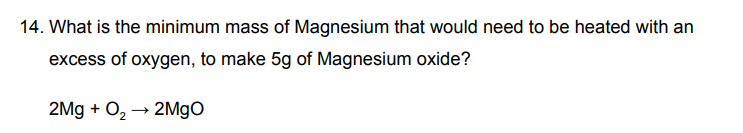C4.2 more equations
{{I GIVE UP{{
calculating mass from an chemical equation (the ones that show the reactants and products)
how much magnesium can make magnesium oxide
2Mg + O2 → 2MgO
it shows 2 moles of magnesium reacting with 1 mole of oxygen to produce 2 moles of magnesium oxide
find the Ar (Mg = 24 and O = 16)
(24 x 2) + (2 x 16) = 48 + 32 = 80g
if you have 5.00g of magnesium you can find the amount of magnesium oxide produced
find how much 1g produces : 80/48 (total over mg) = 1.66666666667
find how much 5g produces (80/48) x 5 = 8.33g
- calculate the mass of calcium oxide that can be made from 10g of calcium carbonate ,
%%CaCO3 → CaO + CO2%%
%%find Ar (Ca = 40, O = 16, C = 12)%%
%%40 + 12 + (16 x 3) = 100g%%
%%CaO = 40 + 16 = 56%%
==1g produces : 100/56 = 1.78571428571==
==10g produces : 1.78571428571 x 10 = 17.9g==
==17.9g==
- 10g gives (10/100) x 56 = 5.6g CaO
(24 x 2) + (16 x 2) = 48 + 32 = 80g
1g : 80 / 48
5g : (80 / 48) x 5 = 8.33g
\
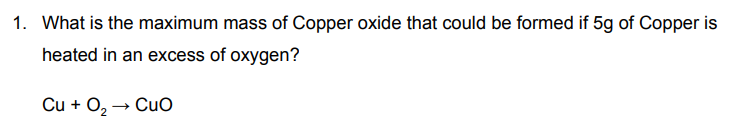
63.5 + 32 = 95.5g
5g : (95.5 / 63.5) x 5 = 7.52g