Pure Chemistry - States of Matter
Matter
anything that has mass and occupies space
made of tiny particles (atoms, molecules, or ions) that are always moving
States of matter refer to the distinct forms that different phases of matter take on:
Solid
Particles are closely packed together in a fixed arrangement.
They vibrate in place but do not move freely.
Solids have a definite shape and volume.
Liquid
Particles are close together but can move past one another.
Liquids have a definite volume but take the shape of their container.
Gas
Particles are far apart and move freely and quickly in all directions.
Gases have neither a definite shape nor volume, expanding to fill their container.
Plasma
A state of matter where gases are energized until atomic electrons are no longer associated with sent nuclei.
Found in stars, including the sun.
Has unique properties like conductivity and responsiveness to electromagnetic fields.
Effect of temperature and pressure on gases:
When temperature increases:
particles move faster;gas volume increases if pressure is constant
Pressure increases (at constant temperature):
gas volume decreases (gas is compressed)
Gas volume is very sensitive to temperature and pressure changes.
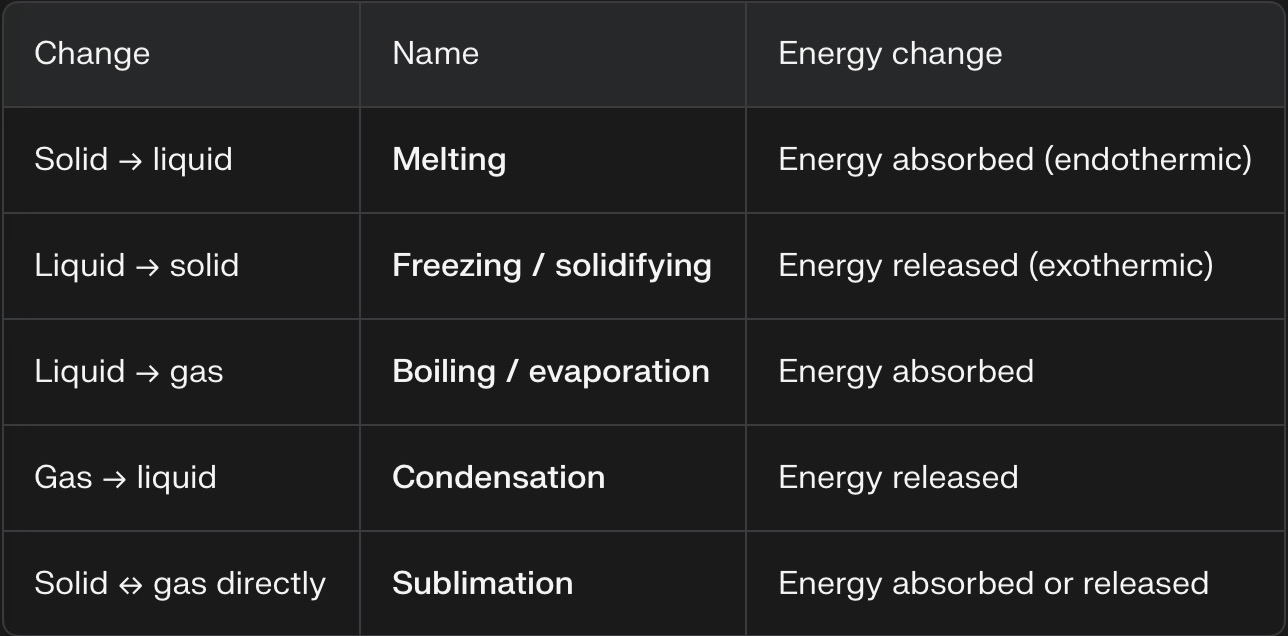
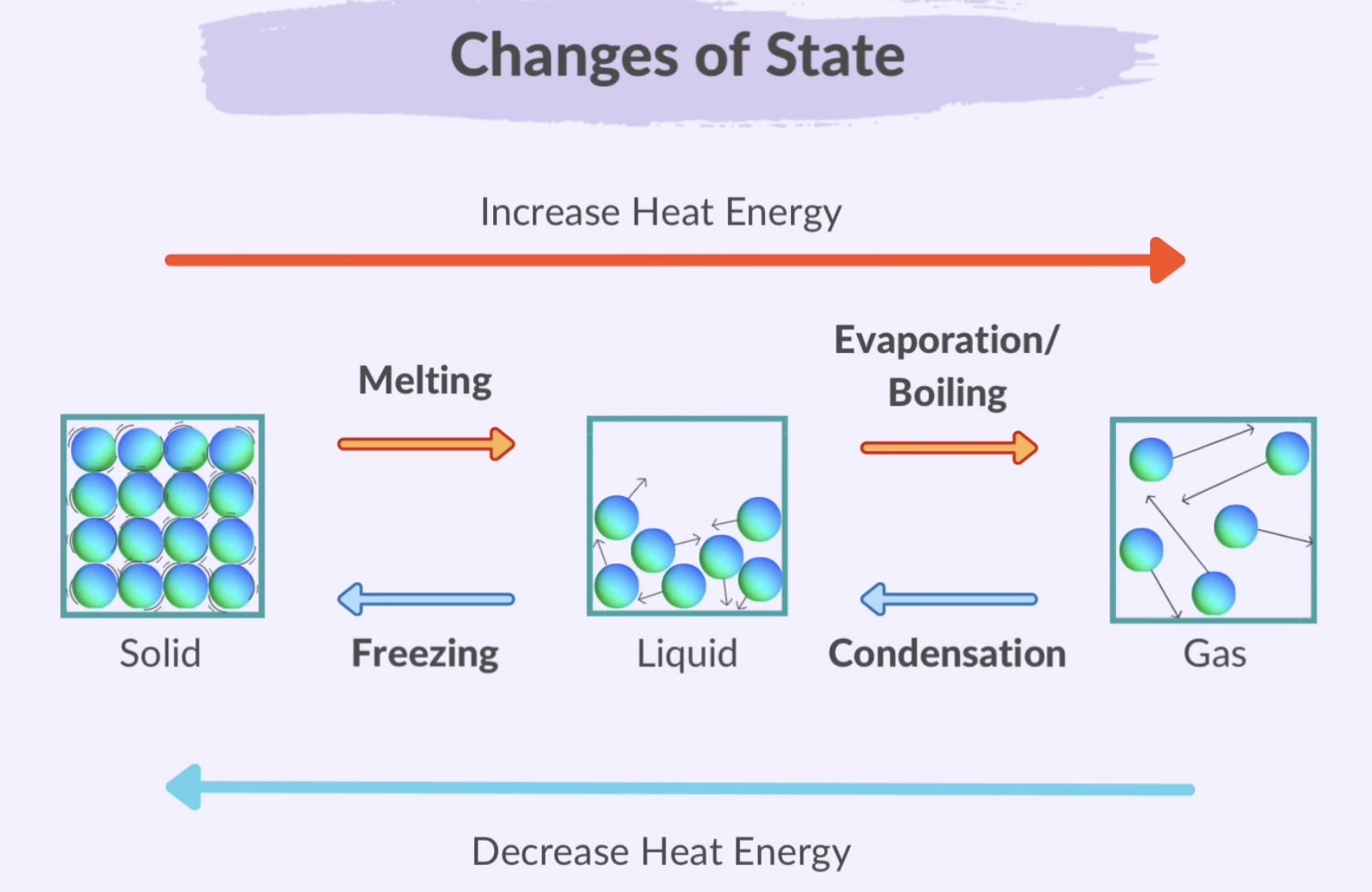
Changes of State
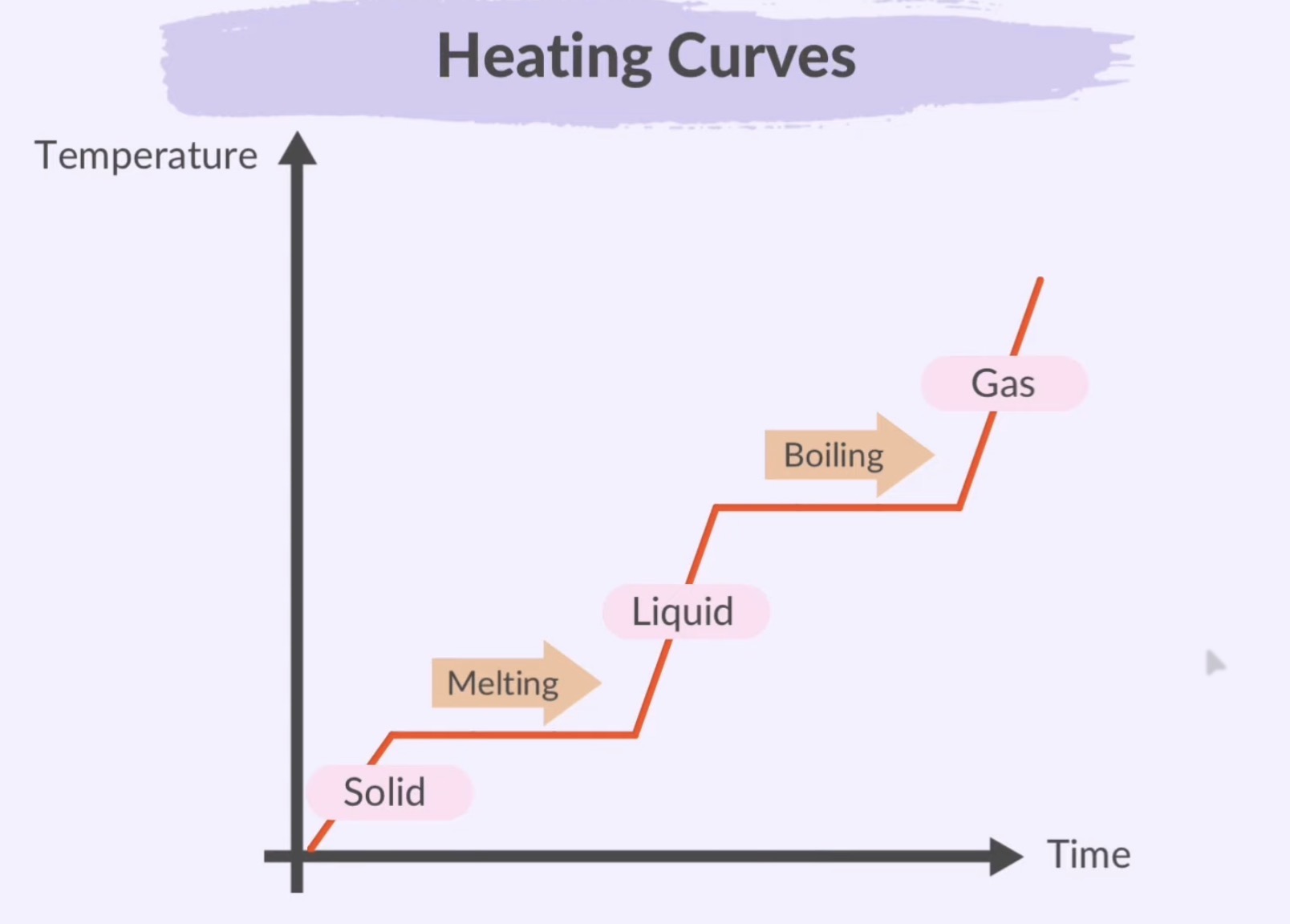
Melting: Solid to Liquid
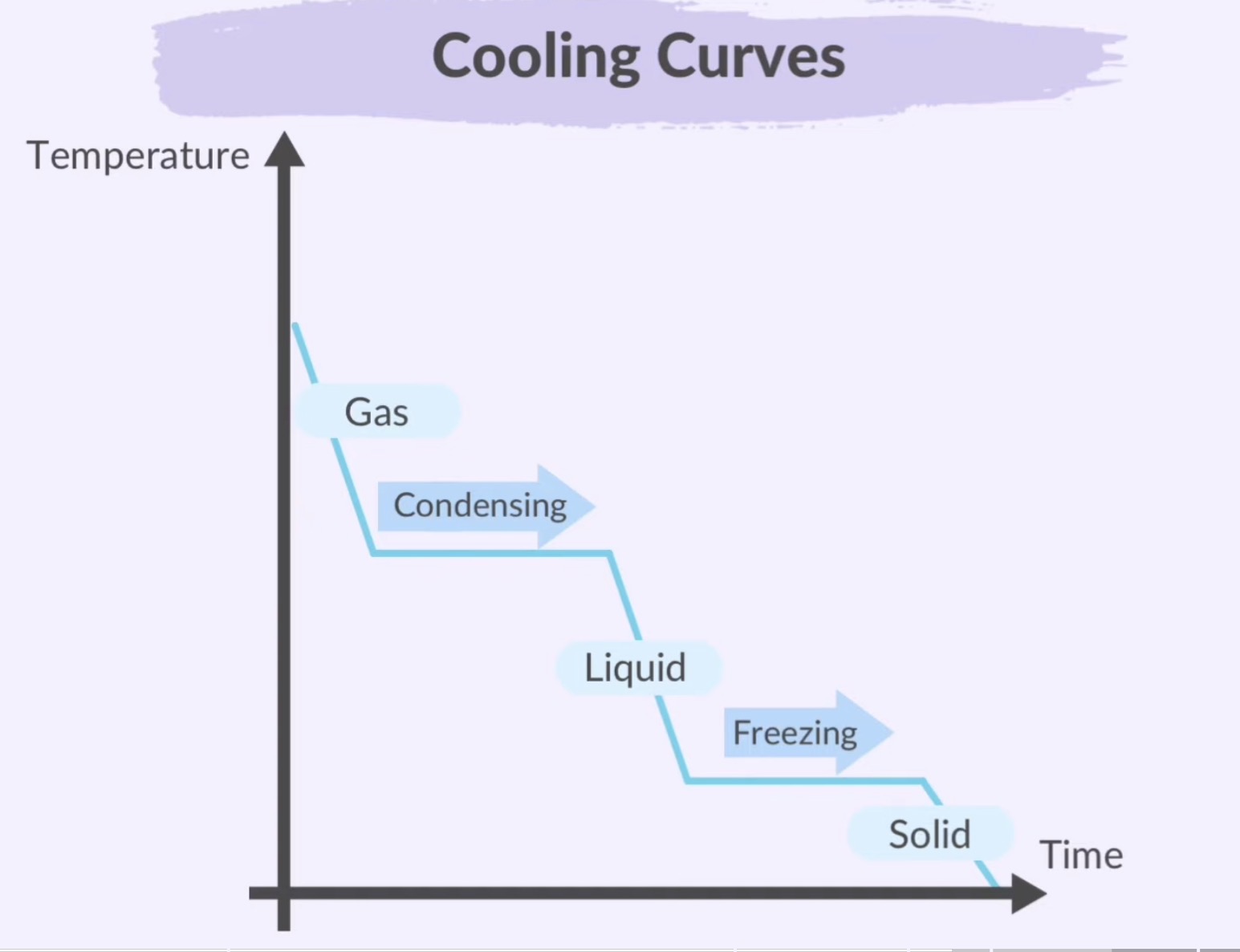
