ENE190 Reviewer
Soil is formed by the process of weathering of rocks which has great variability in its chemical composition.
Primary minerals are those which are not altered chemically since the time of formation and deposition.
These non-clay minerals with low surface area (silica minerals) and with low reactivity.
Quartz, feldspar, micas, amphibole
Secondary minerals are mostly layered alumino-silicates, which are made up of silicon/oxygen tetrahedral sheets and aluminium/oxygen octahedral sheets.
These are clay minerals with high surface area and high reactivity that affect the chemical transport of liquid and vapours.
Basic layers of basic units of clay minerals
Silica layer, (Si4O10)-4
Gibbsite layer is otherwise termed as dioctahedral structure in which two-third of central portion is occupied by Al+3
Brucite layer is termed as trioctahedral structure in which entire central portion is occupied by Mg+2
Description on common clay minerals
Kaolinite formation is favoured when there is abundance of alumina and silica is scarce.
low electrolyte content, low pH and removal of ions that flocculate silica
regions with heavy rainfall that facilitate leaching of above cations
Halloysite is formed by the leaching of feldspar by H2SO4 produce by the oxidation of pyrite.
high-rain volcanic areas
Smectite group of mineral formation are favoured by high silica availability, high pH, high electrolyte content, presence of more Mg+2 and Ca+2 than Na+ and K+.
less rainfall and leaching and where evaporation is high (such as in arid regions)
Illite formation, potassium is essential in addition to the favourable conditions of smectite.
Important properties of clay minerals
Specific surface area (SSA)
surface area of soil particles per unit mass (or volume) of dry soil
High specific surface area is associated with high soil-water-contaminant interaction, which indicates high reactivity.
Increasing reactivity:
Kaolinite < Illite < Montmorillonite
As particle size increases SSA decreases.
Plasticity and cohesion
Clay attracts dipolar water towards its surface by adsorption. This induces plasticity in clay.
plasticity increases with SSA
Water in clays exhibits negative pressure due to which two particles are held close to each other, resulting to apparent cohesion.
Surface charge and adsorption
In general, clay particle surface are negatively charged and its edges are positively charged.
Reason why clay surface is charged:
Isomorphous substitution - shortage of one positive charge, which appears as negative charge on clay surface. Such substitution is therefore the major reason for net negative charge on clay particle surface.
Dissociation of hydroxyl ions or broken bonds at the edges - positive charge can occur on the edges of kaolinite plates due to acceptance of H+ in the acid pH range
Exchangeable cations and cation exchange capacity
Due to negative charge, clay surface attracts cations towards it to make the charge neutral.
Exchangeable cations - replaced by easily available ions present in the pore solution
Cation exchange capacity (CEC) - total quantity of exchangeable cations; unbalanced negative charge existing on the clay surface
Determination of CEC is done after removing all excess soluble salts from the soil.
The adsorbed cations are then replaced by a known cation species and the quantity of known cation required to saturate the exchange sites is determined analytically.
Flocculation and dispersion
When two clay particles come closer to each other it experiences:
interparticle attraction due to weak van-der-Waal’s force
repulsion due to –ve charge
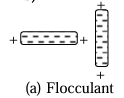
Flocculant structure - when particles are sufficiently close, attraction becomes dominant active force and hence there is an edge to face configuration for clay particles
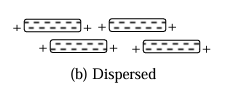
Dispersed structure - when the separation between clay particles increase, repulsion becomes predominant and hence the clay particles follows face to face configuration
Swelling and shrinkage
when exposed to moisture are subjected to excessive swelling and during drying undergo excessive shrinkage
Decrease in swelling of clay minerals:
montmorillonite > illite > kaolinite
Minerals other than clays and silica
Carbonate minerals such as calcite and dolomite are found in some soils in the form of bulky particles, precipitates
Sulphate minerals mainly gypsum are found in semiarid and arid regions.
