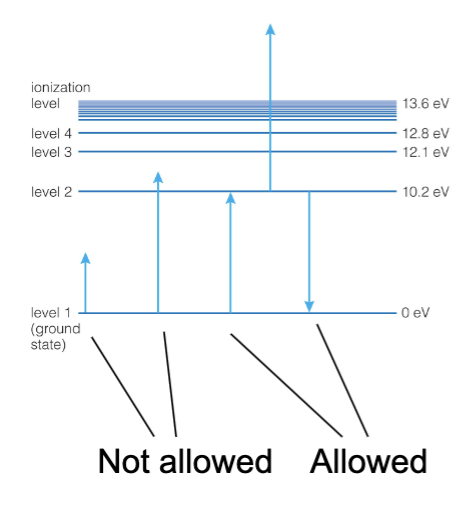
The only allowed changes in energy are those corresponding to a transition between energy levels.
Example: Laser
What have we learned?
What is the structure of matter?
Matter is made of atoms, which consist of a nucleus of protons and neutrons surrounded by a cloud of electrons.
What are the phases of matter?
Adding heat to a substance changes its phase by breaking chemical bonds.
As temperature rises, a substance transforms from a solid to a liquid to a gas, then the molecules can dissociate into atoms.
Stripping of electrons from atoms (ionization) turns the substance into a plasma.
How is energy stored in atoms?
The energies of electrons in atoms correspond to particular energy levels.
Atoms gain and lose energy only in amounts corresponding to particular changes in energy levels.