Unit 07? Orbital Diagrams and e- Configurations
Atomic Orbitals
A region in space in which there is a high probability of finding an e-.
The energy levels are represented by principal quantum numbers, n.
n = 1, 2, 3, 4, etc.
The principal quatum numbers indicate the relative size and energies of the atoic orbitals.
As n increases, the orbital becomes larger and the e- spend more time farther from the nucleus.
n specifies the atom;s major energy levels 9called principal energy levels)
The number of the princpal energy level is equal to the number of sublevels that it can contain.
Quantum #1 has 1 sublevel
Quantum #2 has 2 sublevels
Quantum #3 has 3 sublevels
Quantum #4 has 4 sublevels
Each energy sublevel corresponds to an orbital or a
different shape, which describes where the e- is likely
to be found.
4 sublevels = 4 orbitals = s, p, d, and f
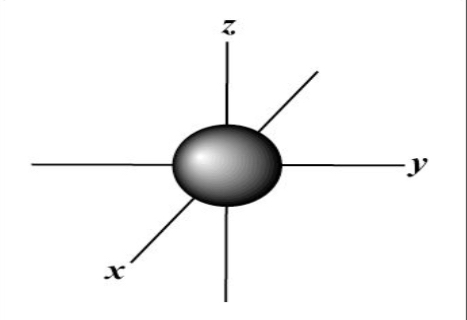
S Orbital
Spherical in shape
Lowest Energy level there is
1 orientation in space (1 orbital)
Holdea a maximum of two e-
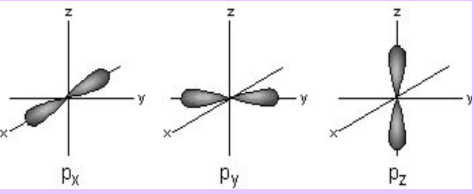
P Orbital
Dumbell shape
Has more energy than s
3 orbitals (3 orientations in space)
Can hold a maximum of 6 e- (2/orbital)
n=2, # e- =2.718 8 (2 from s, 6 from p)
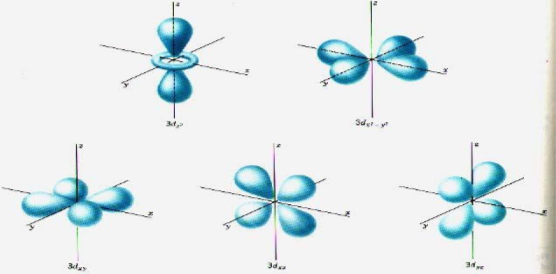
D Orbital
Complex shape
Has more energy than s and p orbitals
5 orbitals (5 orientations in space and shape)
Can hold a max of 10 e-
n = 3, # of e- = 18
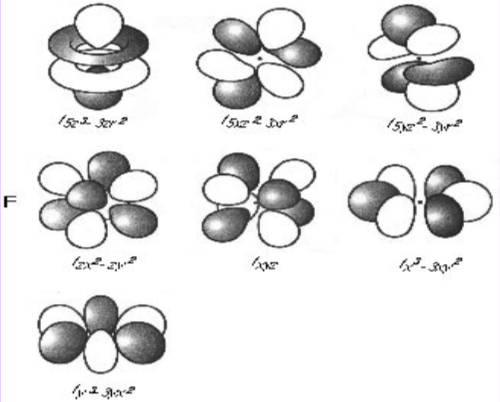
F Orbital
Complex shape
Orbital with the highest energy
7 orbitals (orientations in space and shape)
Hold a max of 14 e-
n = 4, 32 e- total
Orbital Diagrams
e- fill orbitals of lowest energy first
Each orbital can be occupied by up to two electrons
Since each sublevel holds a different number of
orbitals, they hold a different number of total e-
The e- in any orbital spin in opposite directions
↑ represents an e- spinning in one direction
↓ represents an e- spinning in the other direction
e- will not pair until there is one e- in each orbital


