C8 Rate of reaction
C8.1
- find the rate of reaction by the amount of reactant used over a period of time
- or you can find the amount of product produced over time
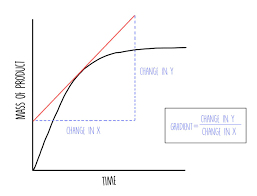
- steeper line on the graph means higher rate of reaction
- to find the gradient of a line that’s curved you need to draw a tangent
- mean rate of reaction = quantity or reactant used or product made / time take
C8.2 collision theory
- particles must collide with enough energy to react
- the minimum amount of energy is called activation energy
- the higher the ratio of SA to volume the more collisions there will be
C8.3 effect of temperature
- reactions increase in speed when temp increases
- because particles collide more frequently and with more energy
- a higher proportion of particles have energy greater than the activation energy
C8.4 effect of concentration/pressure
- increasing the concentration increases the frequency of collisions and therefore increasing rate of reaction
- same for pressure but with gas
- test how much gas different concentrations give off using a gas syringe attached to a conical flask
- another test is to set up a conical flask with the reactants over an X you drew then wait until you can’t see the X because there is too much suspended precipitate (insoluble solid). Repeat for different concentrations and measure with stopwatch
C8.5 effect of catalyst
- used to speed up the rate of reaction and is not used up in reaction, chemically unchanged
- different catalysts for each reaction
- used wherever possible in industry to reduce energy costs and impacts on the environment
- lower the activation energy required
C8.6 reversible reactions
- products can react together to make the reactants
- shown with a half arrow
C8.7 energy and reversible reactions
- in these reactions one is endothermic and the other direction is exothermic
- the amount of energy transferred to the surroundings one way is the exact same energy transferred the opposite direction
- example blue hydrated copper sulphate and white anhydrous copper sulphate, heat 1 and get two. Add water to 2 you get 1, it also gets hot because it’s transferring energy to the surroundings (exothermic)
C8.8 Dynamic equilibrium
- the rate of reaction is equal in both directions, each one is still happening but the mass of reactants and products remain the same. ONLY IN CLOSED SYSTEMS
- Le Chatelier Principle - noticed that when you change the conditions in a system at equilibrium (changing concentration, temp, pressure), the position of equilibrium changes to cancel out the change.
- For example if the concentration of reactant increases the amount of product will increase to try to achieve equilibrium again
C8.9 altering conditions
- increasing pressure favours the reaction that forms fewer molecules of gas, if the forward reaction produces more molecules of gas an increase in pressure would decrease in the amount of product formed
- basically, if the pressure increases on one side the equilibrium will try to shift to reduce pressure.
- increasing the temperature favours the endothermic reaction (increases the product formed)
\ \ \ \ \ \ \ \ \ \ \ \