chemistry 2025-2026
Test Dates
August 28
September 18
October 9
November 6
November 25
December 18
January 22
February 12
March 5
April 2
April 23
May 14
What is Chemistry ?
The study of matter and energy, and the changes it undergoes; fundamental science; often abstract.
Branches of Chemistry:
Organic, Inorganic, Physical, Analytical, and Biochemical.
The Scientific Method and Research Framework
The Scientific Method: a systematic, empirical, and iterative approach to research, used to investigate observations, propose explanations, and test hypotheses.
Key steps:
Observation/Collection of Data: This initial step involves noticing a phenomenon and gathering information (data) through senses or instruments. Data can be qualitative (descriptive) or quantitative (numerical).
Qualitative and Quantitative Data:
Qualitative data describe characteristics or qualities, such as color, odor, or texture. They are non-numerical.
Quantitative data are numerical measurements, such as temperature, volume, or mass. They often involve units and are obtained using measuring instruments.
Formulation and Testing of Hypothesis: A hypothesis is a testable, educated guess or proposed explanation for an observation. It must be specific, falsifiable, and often takes the form of an "if…then…" statement. Testing involves designing and conducting experiments or further observations to collect evidence that either supports or refutes the hypothesis.
The progression from Hypothesis to Theory to Law:
A hypothesis is a tentative explanation.
A theory is a well-substantiated, comprehensive, and testable explanation of a natural phenomenon, supported by a vast body of evidence from many confirmed hypotheses. Theories explain why something happens (e.g., Kinetic Molecular Theory explains gas behavior).
A law is a concise statement or mathematical equation that describes a fundamental relationship in nature, typically describing what happens under certain conditions, often without explaining why (e.g., Law of Conservation of Mass).
Experimental design elements: To ensure reliable and valid results, experiments are carefully designed.
Independent Variable (IV): The variable that is intentionally changed or manipulated by the experimenter. It is the presumed cause.
Dependent Variable (DV): The variable that is measured or observed; its value changes in response to changes in the independent variable. It is the presumed effect.
Control (or Control Group): A standard of comparison, kept under normal or unchanged conditions. It helps ensure that the observed effects are due to the independent variable and not other factors.
Constant (or Controlled Variable): Factors that are kept the same throughout the experiment to ensure fair testing and prevent them from influencing the dependent variable.
Model (Example): A representation of an idea, object, or process that is too difficult or dangerous to observe directly. Models can be physical, conceptual, or mathematical (e.g., Bohr model of the atom, weather simulations).
Properties of Matter
Substance: A type of matter with a uniform and definite composition throughout, possessing distinct properties. Substances can be elements or compounds.
Pure substance: Consists of only one type of matter, with invariant chemical and physical properties (e.g., pure water, gold).
Mixtures: Physical combinations of two or more substances in which each substance retains its own distinct chemical identity and properties. The components can be separated by physical means.
Physical properties: Characteristics of a substance that can be observed or measured without changing the chemical composition or identity of the substance. These properties describe the substance itself (e.g., melting point, boiling point, density, color, hardness, state of matter).
Chemical properties: Describe a substance’s ability or inability to undergo a chemical change or reaction that transforms it into a new, different substance. These properties relate to how a substance interacts with other substances (e.g., flammability, reactivity with acids, oxidation).
Physical Properties and Related Concepts
Physical Properties are observed or measured without changing the composition of the substance. They are inherent characteristics.
Extent of properties:
Extensive properties: Properties that depend on the amount of substance present. They are additive. Examples include mass, volume, and total energy content (e.g., a larger sample of water has more mass and volume).
Intensive properties: Properties that do not depend on the amount of substance present. They are characteristic of the substance itself and help identify it. Examples include density, melting point, boiling point, temperature, color, and hardness (e.g., the density of water is the same regardless of the amount).
Chemical Properties and Reactions
Chemical property: The ability of a substance to chemically combine with or change into one or more new substances. This change involves the rearrangement of atoms and formation of new chemical bonds.
Be able to determine chemical vs physical properties from observations/experiments:
If the substance's chemical identity changes, it's a chemical property (e.g., burning wood changes it to ash and gases, indicating flammability as a chemical property).
If the substance remains chemically the same, it's a physical property (e.g., ice melting into liquid water, where both are HO, is a physical property related to its melting point).
States of Matter and Kinetic Theory
States of matter: The physical forms in which matter exists, primarily distinguished by particle arrangement and energy.
