Carbohydrates
Molecules
Monomers
A small molecule that can bind chemically to other molecules
Polymers
A large molecule that’s formed when monomers link together chemically in a chain
Macromolecules
Complex molecule composed of repeating units of smaller molecules covalently linked together
Structure of Carbohydrates
Carbohydrates/Saccharides are divided into three chemical groups:
Monosaccharides (simple sugars)
Alpha glucose, beta glucose, galactose, fructose
Disaccharides (double sugars)
Sucrose, malatose, lactose
Polysaccharides (complex sugars)
Starch, glycogen, cellulose, chitin
Carbohydrates
A large biological molecule consisting only of carbon, hydrogen, and oxygen
General empirical formula is (CH2O)n, where n is the number of carbons in the molecule, represents carbohydrates
Carbohydrates Role
Energy Source
Carbohydrates broken down to mainly glucose are the preferred source of energy for our body, as cells in our brain, muscle, and all other tissues directly use monosaccharides for their energy needs
Monosaccharides are directly absorbed by the small intestine into the bloodstream, where they are transported to the cells in need
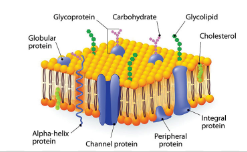
Membrane Carbohydrates
Structural role as a physical barrier
Participate in cell recognition
Structural Support
Diff carbohydrates, particularly those in the form of polysaccharides, contribute to the building of cellular structure
In plants particularly, cellulose creates a solid wall around the plant cells, giving the plant its structure
Biochemical Synthesis
Carbohydrates release carbon atoms as they break down
These serve as the raw material for much of an organism’s biochemistry, as the carbon can then join w other chemicals in the body
Monosaccharides
Simple carbohydrates that consist of 1 monomer subunits
Contains 6 carbon atoms, 12 hydrogen atoms, and 6 oxygen atoms
An energy source, building blocks (monomer)
Most monosaccharide names end w the suffix -ose
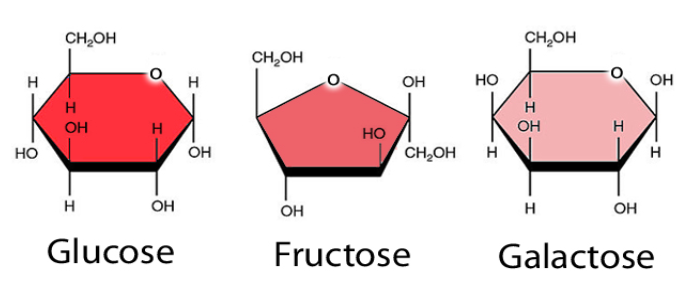
Ex. Glucose, fructose, galactose, ribose, deoxyribose
Glucose is an important source of energy (aka blood sugar)
Fructose is commonly found in fruits and known as fruit sugar
Galactose is found in milk products
Ribose and deoxyribose are sugar components of DNA (C5H10O5)
Monosaccharides are classified based on:
The position of their carbonyl group
The number of carbons in the backbone
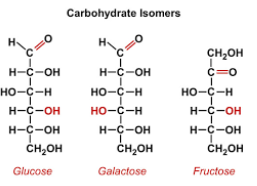
Carbonyl groups - monosaccharides
Monosaccharides can be distinguished by their carbonyl group
Glucose, fructose, and galactose are isomers, meaning they have the same chemical formula (C6H12O6) but a diff atom arrangment
# of carbons - monosaccharides
Monosaccharides can be distinguished by the # of carbons