Soil mineralogy characterization
X-ray diffraction (XRD) analysis - identifies minerals based on this unique crystal structure
Soil sample is initially dried and sieved through 2 mm sieve.
Sieved sample is homogenized in a tumbler mixer for 30 min. Homogeneity is very important.
A control mix of 30 g was taken and ground in lots of 15 g in a gyratory pulverizer.
15 percent by weight of KIO4 (internal standard) was added to 5 g of specimen and again homogenized in a mixer.
Characteristic X-rays of particular wave length are passed through a crystallographic specimen.
The diffraction pattern of the soil specimen is then analyzed for the qualitative and quantitative (not always) assessment of minerals.
Cu- Kα - X-ray wave of monochromatic radiation (Kα) is commonly obtained from copper radiation
XRD output - ordinate represent relative intensity of X-ray diffraction and abscissa represents twice of angle at which a striking X-ray beam of wave length λ makes with parallel atomic planes.
Quantitative determination of mineral composition in soils based on simple comparison of diffraction peak height under peak is complex and uncertain because of different factors such as mineral crystallinity, hydration, surface texture of the specimen, sample preparation, non-homogeneity of soil samples, particle orientation.
Constant mineral standard method - increasing quantity of clay are added to the fixed mass of known standard and the difference in X-ray diffraction intensity when the specimen changes from 100 percent standard to 100 percent clay is noted.
The peak area ratio for each component is then plotted against percentage of clay
Constant clay method - known weight of pure standard mineral is added to clay containing the same components, and the change in the reflection peak-area intensity of each component is measured to estimate the weight proportion of that component.
Applications of soil mineral analysis in geoenvironmental engineering
The soil-water and soil-water-contaminant interaction and hence reactivity is greatly influenced by the mineralogy.
When there are problems associated with expansive soils, the best method for identifying the problem is by conducting XRD and checking for expansive clay minerals.
Acidification of soils - transformation of minerals; micas get transformed to vermiculite by weathering process under acidic condition.
By analyzing mineralogy, the land use practices can be assessed.
Soil-water-contaminant interaction
Unbalanced force fields are generated at the interface of soil water, which increases soil-water interaction.
When particles are finer, magnitude of these forces are larger than weight of these particles. This is mainly attributed to low weight and high surface area of fine particles.
Forces between soil solids:
Electrostatic or primary valence bond - atoms bonding to atoms forming molecules
Secondary valence bond - when atoms in one molecule bond to atoms in another molecule (intermolecular bond).
van der Waals force and hydrogen bonds
van der Waals force is the force of attraction between all atoms and molecules of matter; sufficiently close to each other
Hydrogen bond is formed when a hydrogen atom is strongly attracted by two other atoms.
stronger than Van der Waals force of attraction and cannot be broken under stresses that are normally experienced in soil mechanics.
Secondary valence force existing between molecules is attributed to electrical moments in the individual molecules.
Non-polar - when the centre of action of positive charge coincide with negative charge, there is no dipole or electric moment for the system
Non-polar molecules can become polar when placed in an electric field due to slight displacement of electrons and nuclei.
Polar - for a neutral molecule there can be cases where the centre of action of positive and negative charge does not coincide, resulting in an electric or dipole moment.
unsymmetrical distribution of electrons in silicate crystals
The forces in soil mechanics may be grouped as gravitational forces and surface forces.
In geoenvironmental engineering surface forces are important.
Surface forces are classified as attractive and repulsive forces.
Attractive forces:
Van der Waals London forces - most important in soils and becomes active when soil particles are sufficiently close to each other.
hydrogen bond
cation linkage - acts between two negatively charged particles as in the case of illite mineral structure
dipole cation linkage
water dipole linkage
ionic bond
Repulsive forces:
like charge particle repulsion
cation-cation repulsion
Soil-water interaction
Soil water or pore water - water present in pore spaces of soil
free water or gravitational water - flows freely under gravity under some hydraulic gradient and are free from the surface forces exerted by the soil particle; removed easily from the soil
held water or environmental water - held under the influence of surface forces such as electrochemical forces or other physical forces
structural water - present within the crystal structure of mineral; not very important as far as engineering property of soil is concerned.
adsorbed water
capillary water
Adsorbed water
Strongly attracted to soil mineral surfaces especially clays
Adsorptive forces between soil and water is polar bond and depends on specific surface area of soil.