Solid: Particles are tightly packed in a fixed, orderly arrangement, vibrating in place. Solids have a definite shape and definite volume. They are generally incompressible.
Liquid: Particles are close together but are able to flow past one another, taking the shape of their container. Liquids have a definite volume but an indefinite shape. They are nearly incompressible.
Gas: Particles are widely spaced and move randomly and rapidly. Gases have an indefinite shape and indefinite volume, expanding to fill their container. They are highly compressible.
Plasma: An ionized gas state, consisting of a mix of ions and electrons. It is the most common state of matter in the universe (found in stars, lightning, neon signs).
KMT: Kinetic Molecular Theory of Matter: This theory explains the behavior of matter based on the motion of its particles. Key postulates include:
All matter is composed of tiny, rapidly moving particles.
These particles are in constant, random motion.
The kinetic energy of these particles increases with temperature.
There are forces of attraction between particles, weakest in gases and strongest in solids.
Physical Changes vs Chemical Changes
Physical changes: Altering a substance's form or appearance without changing its chemical composition or identity. No new substances are formed.
Examples: Phase changes (changes in state of matter):
Melting: Solid to liquid (e.g., ice to water)
Freezing: Liquid to solid (e.g., water to ice)
Vaporization (Boiling/Evaporation): Liquid to gas (e.g., water boiling into steam)
Condensation: Gas to liquid (e.g., steam turning into liquid water droplets)
Sublimation: Solid directly to gas (e.g., dry ice to carbon dioxide gas)
Deposition: Gas directly to solid (e.g., formation of frost)
Other physical changes include dissolving, cutting, bending, crushing, or breaking.
Chemical changes (Chemical Reactions): One or more substances (reactants) transform into one or more new and different substances (products) with distinct chemical compositions and properties. These changes are typically irreversible by physical means.
Evidence of chemical change (indicators that a chemical reaction may have occurred):
Color change: A distinct and often unexpected color appears.
Formation of a precipitate: A solid substance forms and settles out of a liquid solution.
Gas evolution: Bubbles (new gas) are produced, indicating a gas is formed.
Temperature change: A significant release (exothermic) or absorption (endothermic) of heat, leading to a noticeable temperature increase or decrease.
Odor change: A new or distinct smell is detected.
Light or sound production: Emission of light or sound.
Irreversibility: It's difficult to reverse the change through simple physical means.
Mixtures
Two primary types of mixtures:
Homogeneous mixture (Solution): A mixture that has a uniform composition and appearance throughout. The components are evenly distributed at a microscopic level, making it difficult to distinguish individual parts. They consist of a single phase. Examples: saltwater, sugar dissolved in water, air (mixture of gases), brass (alloy of copper and zinc), vinegar.
Heterogeneous mixture: A mixture that does not have a uniform composition; its components are not evenly distributed and can often be visibly distinguished. They consist of two or more distinct phases. Examples: oil and water, sand and water, tossed salad, granite, blood, chocolate chip cookie.
Energy and Thermodynamics
Energy definition: The capacity or ability to do work (apply a force over a distance) or produce heat.
Units: Energy is measured in the Joule (J), which is the SI unit. Other common units include the calorie (cal) and kilocalorie (kcal or Cal).
Heat: Energy that flows or is transferred from an object at a warmer temperature to an object at a colder temperature. It is a form of energy transfer, not a property possessed by a system.
Endothermic vs Exothermic processes:
Endothermic: A process in which the system absorbs heat from its
surroundings. The surroundings become colder, and the system's enthalpy increases. Examples: melting ice, evaporating water, photosynthesis, instant cold packs.
Exothermic: A process in which the system releases heat to its surroundings. The surroundings become warmer, and the system's enthalpy decreases. Examples: combustion (burning), freezing water, cellular respiration, instant hot packs.
Law of Conservation of Energy (1st Law of Thermodynamics): States that in any chemical reaction or physical process, energy is neither created nor destroyed. It can only be converted from one form to another (e.g., chemical energy to heat energy, potential energy to kinetic energy) or transferred from one system to another. The total energy of the universe remains constant.
Law of Conservation of Mass: States that in any closed system, during any chemical or physical process, mass is neither created nor destroyed. The total mass of the reactants in a chemical reaction must equal the total mass of the products. This implies that atoms are rearranged, not lost or gained.
Note/Prompt: mentioned as a consideration in the context of energy-mass equivalence. This famous equation by Albert Einstein describes the relationship between mass (m) and energy (E), where c is the speed of light. It shows that mass can be converted into energy and vice-versa, particularly relevant in nuclear reactions. While in typical chemical reactions mass changes are negligible, this law highlights a deeper principle.