● The element's location on the PT tells you how to
complete the orbital diagram.
● Remember the PT is divided into s, p, d, and f
blocks.
● Whichever block the element is located in, will tell
you the ending of your orbital diagram.
● To determine the specifics of the orbital diagram,
you use an Aufbau diagram to show you how the
e- fill the orbitals in terms of energy.
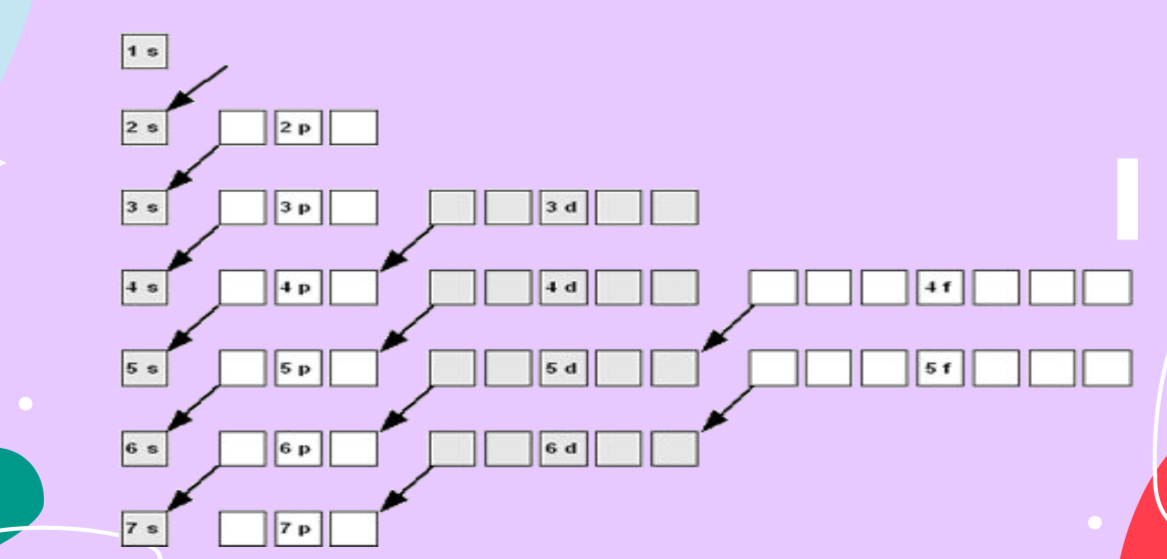
Aufbau Principle — CandyLand!
All orbitals in an energy sublevel are of equal energy
Energy sublevels within a principal energy level have different energies
e- occupy the orbitals of lowest energy first
e- occupy the s orbital, then p, and so on

Pauli Exclusion Principle
An atomic orbital can describe at most 2 e-
e- spin
In order to occupy an orbital, e- must have opposite spins
Arrows are used to represent the e- and their spin
Hund’s Rule — Busdriver!
e- eter each orbital one at a time until all tthe orbitals of that type have one e- with the same spin direction
Orbital Diagrams
If you complete your diagram sucessfully, then the number of arrows should equal the atomic number of the element.
You should also notice your diagram ended in the orbital based one what block the element is located.
Notice toy cannot pair e- until each orbital has at least one e- in it.
Checkpoint 02
Draw the orbital diagram for the following elements.

e- Configuration
The way electrons are arranged in the orbitals around the nucleus
Specific order
Also follows the 3 rules:
Aufbau principla
Pauli Exclusion Principle
Hund’s rule
How to Write e- Configurations
Materials - PT and Aufbau diagram
Locate the element on the PT
Use the aufbau diagram to know the principle quantum number for the orbitals
Fill the orbitals based on Pauli exclusion principle and Hund’s rule
Noble Gas Configuration
Shorthand version of e- configuration
Allows you to “start” and e- configuration from the closest Nobel gas and continue from there
The Nobel gas must come before the element on the PT
Find the Nobel gas that precedes the element
Write the Nobel gas in brackets
Start writing the e- configuration form the noble gas

Exceptions
Although the Aufbau rule accurately predicts the electron configuration of most elements, there are notable exceptions among the transition metals and heavier elements.
the reason these exceptions occurs is that some elements are more stable with fewer electrons in some sub shells and more electrons in others
Their configuration is more stable by having half filled sublevels than having the normal configuration
There are several different exceptions, but you will be responsible for knowing only 5
The five elements you need to know are chromium, molybdenum, copper, silver, and gold
When you look at their locations on the PT, you should notice they are in the transition metals
Chromium and molybdenum would have configurations that ended in d^4
However, it is more stable to have two half filled orbitals instead of one full s and and partial d
For copper, silver, and gold, they would normally end in d^9
Again, it is more stable to have a filled d orbital and a half filled s orbital
So for chromium and molybdenum the ending would be s^1d^5
For copper, silver, and gold the ending would be s^1d^{10}