Adsorbed water or bound water behaves differently from the normal pore water.
It is immobile to normal hydrodynamic forces and its density, freezing point etc. are different from free water.
Hygroscopic water content - dry soil mass can adsorb water from atmosphere even at low relative humidity; vary depending on relative humidity and temperature.
Possible mechanisms for water adsorption:
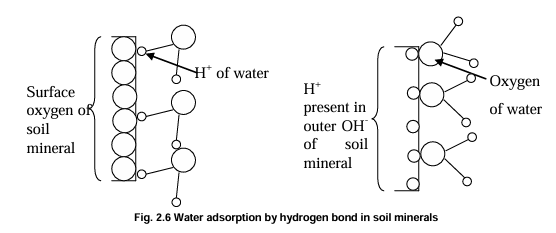
Hydrogen bond and dipole attraction - Surface oxygen can attract positive corner of water molecules (H+) and H+ present in OH- can attract negative corner (O-2) of water molecules.
Hydration of cations - cations get hydrated by water dipole due to the formation of hydrogen bond; cations present in the soil would contribute to the adsorbed water occupy in the porous space of clay mineral
During hydration, these cations engulfs with water molecules and move towards centre space between two clay particles.
Osmosis - clays act as semi-permeable membranes; concentration of cations increases with proximity to clay surface.
Relatively high concentration would induce osmotic flow of water to neutralize the high concentration of cations
Attraction by Van der Waals-London forces - causes attraction of water molecules towards clay surface.
Capillary condensation - water and water vapour can get retained in soil pores by capillary forces and attraction to particle surfaces.
Properties of adsorbed water:
Density - at low water content, the density of adsorbed water is greater than that for normal water. For higher water content, less density variation with reference to free water
Viscosity - not much difference in viscosity between adsorbed and free water. This is a very important observation relative to analysis of seepage, consolidation etc. for unsaturated soils.
Dielectric constant - the property depends on the ease with which the molecules in the material can be polarized. It is observed that dielectric constant of adsorbed water (50) is less as against for free water (80).
Freezing of adsorbed water - adsorbed water exhibit freezing point depression as compared to free water. This is mostly attributed to the less molecular order of adsorbed water as compared to free water.
Other properties - energy is released when water is adsorbed by clay surface. There is a time-dependent increase in moisture tension of water after mechanical disturbance of at-rest structure of clay-water.
Diffused double layer (DDL)
The diffusion tendency of adsorbed cations and electrostatic attraction together would result in cation distribution adjacent to each clay particle in suspension.
The result of clay-water-electrolyte interaction; term for the charged clay surface and the distributed ions adjacent to it
Adsorbed cations - cations are held strongly on the negatively charged surface of dry fine-grained soil or clays.
Cations in excess of those needed to neutralize electronegativity of clay particles and associated anions are present as salt precipitates.
When dry clays come in contact with water, the precipitates can go into solution. The adsorbed cations would try to diffuse away from the clay surface and tries to equalize the concentration throughout pore water.
Close to the surface there is high concentration of ions which decreases outwards.
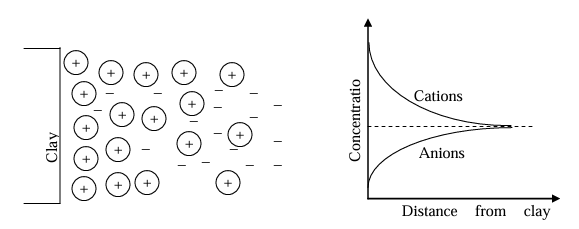
Double layers of ions (a) compressed layer and (b) diffused layer thus DDL.