Practice Scenarios and Properties Review
Practice Question 1 (Observations): A chemist notes before/after reaction:
Before: 25.0 mL blue liquid solution; shiny metallic solid; both at 25 °C.
Quantitative Observations: 25.0 mL (volume), 25 °C (temperature).
Qualitative Observations: blue liquid solution, shiny metallic solid.
After 10 minutes: volumes: 25.0 mL (unchanged); product is a clear solution; metallic solid becomes brown; temperature rises to 65 °C.
Quantitative Observations: 25.0 mL (volume), 65 °C (temperature).
Qualitative Observations: clear solution, metallic solid becomes brown (color change), temperature rise (implies heat release).
Task: List qualitative and quantitative observations. (This task encourages identification of direct measurements vs. descriptive characteristics, which is crucial for data collection.)
Practice Question 2 (Properties of Aluminum): Aluminum melting point ≈ 932 K; boiling point ≈ 2740 K; characteristics: shiny, malleable, ductile; reacts with acids, bases, and various metal ions; good conductor of heat and electricity.
Physical Properties: melting point , boiling point , shiny appearance, malleable (can be hammered into thin sheets), ductile (can be drawn into wire), good conductor of heat, good conductor of electricity. These describe aluminum without changing its identity.
Chemical Properties: reacts with acids, reacts with bases, reacts with various metal ions. These describe how aluminum undergoes chemical changes.
Practice Question 3 (Physical vs Chemical change): Two samples heated to just over 100 °C in absence of oxygen; sodium melts, water boils/vaporizes; upon mixing sodium with water after cooling, vigorous reaction occurs with some fire/explosion; pH of water increases. Task: Identify one example of a physical change and one of a chemical change.
Physical Change Examples: sodium melts, water boils/vaporizes. In both cases, the substances change state but remain chemically the same (Na is still Na, HO is still HO).
Chemical Change Example: upon mixing sodium with water after cooling, vigorous reaction occurs with some fire/explosion; pH of water increases. The vigorous reaction, fire/explosion, and change in pH (indicating formation of a new, basic substance, NaOH, from Na and HO) are strong evidence of a chemical change.
SI Base Units
The International System of Units (SI) is the modern form of the metric system and is the most widely used system of measurement. It defines seven base units from which all other units are derived.
Time: second (s)
Length: meter (m)
Mass: kilogram (kg)
Temperature: kelvin (K) (Note: Celsius (°C) is commonly used, but Kelvin is the absolute temperature scale used scientifically, where is absolute zero, and )
Amount of substance: mole (mol) (The mole is a counting unit, like a dozen, representing particles, known as Avogadro's number).
Electric current: ampere (A)
Luminous intensity: candela (cd)
SI Derived Units
Derived units are combinations of base units. They are used to measure other physical quantities.
Volume: The amount of space an object occupies. Common derived units include:
(commonly used for liquids and gases).
(commonly used for smaller volumes).
Density: A measure of how much mass is contained in a given volume. It is an intensive property.
Formula:
Common unit: grams per cubic centimeter (g/cm) or grams per milliliter (g/mL) for solids/liquids, and grams per liter (g/L) for gases.
Scientific Notation and Significant Figures
Scientific notation: A way to express very large or very small numbers concisely and to indicate the precision of a measurement. It uses powers of 10. A number in scientific notation is written as a coefficient (a number between 1 and 10) multiplied by raised to an exponent (e.g., ).
Operations rules (conceptual):
Adding/Subtracting: The numbers must have the same exponent before adding or subtracting their coefficients. The exponent in the result remains the same. The final answer should be rounded to the least number of decimal places in the coefficients when they are aligned.
Multiplying/Dividing: Multiply or divide the coefficients normally. For multiplication, add the exponents. For division, subtract the exponents. The number of significant figures in the result is limited by the least number of significant figures in any of the original numbers.
Significant Figures (Sig Figs): All the digits in a measurement that are known with certainty plus one estimated digit. They communicate the precision of a measurement. Rules for determining significant figures:
Non-zero digits are always significant (e.g., has 4 sig figs).
Zeros between non-zero digits are significant (e.g., has 4 sig figs).
Leading zeros (zeros before non-zero digits) are never significant (e.g., has 2 sig figs).
Trailing zeros (zeros at the end of a number) are significant only if the number contains a decimal point (e.g., has 4 sig figs; has 1 sig fig).
