Nuclear Chemistry
Chemical Symbol:

Mass number - atomic number = neutron number
Chemical vs. Nuclear Reactions
Chemical Reactions | Nuclear Reactions |
Occur when bonds are broken | Occur when nuclei emit particles or rays. |
Atoms are unchanged, but can be rearranged | Atoms usually converted into different elements |
Involve valence electrons only | May involve protons, neutrons, and electrons |
Associated with small energy changes | Associated with large energy changes |
Reaction rate influenced by temperature, particle size, concentration, etc. | Reaction rate not influenced by temperature, particle size, concentration, etc. |
Nuclear Fission
Splitting of a nucleus
Chain reaction, one particle sets off another atom
Nuclear reactors control these situations, allowing energy to be released slowly.
Nuclear Fusion
Combines nuclei
Does not normally happen because protons repel each other
Inexpensive, does not produce radioactive waste, but is unrealistic power source as of now because requires lots of energy and very hard to control.
Examples: Stars, hydrogen bomb
Nuclear stability
Very stable - Atomic numbers 1-20 tend to have 1:1 ratio of protons to neutrons
Marginally stable - Atomic numbers 21-82 tend to have 1:1.5 ratio of protons to neutrons.
Unstable/radioactive - Atomic number >82 tend to have >1:1.5 ratio of protons to neutrons
Strong force keeps nucleus together despite proton repulsion
If there are too many neutrons strong force will fail, and release particles out of the nucleus.
Types of Decay
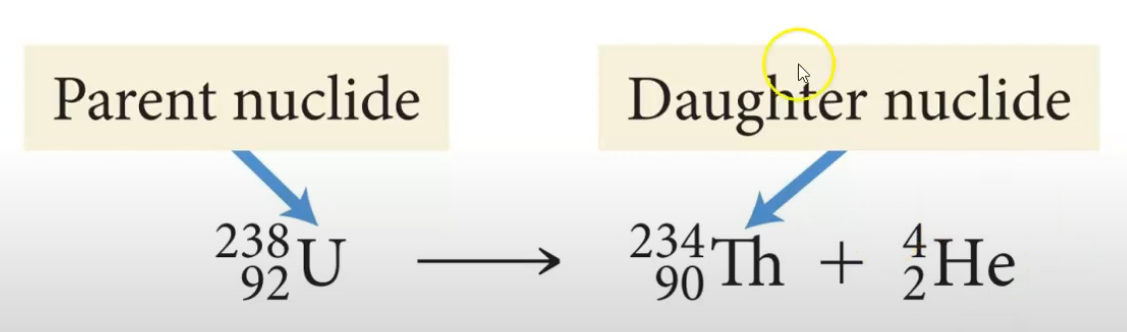
Alpha Decay - Unstable nucleus emits a particle composed of 2 protons and 2 neutrons (helium nuclei). They have a charge of 2+ because there are no electrons. Can be written out with helium symbol and the mass and atomic number or with alpha alpha symbol.
Beta Decay: Neutrons converted into protons, and electrons (β particles) shown as (β or e-). Have -1 charge.
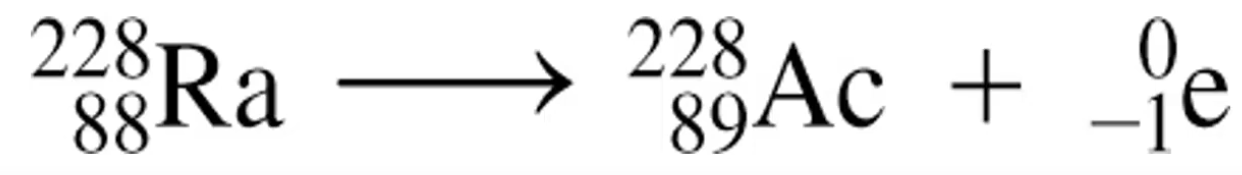
Gamma Emissions:
High energy released
Particles are not lost from nucleus
Usually occurs after nucleus goes through decay and remaining particles rearrange. No charge on gamma emissions, just energy

Electron Capture: nucleus captures low energy electron
usually occurs when mass number less than normal
Positron emission: like beta emission but with a positive charge.
usually occurs when mass number less than normal
Decay can take multiple steps in a decay series.