Freezing: Liquid to Solid
Boiling (Evaporation): Liquid to Gas
Condensation: Gas to Liquid
Sublimation: Solid to Gas without passing through the liquid state
Deposition: Gas to Solid without passing through the liquid state
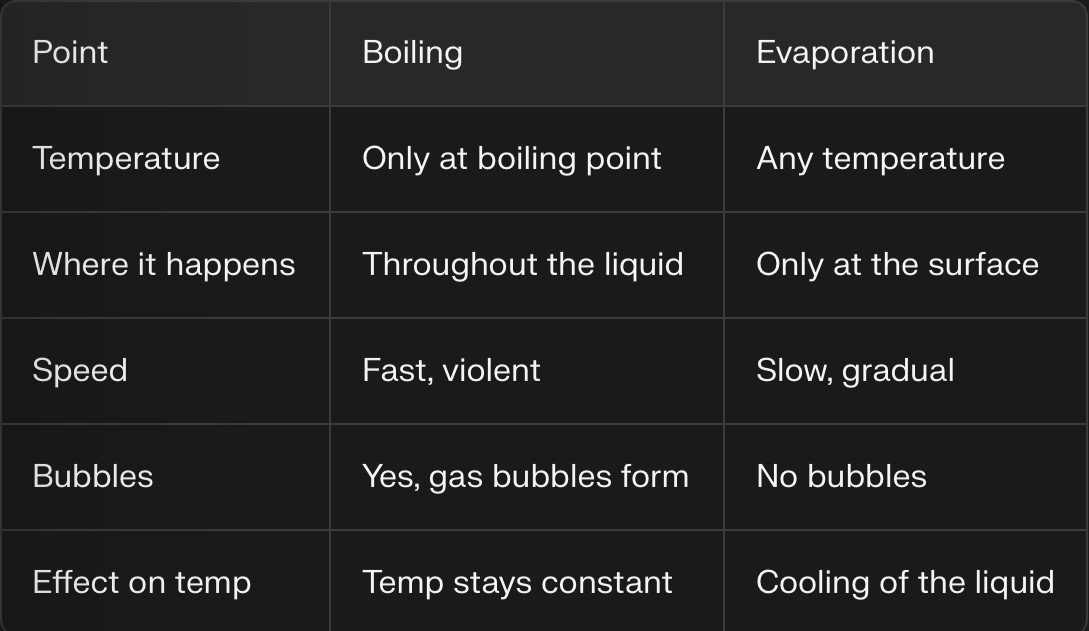
Evaporation v.s. Boiling
Boiling
happens only at the boiling point of the liquid
occurs throughout that whole liquid (bubbles of gas form inside and rise to the surface
a fast, vigorous process driven by an external heat source
temp of the liquid stays constant during boiling until all the liquid has turned to gas
Evaporation
happens at any temperature, below the boiling point
occurs only at the surface of the liquid, no bubbling