Measurements, Accuracy, and Precision
Accuracy: Refers to how close a measured value is to the true or accepted value. A measurement is accurate if it is very close to the standard or known value.
Precision: Refers to how close a series of measurements are to one another (reproducibility). A measurement is precise if repeated measurements yield very similar results, even if they are not close to the accepted value.
Relationship: High precision does not guarantee high accuracy, and vice versa. It is possible to have precise measurements that are inaccurate (e.g., all measurements are consistently off by the same amount due to a faulty instrument). Conversely, it's possible for one measurement to be accurate by chance but part of an imprecise set.
Percent Error and Percent Difference concepts introduced (formulas discussed in course materials): These are quantitative ways to express accuracy and precision.
Percent Error formula (standard): Used to quantify accuracy by comparing an observed or calculated value to an accepted value. The absolute value is used to ensure a positive result.
Practice Problems: Density and Measurements
Example: Calculate average density from three measurements: , , .
Average Density
Accepted density value given: .
Percent error calculation example (as shown in class materials): Using the calculated average density and the accepted density:
In this specific example,
Dimensional Analysis and Unit Conversions
Dimensional analysis: A powerful problem-solving technique that uses the units of measurements to guide the setup of calculations. It involves treating units as algebraic quantities that can be multiplied, divided, and canceled. This method helps ensure that calculations are set up correctly and results have the correct units.
Steps
write down the original measurement as a fraction.
since opposites cancel when multiplying fractions, find a conversion factor(s) that will remove the original unit and produce the new unit.
check the sig-figs.
Conversion factors: Ratios of equivalent values expressed in different units. They are derived from equalities (e.g., can give conversion factors or ). Conversion factors are essential for changing units without changing the actual value of the quantity.

Elements, Ions, and Isotopes
Element: A pure substance that cannot be separated into simpler substances by ordinary physical or chemical processes. Each element is defined by its atomic number (number of protons).
Ions: Atoms or groups of atoms that have gained or lost one or more electrons, resulting in a net electrical charge (imbalance of protons and electrons). Positively charged ions are cations, and negatively charged ions are anions.
Isotopes: Atoms of the same element (meaning they have the same number of protons and thus the same atomic number) but have different numbers of neutrons. This difference in neutron count leads to different mass numbers and atomic masses for the isotopes of an element.
Compounds
Compound: A pure substance formed when two or more different elements are chemically combined in fixed proportions by atoms. This chemical combination involves the formation of chemical bonds.
Compounds have properties distinct from those of their constituent elements.
There are infinite possibilities for compounds due to the variety of ways elements can combine.
Unlike mixtures, compounds can only be broken down into their constituent elements or simpler compounds by chemical reactions, not by physical means.
Atomic/Molecular Structure: Core Terms
Atom: The fundamental building block of an element; it is the smallest unit of an element that retains the chemical identity of that element. Atoms consist of a nucleus (protons and neutrons) and orbiting electrons.
Proton: A positively charged () subatomic particle located in the nucleus of an atom. Its number (atomic number, Z) determines the element's identity.
Neutron: A neutral (no charge) subatomic particle located in the nucleus of an atom. It has a mass approximately equal to a proton.
Electron: A negatively charged ( ) subatomic particle that orbits the nucleus in electron shells or orbitals. Electrons determine an atom's chemical behavior and bonding characteristics.
Atom vs Ion: An atom is electrically neutral (protons = electrons), while an ion has a net positive or negative charge due to an unequal number of protons and electrons.
Isotopes vs Ions: Isotopes of an element differ in neutron count (affecting mass), while ions of an element differ in electron count (affecting charge).
Atomic Numbers, Mass Numbers & Charge
Atomic Number (Z): The number of protons in the nucleus of an atom. It uniquely identifies an element and determines its position on the periodic table. For a neutral atom, .
Mass Number (A): The total number of protons and neutrons in the nucleus of an atom. It is an integer and helps distinguish between isotopes.
where N is the number of neutrons.
Average Atomic Mass: The weighted average of the masses of all naturally occurring isotopes of an element. This is the value typically listed on the periodic table.
Charge: The net electrical charge of an atom or ion. It is determined by the difference between the number of protons and electrons.
If electrons > protons, the charge is negative (anion).
If protons > electrons, the charge is positive (cation).
Ions: Positive and Negative
Anions are formed when a neutral atom gains one or more electrons, acquiring a net negative charge.
Cations are formed when a neutral atom loses one or more electrons, acquiring a net positive charge.