A high concentration of cations close to clay surface gradually reduces, and reaches equilibrium concentration at a distance away from clay surface.
For anions, concentration increases with distance from clay surface.
In addition to ion quantification, DLVO describe the repulsive energies and forces of interaction between clay particles and prediction of clay suspension stability.
The theory predicts ion distribution reasonably for only smectite particles suspended in monovalent ion solution at low concentration.
Assumptions which pertain to the formulation of DDL theory:
Ions in the double layer are point charges and there are no interactions among them.
Charge on particle surface is uniformly distributed.
Platy particle surface is large relative to the thickness of double layer (to maintain one dimensional condition).
Permittivity of medium adjacent to particle surface is independent of position.
Permittivity is the measure of the ease with which a molecule can be polarized and oriented in an electric field.
Different models representing double layer:
Helmholtz double layer - surface charge of clays are neutralized by opposite sign counter ions placed at a distance away from the surface
In this model, positive charges are considered to be stationery, which is against the reality that cations are mobile.
Gouy Chapman model - interfacial potential at the charged surface can be attributed to the presence of a number of ions of given sign attached to the surface and to an equal number of opposite charge in the solution.
Electric potential in double layer relates ion distribution to electric potential (Blotzmann equation) and relates electric potential and distance (Poisson equation); does not consider other interaction energy components.
This model is accurate only if the soil behaves like a true parallel particle system.
Stern model - total cations required to balance the net negative charge on clay surface consists of two layers.
Electric double layer (EDL) or Stern layer - clay surface charges and the adsorbed group of cations
Boltzmann distribution - other group of cations are diffused in a cloud surrounding the particle
Cation exchange capacity (CEC)
The sum of exchangeable cations soil can adsorb per 100 g of dry soil; replaced easily by another incoming cation
Replaceability of cations depends on valency, relative abundance of different ion type and ion size
Typical replaceability series:
Na+ < Li+ < K+ < Cs+ < Mg+2 < Ca+2 < Cu+2 < Al+3 < Fe+3
The rate of exchange reaction would essentially depend on clay type, pore solution concentration, temperature, pH
In kaolinite, the reaction takes place quickly
In illite, a small part of the exchange sites may be between unit layers of minerals and hence would take more time.
In smectite minerals, much longer time is required because the major part of exchange capacity is located in the interlayer region.
For a pore solution containing both monovalent and divalent cations, the ratio of divalent to monovalent cations is much higher in adsorbed layer than in the equilibrium solution.
Exchangeable sodium percentage (ESP) - sodium present in the adsorbed layer is normalized with respect to total exchange capacity
Clays with ESP > 2 percent is considered as dispersive.
Factors influencing CEC of the soil:
pH of the soil - CEC of the soil increases with an increase in pH
Presence of organic matter - presence of organic carbon in clays reduces its CEC; in coarser fractions, an increase in CEC with increasing organic matter contents
Temperature - ion exchange capacity decreases with an increase in temperature
Particle size - CEC increase with decreasing particle
Calcium carbonate contents (CaCO3) - higher amount of CaCO3 in soil leads to higher CEC
Mineralogy - active clay minerals increase CEC of the soil.
CEC is important in assessing the chemical properties of the soil in terms of its reactivity, contaminant retention mechanism.
Determination of CEC by ammonium replacement method.
This method involves saturation of the cation exchange sites on the soil surface with ammonium, equilibration, and removal of excess ammonium with ethanol, replacement and leaching of exchangeable ammonium with protons from HCl acid.
Quantification of soil water
Capacity factor - water present in the soil voids are quantified as water content
Intensity factor - energy status of water
Gravimetric water content - ratio of weight of water to the weight of soil solids (weight basis)
Volumetric water content - ratio of volume of water (Vw) to the total volume of soil (V)
Mechanical energy of water
Kinetic energy is important in granular soils where velocity is significant and also in the case of preferential flow in soils.