Ribose and Deoxyribose are a pentose sugar (5 carbons)
Glucose is a hexose sugar (6 carbons)
In solutions
Monosaccharides form a ring structure when dissolved in water
The hydroxyl group on carbon 1 can end up either above or below the ring
α-glucose - below the ring
β-glucose - above the ring
Dry State
Monosaccharides are linear in a dry state
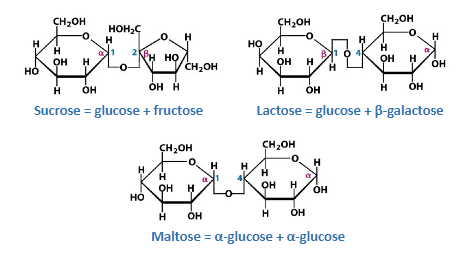
Disaccharides
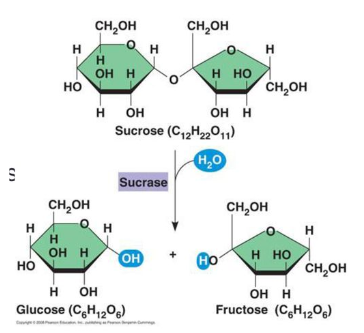
Formed when two monosaccharide molecules join in a dehydration rxn to form a glycosidic bond
An H2O molecule is removed in the process
Used as energy storage and as a building block for larger molecules
Some important disaccharide molecules include:
Sucrose (table sugar)
Maltose (grain sugar)
Lactose (milk sugar)
These types of disaccharide formed depends on the monomers involved & their form (alpha or beta)
Polysaccharides
A long chain of monosaccharides linked by glycosidic bonds is a polysaccharide
The chain may be branched or unbranched and it may contain different types of monosaccharides
Primary examples include:
Starch
Glycogen
Cellulose
Starch and glycogen are examples of storage for carbohydrates
Cellulose and chitin are examples of a structural complex carbohydrates
Disaccharides and polysaccharides can be broken down into smaller, simpler sugars via hydrolysis
A chemical rxn using water to break bonds to form two or more new substances
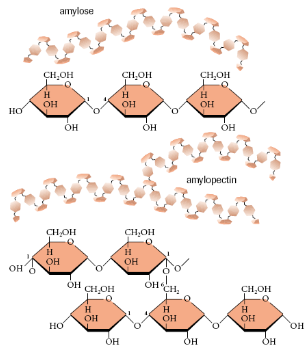
Starch
2-6 thousand glucose molecules bonded together forming amylose (unbranched, coiled α-glucose chain) or amylopectin (branched α-glucose chain)
Storage molecules used by plants
Glucose made through photosynthesis can be stored as a starch polymer
Glycogen
The storage form of glucose in humans and other vertebrates and is comprised of monomers of glucose
Stored in liver and muscle cells
Whenever blood glucose levels decrease, glycogen breaks down to release glucose
Good for long-term source of energy
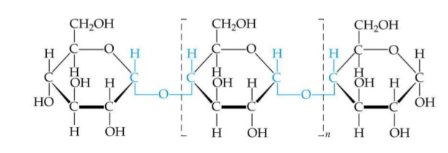
Cellulose
Structural component of plant cell walls that provides support
Straight chain of β-glucose monomers w β 1-4 linkages
Linkages cause inversion of every other monomer
Form bundles of fibres
Humans cannot digest cellulose bc we lack the enzymes in our digestive systems to break down the bonds within cellulose
The linear shape of cellulose allows it to interact w water
Chitin
Exoskeleton of insects and crustaceans
Contains a glucose-like monomer w a nitrogen group at C-2
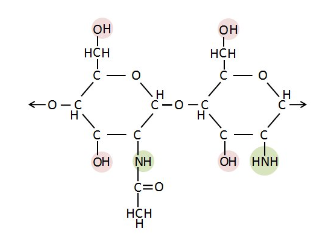
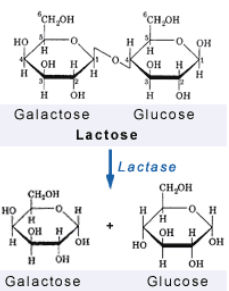
Lactose Intolerance
Lactase is an enzyme that breaks down the glycosidic bond in lactose
Although almost all babies are born w the ability to produce lactase, some people lose that as they get older
Lactose intolerant lmao