Atomic Structure: Protons, Neutrons, Electrons in Atoms
The atom consists of a dense, positively charged nucleus surrounded by a cloud of negatively charged electrons.
Proton: Located in the nucleus; mass approximately 1 atomic mass unit (amu); charge of elementary charge ( ).
Neutron: Located in the nucleus; mass approximately 1 amu (slightly heavier than proton); charge 0.
Electron: Orbits the nucleus; very small mass (approximately or about amu); charge of elementary charge ().
Bonding and Crystal Structures
Bond: A strong electrostatic attraction between two or more atoms or ions that holds them together in a molecule or crystal lattice. This attraction arises from the interaction of their electrons.
Ionic bond: A type of chemical bond formed by the electrostatic attraction between oppositely charged ions. This typically occurs when electrons are completely transferred from a metal atom to a nonmetal atom, resulting in positively charged cations and negatively charged anions.
Covalent bond: A chemical bond formed by the sharing of one or more pairs of electrons between two atoms, typically between two nonmetal atoms.
Molecule: The fundamental discrete unit of a covalently bonded compound, consisting of two or more atoms held together by covalent bonds (e.g., molecule).
Crystal lattice: A highly ordered, three-dimensional arrangement of atoms, ions, or molecules in a crystalline solid. It is the fundamental repeating unit of an ionic compound (e.g., crystal) or network solids.
Isotopes & Isotopic Abundances (Summary)
Isotopic abundances: The relative percentage of each isotope found in a naturally occurring sample of an element. These abundances are used to calculate the element's average atomic mass, reflecting the weighted average of the masses of its isotopes.
Historical Atomic Theory (Key Milestones)
Democritus (Ancient Greece): Proposed the earliest concept of atoms as indivisible, indestructible particles, suggesting that all matter is composed of tiny units called "atomos" (uncuttable).
Aristotle (Ancient Greece): Rejected the atom concept in favor of the idea that all matter is composed of four elements (earth, air, fire, water) and could be infinitely divided. His influence suppressed atomic theory for nearly two millennia.
John Dalton (early 1800s): Revived and formalized atomic theory with his postulates:
All matter is composed of extremely small, indivisible particles called atoms.
Atoms of a given element are identical in size, mass, and other properties; atoms of different elements differ in these properties.
Atoms cannot be created, divided into smaller particles, or destroyed.
Atoms combine, separate, or rearrange in simple whole-number ratios in chemical reactions.
J.J. Thomson (late 1800s): Discovered the electron using cathode-ray tubes. He proposed the "plum pudding model," depicting the atom as a sphere of uniformly distributed positive charge with negatively charged electrons embedded within it.
Robert Millikan (early 1900s): Performed the oil drop experiment, which accurately measured the charge of a single electron. From this, the mass of an electron was calculated to be approximately .
Ernest Rutherford (early 1900s): Conducted the Gold Foil Experiment, demonstrating that atoms have a small, dense, positively charged nucleus at their center, with electrons orbiting it. This overturned Thomson's model and proposed the nuclear model of the atom.
1920s-1930s: Further development led to the proton being discovered as the positive particle in the nucleus.
James Chadwick (1932) proposed the existence of the neutron, a neutral particle within the nucleus, which accounted for the remaining mass of atoms.
Bohr Model and Hydrogen Emission
Bohr model (1913): Proposed for the hydrogen atom, it describes a central positive nucleus around which a single electron orbits in specific, quantized energy levels.
These energy levels are designated by a principal quantum number, , with representing the lowest energy state (ground state) and higher values (up to generally) representing excited states.
When an electron absorbs energy, it jumps from a lower energy level (initial state, ) to a higher energy level (excited state, ).
When an electron relaxes back to a lower energy level, it emits the absorbed energy as a photon of light. The energy of the emitted photon () is precisely equal to the energy gap between the two levels:
(The absolute value is used as energy transitions are typically positive values representing magnitude of energy absorbed/emitted).
Additionally, the photon energy is related to its frequency () and wavelength ():
where is Planck's constant and is the speed of light.
The simple Bohr model successfully explains the line emission spectrum of hydrogen, but it does not accurately describe the spectra or behavior of multi-electron atoms.
Energies of Orbitals (Hydrogen-like model)
For a hydrogen atom (or any one-electron species, hydrogen-like atom), the energy of an electron in a specific orbit (or shell) can be calculated using the formula:
where is the energy of the electron in the energy level, J is Joules, eV is electron volts, and is the principal quantum number.