Preferential flow is caused in soils due to the formation of macrocracks which is mostly attributed to the shrinkage cracks in soil, holes or burrows created by animals, cracks caused by the roots of plants
Potential energy is the most important energy component of water present in the porous media.
The rate of decrease in PE is termed as hydraulic gradient (i).
PE of water is termed as soil-water potential.
Gravitational potential - due to the difference in elevation between two reference points and hence it is also known as elevation head (z); above is negative, below is positive
Matric potential - due to the adsorptive and capillary forces present in the soil; retains water towards the soil surface (always negative)
Pressure potential - below the ground water table (always positive)
Osmotic potential - caused due to salts and contaminants (solutes) present in the soil pore water (always negative)
A higher relative vapour pressure is associated with high water content of the soil sample.
As water content increases, matric suction reduces.
As concentration of solute increases, osmotic potential also increases.
Soil water moves from higher ψt to lower ψt (total soil-water potential)
If only about liquid flow, ψ0 is considered negligible because the solutes also move along with the flowing water
Hydraulic potential causing flow - ψt can be rewritten as ψg + ψp + ψm
Under hydraulic equilibrium, ψt is same everywhere, spatially.
Hydrologic horizons
Groundwater zone - phreatic zone; exists below ground water table and hence will be fully saturated.
All the voids in this zone are filled with water and water pressure will be always positive.
Saturated soil mechanics can be applied
Vadoze zone - partially saturated zone or unsaturated zone above water table; extends from the top of the groundwater table to the ground surface
voids are filled with air or water and the relative percentage is decided by the amount of saturation.
Lowest portion of this zone can be nearly saturated due to the phenomenon of capillary rise. However, the water in this zone will be held under tension.
Root zone - top portion of vadoze zone close to ground surface where the plants and trees grows.
subjected to evaporation and evapotranspiration and is in direct interaction with the atmosphere
During precipitation, infiltration and flow of water to the subsurface occurs through this zone
Moisture dynamics is applied
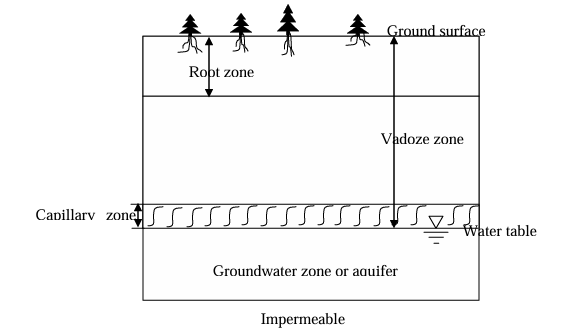
Unsaturated soil (vadoze zone)
Soil suction - energy required for extracting unit volume of water from soil; interaction of solid, water and air phase present in the soil develops a complex energy state resulting in negative pore water pressure
Soil-water characteristic curve (SWCC) or water retention characteristic curve (WRCC) - study of unsaturated soil behaviour is dependent on the basic relationship between suction and water content or saturation
mandatory for studying the behaviour of unsaturated soil
Total soil suction - sum of matric suction (ψm) and osmotic suction (ψo)
Due to hysteresis, drying WRCC has higher suction than wetting curve for particular water content.
Key points that are relevant for WRCC:
Volumetric water content at saturation - water content at which the soil is completely saturated; depicts the initial state for the evaluation of the drying path
Air-entry value - air enter the largest pore present in the soil sample during a drying process
Residual water content - minimum water content below which there is no appreciable change in θ
Water-entry value - matric suction at which the water content of the soil starts to increase significantly during the wetting process.
Desaturation of soil specimen:
Stage 1: capillary saturation zone - soil remains saturated with the pore-water held under tension due to capillary forces
Stage 2: desaturation zone - sharp decrease in water content and the pores are increasingly occupied by air
Stage 3: zone of residual saturation - little hydraulic flow, some water vapour movement.
Beyond Stage 3, increase in soil suction does not result in significant changes in water content.
