Polarity
Polar molecules
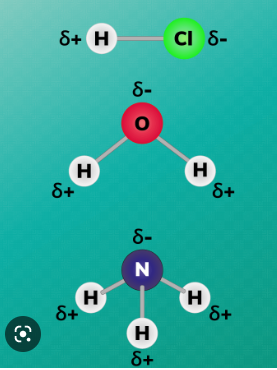
^^POLAR MOLECULES^^ = uneven distribution of charge.
1 side of a molecule is %%negative;%% one side is ==slightly positive==. Creating Poles.
- atoms bonded with a large difference in electronegativity are polar.
Non-polar molecules
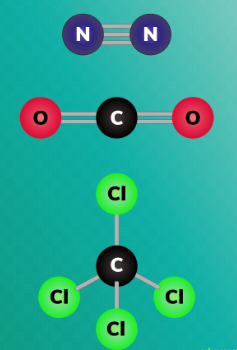
^^NON-POLAR MOLECULES^^
- no difference in charge on the outside of the molecule.
- Electrons are evenly distributed.
- Uniform charge on the outside of the molecule.
- Any atoms being bonded with itself
VSEPR Theory

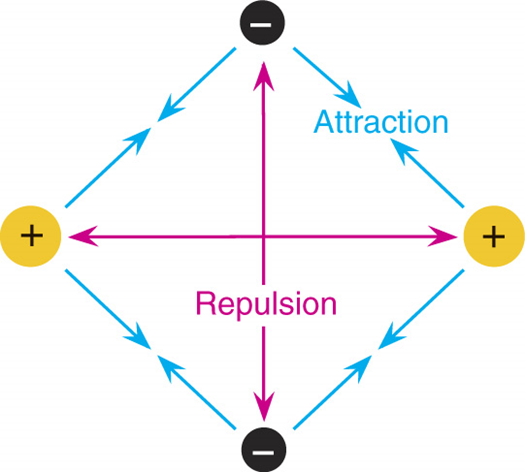
The shape of a molecule is determined by the repulsion of electron pairs.
pairs of electrons (in bonds and in lone pairs) repel each other.
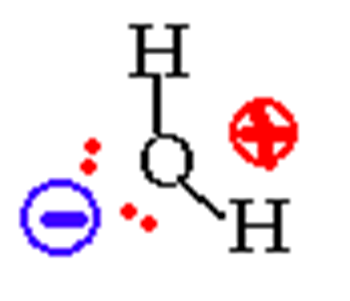
Lone pairs are stubborn and want to be at the top without anyone disturbing them.
The pairs being bonded that is the same want to be as far away from each other as possible.
- IDENTIFY THE CENTRAL ATOM
- FORM A COVALENT BOND
- DETERMINE IF THERE ARE LONE PAIRS
- DETERMINE SHAPE
Linear
- for non-polar molecules
- They should be arranged in such a manner that repulsion can be minimized (pointing in the opposite direction).
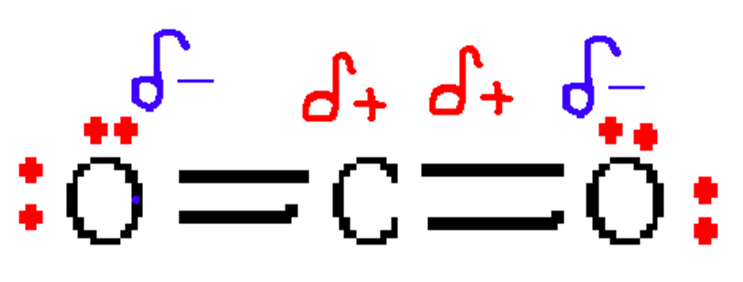
As oxygen is being bonded with carbon on both sides with no lone pairs, the shape will be linear.
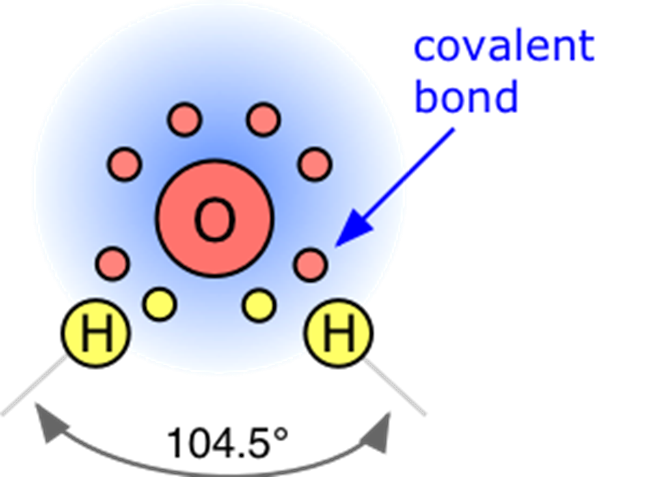
Bent
- polar molecules
- there are lone pairs that wanna be separated from the rest of the pairs, creating a bent shape.
Trigonal planar
- non-polar
- 3 molecules are attached to a central atom.
- number of lone pairs are equal on each bonding atom
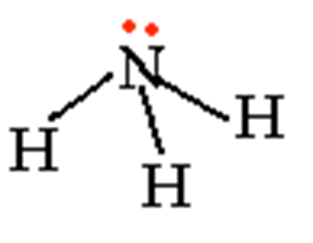
Trigonal pyramid
- Polar
- Lone pairs on the central atom cause it to rise
Tetrahedral