a slower process, rate increases with temperature, surface area, and air movement
often causes cooling of the remaining liquid because fastest moving molecules escape first
Graphical Representation:
Phase Change Diagram: A graph plotting temperature against time that illustrates the changes of state during heating or cooling.
Temperatures rises excepts at melting and boiling, where it plateus
Temperature falls excepts at freezes and condensation, where it is constant
Plateaus show the stage change (solid ←→ liquid, liquid ←→ gas)
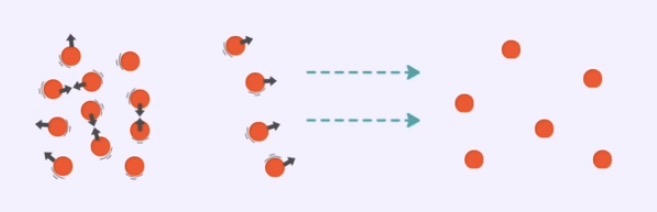
Diffusion - movement of particles from a region of high concentration to low concentration
Occurs in gases and liquids (not in solids)
Faster at higher temperatures
When particles are in a high concentration area, they collide frequently and move randomly.
As they collide and exchange energy, some particles gain enough energy to break away and move to areas with lower particle concentration.
This continuous motion and collision causes particles to spread out until they are evenly distributed.
Kinetic Particle Theory: all matter is made of tiny particles in constant random motion
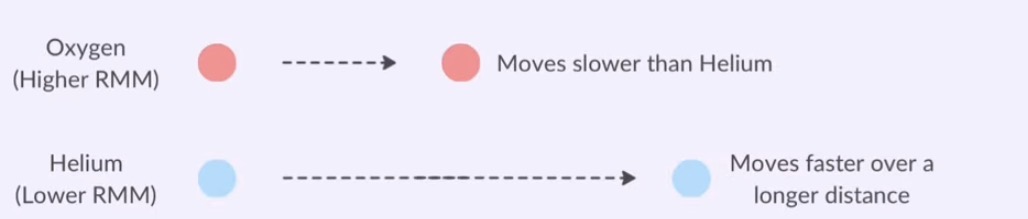
Relative Molecular Mass on Diffusion of Gases:
Lighter gas particles are able to move faster and cover greater distance than heavier ones.
Gas will diffuse more quickly when it has a lower relative mass.