Specific capacity or differential water capacity - slope of WRCC describing water storage and water availability to plants.
As C(θ) is more, water drained out or water availability from that soil is more.
Complexity in modelling the behaviour of unsaturated soil
As against the steady state behaviour in saturated soil, an unsaturated soil therefore exhibit transient behaviour.
In the case of saturated soil, hydraulic conductivity (ks) remains constant with time since pores are filled with water.
For a particular soil, ku (unsaturated) increases as suction decreases till it approaches ks
suction decreases due to the increase in water content which results in the increase in ku.
Different soil-water-contaminant interaction mechanisms
The major role of a geoenvironmental engineer is to predict the fate of contaminants in the subsurface and minimize its migration towards groundwater source.
Fate prediction is very essential to understand the presence of contaminants in groundwater sources or subsurface for long term (50 to 200 years)
Chemical mass transfer and attenuation:
Sorption - contaminant partitioning; contaminants clings on to the soil solids
Dissolution/ precipitation - addition or removal of contaminants
Acid-base reaction - proton transfer
Redox reaction - electron transfer
Hydrolysis/ substitution/ complexation/ speciation - ligand-cation complexes.
Mass transport:
Advection - fluid flow
Diffusion - molecular migration
Dispersion - mixing
Other factors:
Biological transformations
Radioactive decay
Attenuation - reduction in contaminant concentration during fluid transport due to retardation, retention and dilution
Retention - strong sorption of contaminants on the soil particles such that the concentration of pore fluid decreases with time.
Chemical mass transfer and irreversible sorption removes the contaminants from the moving pore fluid.
Retardation - reversible sorption and hence release of contaminant would eventually occur; with much delay
Dilution - the effect of contaminated pore fluid is reduced when it interacts with fresh water which delays the contaminant migration
For an effective waste management, retention process is more ideal than retardation.
Important reactions determining attenuation:
Hydrolysis - reaction of H+ and OH- ions of water with the solutes and elements present in the pore water.
The reaction increases with decrease in pH, redox potential and organic content and increases with temperature.
Oxidation-reduction (redox) reaction - transfer of electrons between the reactants.
microorganisms act as catalysts for reactions (redox) involving molecular oxygen, soil organic matter and organic chemicals.
Factors affecting Eh (redox potential) include pH, oxygen content or activity, and soil water content.
Complexation - reaction between metallic cations and anions called ligands
Precipitation and dissolution - dissolution increases and precipitation decreases the concentration of contaminants in pore water.
Dissolution - complete solubility of an element in groundwater.
Precipitation - dissolved element comes out of the solution due to the reaction with dissolved species.
pH governs the solubility limit and hence when pH changes, there is a possibility of precipitation reaction.
Exsolution and volatilization - mass transfer between gaseous and liquid or solid phase; removes mass from pore fluid to gaseous phase
governed by the vapour pressure (pressure of gaseous phase) with respect to liquid or solid at a particular temperature.
Radioactive decay - unstable isotopes decay to form new ones with release of heat and particles from element nucleus.
α decay - the element looses a helium particle
β decay - the element looses an electron
Sorption and partitioning - when contaminant laden pore water flow past the soil surface, mass transfer of these contaminants takes place on to the solids.
The amount of partitioning depends on the soil surface (sorbent) and the reactivity of contaminant (sorbate)
Physical (ad)sorption - attraction of contaminant on to the soil surface mainly due to the surface charge (electrostatic force of attraction); reversed easily by washing with extracting solution
Chemical sorption - strong force of attraction due to the formation of bonds such as covalent bond
Biological transformation - degradation or assimilation of contaminants (mostly organic) by microorganisms present in the soil
aerobic conditions are oxidation reaction - hydroxylation, epoxidation, and substitution of OH groups on molecules
Anaerobic biotic transformation processes are mostly reduction reaction - hydrogenolysis, H+ substitution for Cl– on molecules, and dihaloelimination
Major application is in organic contaminant remediation