The negative sign indicates that the electron is bound to the nucleus; a value of 0 J (or 0 eV) would represent an electron completely free from the atom (ionization).
Orbit (n) and energy values (example table for hydrogen):
(Ground state): (most stable)
:
:
:
:
:
:
Transitions and Visible Spectrum (Hydrogen)
When electrons in excited hydrogen atoms relax from higher energy levels to the energy level, they emit photons that fall within the visible region of the electromagnetic spectrum. This series of spectral lines is known as the Balmer series.
The formula for the energy of an emitted photon is: and since frequency () is related to the speed of light () and wavelength () by , we can also write: .
Specific transitions leading to visible light (Balmer series):
Each corresponds to different colors within the violet, blue, and red range, respectively, due to their distinct photon energies and wavelengths.
Wavelengths, Frequencies, and Photon Energies (Visible Spectrum)
The visible spectrum constitutes a small portion of the electromagnetic spectrum, characterized by specific ranges of wavelengths, frequencies, and photon energies. These properties are inversely related: higher frequency means shorter wavelength and higher photon energy.
Visual color associations:
Violet: 400-450 nm
Blue: 450-495 nm
Green: 495-570 nm
Yellow: 570-590 nm
Orange: 590-620 nm
Red: 620-750 nm
Planck’s Constant, Photons, and the Photoelectric Effect
Planck’s constant (): A fundamental physical constant that quantifies energy relationships in quantum mechanics. Its value is approximately . It relates the energy of a photon to its frequency.
Photon: a massless particle; depending on energy levels, it can exhibit both wave-like and particle-like properties, allowing it to interact with electrons and influence their transitions between energy states.
Photon energy: The energy carried by a single quantum of light (a photon).
Electron volt (eV): A unit of energy commonly used in atomic and nuclear physics; .
Photoelectric effect: The phenomenon where electrons are ejected from a metal surface when light of a specific minimum frequency (threshold frequency) shines on it. This effect provided crucial evidence for the particle nature of light.
Einstein explained this by proposing that light consists of discrete energy packets called photons. Each photon carries a quantum of energy ().
If a photon's energy is equal to or greater than the metal's work function (the minimum energy required to eject an electron), an electron will be emitted.
Einstein’s photon concept: Light exhibits wave-particle duality, meaning it can behave as both a wave and a stream of particles (photons). These photons are massless particles that carry a specific quantum of energy.
Wave-Particle Duality & Light Spectra
Wave-particle duality: The concept that all matter and energy exhibit properties of both waves and particles.
Frequency–wavelength–energy relationships for electromagnetic radiation:
The speed of light () is constant in a vacuum and relates wavelength () and frequency ():
The energy () of a photon is directly proportional to its frequency and inversely proportional to its wavelength:
Spectrum types:
Continuous spectrum: Produced when white light (e.g., from a heated solid or liquid) is passed through a prism, showing a continuous band of all wavelengths/colors (e.g., rainbow).
Emission (bright line) spectrum: Produced by excited atoms of an element in gaseous form, showing a series of bright, discrete lines against a dark background. Each element has a unique emission spectrum, acting as a
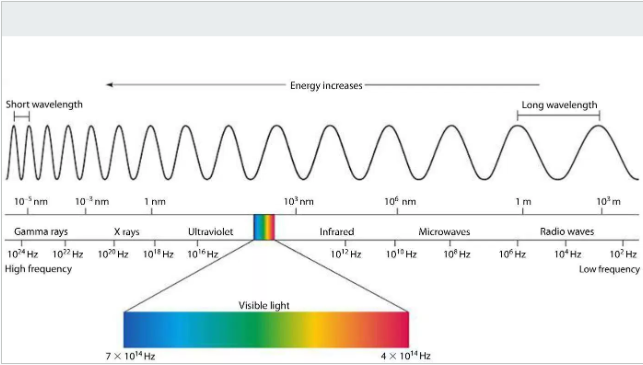
Electron Configurations
Describes the arrangement of electrons in atoms. Key principles include:
Aufbau Principle: Electrons fill orbitals starting from the lowest energy levels.
Pauli Exclusion Principle: No more than two electrons in an orbital, with opposite spins.
Hund's Rule: Equal energy orbitals are filled singly first.
Periodic Trends and Electron Behavior
Discusses ion configurations, the Octet Rule, and stability in terms of valence electrons.
Metalloids lose or gain electrons.
Differences in properties are linked to atomic structure and electron configuration.